Abstract
Objective
Danshensu, a phenylpropanoid compound, is derived from the dry root and rhizome of Danshen (Salvia miltiorrhiza), a traditional Chinese medicinal herb. Evidence suggests that danshensu protects isolated rat hearts against ischemia/reperfusion injury by activating the protein kinase B (Akt)/extracellular signal-regulated kinase (ERK) pathway or by inhibiting autophagy and apoptosis through the activation of mammalian target of rapamycin (mTOR) signaling. Furthermore, danshensu promotes the postischemic regeneration of brain cells by upregulating the expression of brain-derived neurotrophic factor (BDNF) in the peri-infarct region. However, basic and clinical studies are needed to investigate the antidepressant effects danshensu and determine whether brain mTOR signaling and BDNF activation mediate these effects. The aforementioned need prompted us to conduct the present study.
Methods
Using a C57BL/6 mouse model, we investigated the antidepressant-like effects of danshensu and the mechanisms that mediate these effects. To elucidate the mechanisms, we analyzed the roles of Akt/ERK–mTOR signaling and BDNF activation in mediating the antidepressant-like effects of danshensu.
Results
Danshensu exerted its antidepressant-like effects by activating the α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor (AMPAR) of Akt/ERK–mTOR signaling and promoting BDNF release. Treatment with danshensu increased the level of glutamate receptor 1 phosphorylation at the protein kinase A site.
Conclusion
Our study may be the first to demonstrate that the antidepressant effects of danshensu are dependent on the activation of the AMPAR–mTOR signaling pathway, are correlated with the elevation of BDNF level, and facilitate the insertion of AMPAR into the postsynaptic membrane. This study also pioneers in unveiling the potential of danshensu against depressive disorders.
Keywords: Danshensu, α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor, Mammalian target of rapamycin, Brain-derived neurotrophic factor, Antidepressant
INTRODUCTION
Major depressive disorder, a complex and diverse condition, affects approximately 20% of all individuals during their lifetime and is a leading cause of disability worldwide [1-3]. Current therapeutic approaches for this disorder focus on the monoamine hypothesis of depression. These approaches involve the use of conventional antidepressants that inhibit the reuptake of monoamine neurotransmitters, thus increasing their synaptic levels and mediating their interaction with postsynaptic receptors. However, the efficacy of current therapeutic approaches is limited, with only approximately 50% of all patients recovering within 6 months after diagnosis. Moreover, a noticeable gradual decline has been noted in the rate of recovery. Despite regular therapy, many patients continue to experience recurrent depressive episodes. The current monoamine-based antidepressants must be used for several weeks for obtaining therapeutic benefits [4]. The delayed benefits and limited efficacy of current antidepressants pose major challenges, particularly for patients with depression who are at risk of suicide. These challenges indicate the need for next-generation antidepressants with rapid benefits and robust efficacy. Studies have demonstrated that a single subanesthetic dose of ketamine, an N-methyl-D-aspartate receptor (NMDAR) antagonist, exerts rapid and lasting antidepressant effects [5,6]. Subsequent preclinical studies have explored the mechanisms underlying the rapid antidepressant effects of ketamine, revealing that this drug promotes the activation of α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor (AMPAR) by inhibiting the glutamate NMDAR. The activated AMPAR further activates the downstream protein kinase B (Akt)/extracellular signal-regulated kinase (ERK) pathway and then the mammalian target of rapamycin (mTOR) pathway [7,8]. Rapid activation of “The AMPAR-Akt/ERK-mTOR pathway may serve as a mechanism.” that is shared by various fast-acting antidepressants [9-14]. Therefore, agents that can induce the signaling cascades that mediate the rapid antidepressant effects of ketamine—for example, the AMPAR-Akt/ERK– mTOR pathway—hold promise as fast-acting antidepressants.
Danshen, which is derived from the dry root and rhizome of Salvia miltiorrhiza, is commonly used in China for the treatment of various microcirculatory disturbance–related conditions and central nervous system disorders [15,16]. The major water-soluble component of danshen is danshensu—the phenylpropanoid compound of 3-(3,4-dihydroxyphenyl)-2-hydroxypropanoic acid. Several studies have reported the biological activities of danshensu, including enhancing microcirculation, suppressing reactive oxygen species generation, inhibiting platelet adhesion and aggregation, and protecting the myocardium from ischemia and endothelial cells from inflammation-induced injury [15,17-20]. Evidence suggests that danshensu protects isolated hearts against oxidative stress due to ischemia/reperfusion injury, indicating the cardioprotective benefits of danshensu [15]. The mechanisms underlying its cardioprotective effects include the activation of the Akt/ERK1/2/nuclear factor erythroid 2-related factor 2 (Nrf2) pathway, which strengthens the antioxidant defense system [21-23]. Danshensu reduced 6-hydroxydopamine-induced cytotoxicity and reactive oxygen species generation in PC12 cells as well as reduced dopaminergic neuronal loss in zebrafish by regulating the phosphoinositide 3-kinase (PI3K)/Akt/heme oxygenase 1 pathway [24]. This compound also mitigated motor dysfunction in animal models of Parkinson’s disease; danshensu protected neurons against rotenone toxicity, possibly by exerting antioxidative effects through the activation of the PI3K/Akt/Nrf2 pathway [25]. Furthermore, danshensu promotes the regeneration of brain cells after an ischemic event, possibly by upregulating the expression of vascular endothelial growth factor (VEGF), brain-derived neurotrophic factor (BDNF), and endothelial nitric oxide synthase in the peri-infarct region [26]. Recently, the therapeutic benefits of danshensu against mental disorders have attracted increasing research attention. Danshensu ameliorated cognitive impairment in mice with streptozotocin-induced diabetes by alleviating neuroinflammation [27]. In mice, danshensu activated the dopamine receptor D1 by inhibiting the expression of monoamine oxidase A, thereby exerting anxiolytic-like effects [28].
Evidence suggests that danshensu exerts rapid antidepressant effects by activating the mTOR signaling cascade through the activation of the Akt/ERK pathway [8]. However, these effects are yet to be explored. Moreover, the effects of danshensu on mTOR signaling downstream of the Akt/ERK pathway in the brain remain unknown. Therefore, in this study, we investigated the acute antidepressant-like effects of danshensu and measured the danshensu-induced changes in the expression levels of Akt, ERK, mTOR, and BDNF. We further used pharmacological inhibitors to determine the potential associations between the antidepressant-like effects of danshensu and the activation of AMPAR, Akt, ERK, and mTOR. In addition, we evaluated the effects of danshensu on the expression levels of BDNF and phosphorylated Akt, ERK, and mTOR with or without pretreatment with relevant inhibitors.
METHODS
Animals
Male C57BL/6 mice (age: 6–8 weeks; weight: 23–25 g) were used for this study. The mice were acclimatized at our laboratory animal center for at least 7 days before the initiation of experiments. In this period, the mice were housed under regulated environmental conditions (temperature, 23°C±1°C; humidity, 55%±5%; 12-h light/dark cycle) and were provided with food and water ad libitum. Then, they were transferred to the experimental room for further testing. Approval for all animal experiments was granted by the Institutional Animal Care and Use Committee of China Medical University, Taiwan (permit number: CMUIACUC-2021-112).
Treatment
Danshensu (MedChemExpress, Monmouth Junction, NJ, USA) was dissolved in either 0.9% normal saline or 0.5% ethanol. Desipramine (traditional tricyclic antidepressant; Sigma-Aldrich, Saint Louis, MO, USA), 2,3-dioxo-6-nitro-7-sulfamoyl-benzo(f)quinoxaline (NBQX [AMPAR inhibitor]; Sigma-Aldrich, Saint Louis, MO, USA), and rapamycin (mTOR inhibitor; TOKU-E, Bellingham, WA, USA) were dissolved in 0.9% normal saline. MK2206 (Akt inhibitor; MedChemExpress, Monmouth Junction, NJ, USA) and SL327 (ERK inhibitor; Sigma-Aldrich, Saint Louis, MO, USA) were dissolved in 0.5% ethanol. Danshensu and inhibitors were then intraperitoneally injected (separately) into the experimental mice at a dose of 0.01 mL per gram of body weight. To conduct the forced swimming test (FST), the mice were treated with saline, desipramine (20 mg/kg; positive control), or danshensu (5, 10, or 30 mg/kg) 30 min before the test. Notably, a group of mice was treated with rapamycin (20 mg/kg) or NBQX (30 mg/kg) 30 min before the administration of danshensu (10 mg/kg; this was found to be the optimal dose) or saline. Another group of mice were treated with MK2206 (60 mg/kg) or SL327 (30 mg/kg) 60 min before the administration of danshensu (10 mg/kg) or saline.
FST
The FST is a behavioral test for rodents. This test evaluates the efficacy of antidepressants by measuring the duration of immobility [29,30]. Duman’s group was the first to discover that mTOR activation in brain is required for the rapid antidepressant mechanism of ketamine soon after that, mTOR became a major target in the studies of depression and antidepressant action [9]. A day before the 5-min formal FST, the mice were subjected to a 15-min swimming session, which was conducted in a water-filled transparent cylindrical tank, following established protocols. For the formal FST, mice were placed in a cylindrical container (height: 40 cm; diameter: 20 cm) filled with water (depth: 10 cm) at a temperature of 25°C for a duration of 5 minutes. Their behaviors in this test were captured by a digital camera, and the duration of immobility was automatically measured using EthoVision (Noldus Information Technology, Wageningen, The Netherlands).
Open field test
To rule out the possibility of false-positive results in the FST, we evaluated the locomotor activity of the mice through the open field test (OFT) [30]. In this experiment, the mice were housed in a plastic enclosure containing a 60-cm×60-cm base, enclosed by walls standing at a height of 50 cm. Following treatment, the mice were given a 5-minute period to explore the cage. Their locomotion was captured via a digital camera, and the overall movement within the 5-minute timeframe was quantified using EthoVision.
Western blotting
After the FST, four mice were euthanized. The hippocampus was excised, dried with liquid nitrogen, and stored at -80°C for further analyses. Subsequently, each tissue sample was homogenized and mixed with 250 μL of lysis buffer (per tube). Subsequently, the mixture underwent centrifugation at 15,000 rpm for 15 minutes at 4°C. The resulting supernatant was then transferred to a fresh centrifuge tube, supplemented with sample buffer, and heated at 100°C for 7 minutes. Next, the mixture was cooled in a cooling machine set at 4°C and stored at -80°C. Western blotting was performed to measure the expression levels of target proteins in the hippocampus of the treated mice. For western blotting, we followed the protocol described by Chen et al. [11].
Statistical analysis
The normality of data distribution was assessed using the Shapiro–Wilk test. The results of the behavioral experiments were analyzed through one-way analysis of variance, followed by the Tukey post hoc test. The results of western blotting were analyzed using the Kruskal–Wallis test, followed by the Conover–Iman post hoc test. All tests were two-tailed, and a p-value of <0.05 indicated statistical significance. Parametric data were analyzed using SPSS (version 12.0; SPSS Statistics, SPSS Inc., Chicago, IL, USA), whereas nonparametric data were analyzed using Microsoft Office Excel® (version 2019; Microsoft Corp., Redmond, WA, USA; add in: Real Statistics Resource Pack).
RESULTS
Effects of danshensu on the duration of immobility in the FST and the levels of phosphorylated mTOR, Akt, and ERK
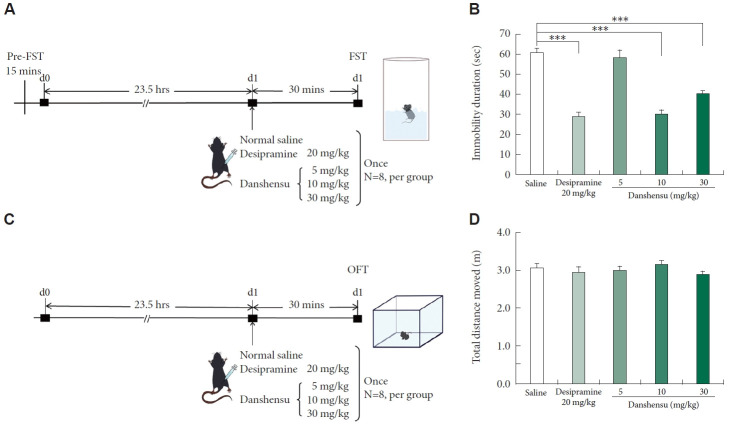
The FST is commonly conducted to determine the antidepressant effects of an agent [29,30]. We conducted this test to evaluate the effects of danshensu on immobility in the mice (Figure 1A). To rule out false-positive results in the FST, we also performed the OFT (Figure 1C) [30]. Treatment with 10 and 30 mg/kg danshensu significantly reduced the immobility duration by 31% and 20%, respectively, compared with the results observed in the control mice (Figure 1B). Furthermore, a single dose of desipramine (20 mg/kg) significantly reduced the immobility duration. Neither danshensu nor desipramine affected the locomotor activity of the mice (Figure 1D). These findings suggest that danshensu exerts antidepressant-like effects.
Figure 1.
Performance of study groups in the FST and OFT. In this study, male C57BL/6 mice were subjected to various treatments: normal saline, desipramine (20 mg/kg; positive control), and danshensu (5, 10, or 30 mg/kg). The placebo or drug was administered through intraperitoneal injection. Subsequently, the behaviors of the treated mice were assessed through the FST (A) and OFT (C). For the FST, the mice were treated with saline, desipramine (20 mg/kg), or danshensu (5, 10, or 30 mg/kg) 30 min before the test, which was conducted 15 min after the initial treatment. Treatment with danshensu and desipramine led to significant reductions in the duration of immobility during the FST (B; analysis of variance; F[4,35]=35.760; p<0.001; N=8 per group). For the OFT, the mice were treated with saline, desipramine (20 mg/kg), or danshensu (5, 10, or 30 mg/kg) 30 min before the test. The total distance traveled, which indicates the level of locomotor activity, did not differ significantly between the treatment (danshensu- and desipramine-treated) groups and the saline-treated group (D; analysis of variance; F[4,35]=0.741; p>0.05; N=8 per group). Compared with saline treatment: Tukey post hoc analysis. Data are presented in terms of the mean±standard error of the mean values. ***p<0.001. FST, forced swimming test; OFT, open field test.
Previous research has shown that danshensu can enhance Akt and ERK signaling [21-23], and that the rapid antidepressant effects of ketamine rely on Akt/ERK downstream mTOR activation [8]. In this study, we investigated whether danshensu promotes the activation of the Akt/ERK–mTOR pathway. Through western blotting, we measured the levels of phosphorylated Akt, ERK, and mTOR after danshensu treatment Figure 2A). The activation of mTOR exhibited a dose-dependent pattern (Figure 2B). Specifically, at higher doses (10 and 30 mg/kg), but not at a lower dose (5 mg/kg), danshensu promoted the activation of mTOR. This pattern was consistent with the reduction in the immobility duration during the FST. Furthermore, a significant increase was noted in the level of phosphorylated Akt across all doses of danshensu (5, 10, and 30 mg/kg) (Figure 2C). Treatment with 10 and 30 mg/kg danshensu resulted in significant increases in the level of phosphorylated ERK; however, treatment with 5 mg/kg danshensu exerted no significant effects (Figure 2D). Although desipramine exerted no significant effect on the levels of phosphorylated mTOR or ERK, it altered the level of phosphorylated Akt.
Figure 2.
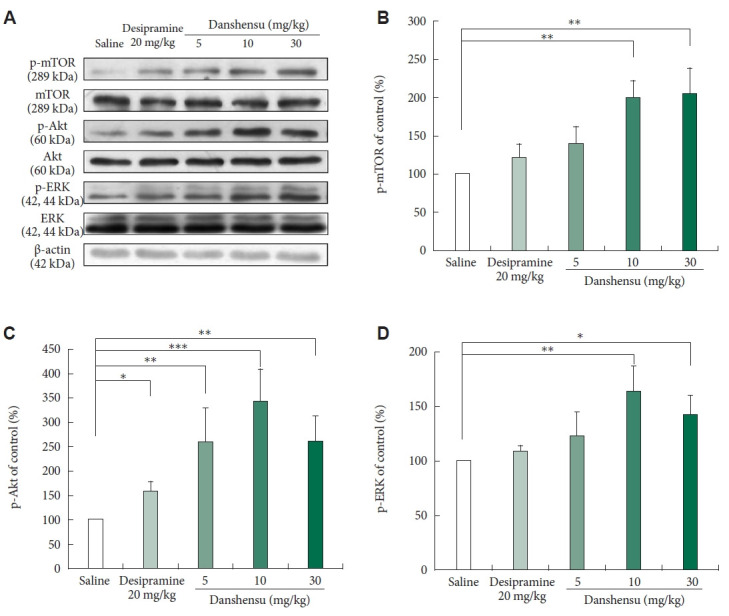
Effects of danshensu and desipramine on the levels of phosphorylated mTOR, Akt, and ERK. We evaluated the effects of different intraperitoneal doses of danshensu (5, 10, and 30 mg/kg) and a single dose of desipramine (20 mg/kg) on the activation of mTOR, Akt, and ERK in the hippocampus of mice. For this, we measured the levels of phosphorylated mTOR, Akt, and ERK through western blotting (A). Densitometric analyses of the blots (normalized to β-actin levels) confirmed the increased levels of phosphorylated mTOR in mice treated with 10 or 30 mg/kg danshensu but not in those treated with 5 mg/kg danshensu or desipramine (B). Treatment with danshensu dose-dependently increased the level of phosphorylated Akt in the mouse hippocampus (C). A significant increase was noted in the level of phosphorylated ERK after treatment with 10 or 30 mg/kg danshensu (D). No between-group differences were noted in the level of total mTOR, Akt, or ERK. Between-group comparisons were performed using the nonparametric Kruskal–Wallis test, followed by the Conover–Iman post hoc test. N=4 per group. Data are presented in terms of the mean±standard error of the mean values. *p<0.05; **p<0.01; ***p<0.001. p-mTOR, phosphorylated mTOR; mTOR, mammalian target of rapamycin; p-Akt, phosphorylated Akt; Akt, protein kinase B; p-ERK, phosphorylated ERK; ERK, extracellular signal-regulated kinase.
Treatment with 10 mg/kg danshensu resulted in significant increases in the levels of phosphorylated mTOR, Akt, and ERK; danshensu exerted antidepressant effects. Therefore, this dose was used in the subsequent experiments.
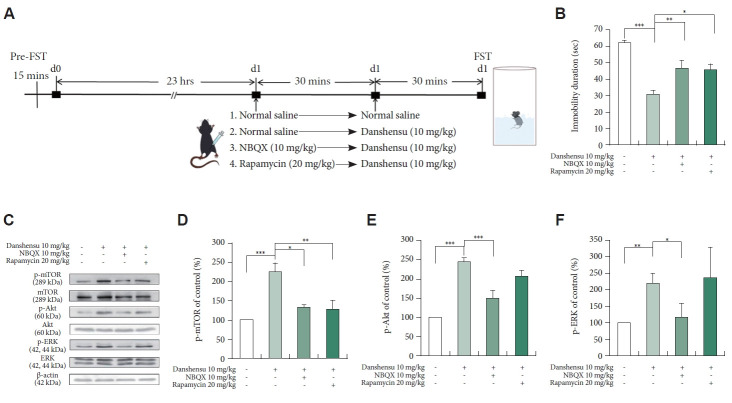
Effects of NBQX and rapamycin on the danshensu-induced reduction in immobility in the FST
To determine the roles of AMPAR and mTOR in mediating the antidepressant-like effects of danshensu, we treated the mice with NBQX or rapamycin before danshensu treatment and then conducted the FST. The timeline for pretreatment with inhibitors is depicted in Figure 3A. The danshensu-induced reduction in immobility was prevented by NBQX and rapamycin (Figure 3B). This finding confirms that the antidepressant-like effects of danshensu are dependent on the activation of mTOR and AMPAR.
Figure 3.
Effects of pretreatment with NBQX and rapamycin. We evaluated the effects of pretreatment with NBQX (10 mg/kg) and rapamycin (20 mg/kg) on the duration of immobility during the FST and the levels of phosphorylated mTOR, Akt, and ERK in the hippocampus of mice treated with danshensu (10 mg/kg). The timeline for the FST under different treatment conditions is shown (A). Pretreatment with NBQX and rapamycin prevented the danshensu-induced reduction in the duration of immobility during the FST (B; analysis of variance; F[3,28]=14.831; p<0.001; N=8 per group). Compared with saline treatment: Tukey post hoc analysis. Western blotting was performed to evaluate the activation of mTOR, Akt, and ERK (C). Densitometric analyses of the blots (normalized to β-actin levels) confirmed the increased levels of phosphorylated mTOR (D), Akt (E), and ERK (F) in mice treated with danshensu. Pretreatment with NBQX prevented the danshensu-induced increases in the levels of phosphorylated mTOR (D), Akt (E), and ERK (F). Pretreatment with rapamycin blocked the danshensu-induced increase in the level of phosphorylated mTOR (D) but not the level of phosphorylated Akt (E) or ERK (F). N=4 per group. Between-group comparisons were performed using the nonparametric Kruskal–Wallis test, followed by the Conover–Iman post hoc test. Data are presented in terms of the mean±standard error of the mean values. *p<0.05; **p<0.01; ***p<0.001. FST, forced swimming test; NBQX, 2,3-dioxo-6-nitro-7-sulfamoyl-benzo(f)quinoxaline; p-mTOR, phosphorylated mTOR; mTOR, mammalian target of rapamycin; p-Akt, phosphorylated Akt; Akt, protein kinase B; p-ERK, phosphorylated ERK; ERK, extracellular signal-regulated kinase.
Effects of NBQX and rapamycin on the danshensu-induced activation of mTOR, Akt, and ERK
To determine the roles of AMPAR and mTOR in the danshensu-induced activation of mTOR signaling-related proteins, we measured the levels of phosphorylated mTOR, Akt, and ERK in the mice treated with danshensu with or without pretreatment with NBQX or rapamycin. A single injection of danshensu significantly increased the levels of phosphorylated mTOR, Akt, and ERK (Figure 3C); however, the levels of total mTOR, Akt, and ERK remained unchanged. Pretreatment with NBQX prevented the danshensu-induced increases in the levels of phosphorylated mTOR (Figure 3D), Akt (Figure 3E), and ERK (Figure 3F), indicating that danshensu-induced activation of AMPAR is upstream of the activation of Akt, ERK, and mTOR. However, pretreatment with rapamycin prevented the danshensu-induced increase in the level of phosphorylated mTOR (Figure 3D) but did not attenuate phosphorylated Akt or ERK (Figure 3E and F); this finding suggests that danshensu-induced mTOR activation is downstream of the activation of Akt and ERK.
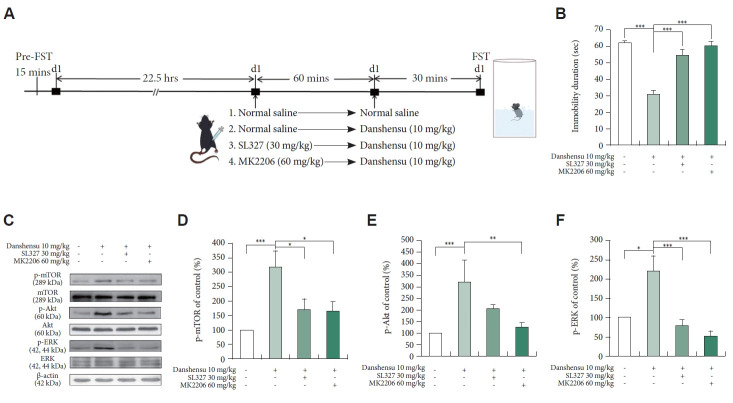
Effects of SL327 and MK2206 on the danshensu-induced reduction in immobility in the FST
To determine the roles of ERK and Akt in mediating the antidepressant effects of danshensu, we treated the mice with SL327 or MK2206 before danshensu treatment and then conducted the FST. The timeline for pretreatment with inhibitors is depicted in Figure 4A. The antidepressant-like effects of danshensu were blocked by SL327 and MK2206 (Figure 4B). However, no significant difference was noted between the SL327- or MK2206-treated mice and the saline-treated mice. These findings confirm that the antidepressant-like effects of danshensu are dependent on the activation of ERK and Akt.
Figure 4.
Effects on pretreatment with SL327 and MK2206. We evaluated the effects of SL327 (30 mg/kg) and MK2206 (60 mg/kg) on the duration of immobility during the FST and the levels of phosphorylated mTOR, Akt, and ERK in the hippocampus of mice treated with danshensu (10 mg/kg). The timeline for the FST under different treatment conditions is shown (A). Pretreatment with SL327 and MK2206 prevented the danshensu-induced reduction in the duration of immobility during the FST (B; analysis of variance; F[3,28]=31.316; p<0.001; N=8 per group). Compared with saline treatment: Tukey post hoc analysis. Western blotting was performed to evaluate the activation of mTOR, Akt, and ERK (C). Densitometric analyses of the blots (normalized to β-actin levels) confirmed the increased levels of phosphorylated mTOR (D), Akt (E), and ERK (F) in mice treated with danshensu. Pretreatment with SL327 and MK2206 prevented the danshensu-induced increase in the level of phosphorylated mTOR (D). Pretreatment with MK2206, but not SL327, prevented the danshensu-induced increase in the level of phosphorylated Akt (E). Furthermore, pretreatment with SL327, but not MK2206, prevented the danshensu-induced increase in the level of phosphorylated ERK (F). No significant between-group difference was observed in the level of total Akt, ERK, or mTOR. N=4 per group. Between- group comparisons were performed using the nonparametric Kruskal–Wallis test, followed by the Conover–Iman post hoc test. Data are presented in terms of the mean±standard error of the mean values. *p<0.05; **p<0.01; ***p<0.001. FST, forced swimming test; p-mTOR, phosphorylated mTOR; mTOR, mammalian target of rapamycin; p-Akt, phosphorylated Akt; Akt, protein kinase B; p-ERK, phosphorylated ERK; ERK, extracellular signal-regulated kinase.
Effects of SL327 and MK2206 on the danshensu-induced activation of mTOR, Akt, and ERK
To determine the roles of Akt and ERK in the danshensu-induced activation of the AMPAR–mTOR pathway, we measured the levels of phosphorylated mTOR, Akt, and ERK in the mice treated with danshensu with or without pretreatment with SL327 or MK2206. Figure 4C depicts the target protein bands obtained after western blotting. Pretreatment with SL327 prevented the danshensu-induced increases in the levels of phosphorylated mTOR (Figure 4D) and ERK (Figure 4F), but not that of phosphorylated Akt (Figure 4E). This result indicated that ERK activation is upstream of mTOR activation. However, pretreatment with MK2206 prevented the danshensu-induced increases in the levels of phosphorylated mTOR (Figure 4D), Akt (Figure 4E), and ERK (Figure 4F), suggesting that the activation of Akt and ERK is upstream of mTOR activation. Together, these findings imply that, similar to ketamine [8], danshensu exerts its effects by activating the AMPAR-Akt/ERK–mTOR pathway.
Danshensu-induced changes in the patterns of AMPAR insertion into the cell membrane
AMPAR has been implicated in mediating the antidepressant effects of new-generation glutamate-based antidepressants [11-13,31-33]. In this study, we investigated the effects of danshensu on the activated forms of the glutamate A1 (GluA1) subunit of AMPAR at its protein kinase A (GluA1ser845) [34-36] and protein kinase C (GluA1ser831) [37,38] sites; thus, we assessed the danshensu-mediated changes in the patterns of AMPAR insertion into the cell membrane. We found that the levels of phosphorylated GluA1ser845 and GluA1ser831 were increased after treatment with 10 and 30 mg/kg danshensu, but not after treatment with 5 mg/kg danshensu or desipramine (Figure 5A-C). Pretreatment with NBQX, but not rapamycin, prevented the danshensu-induced increases in the levels of phosphorylated GluA1ser845 and GluA1ser831 (Figure 5D-F). Furthermore, pretreatment with SL327 or MK2206 significantly prevented the danshensu-induced increases in the levels of phosphorylated GluA1ser845 and GluA1ser831 (Figure 5H and I). However, the level of total GluA1 remained unchanged across treatments (Figure 5A, D, and G). Our findings suggest that danshensu promotes the insertion of AMPAR into the cell membrane—a process that is dependent on AMPAR activation.
Figure 5.
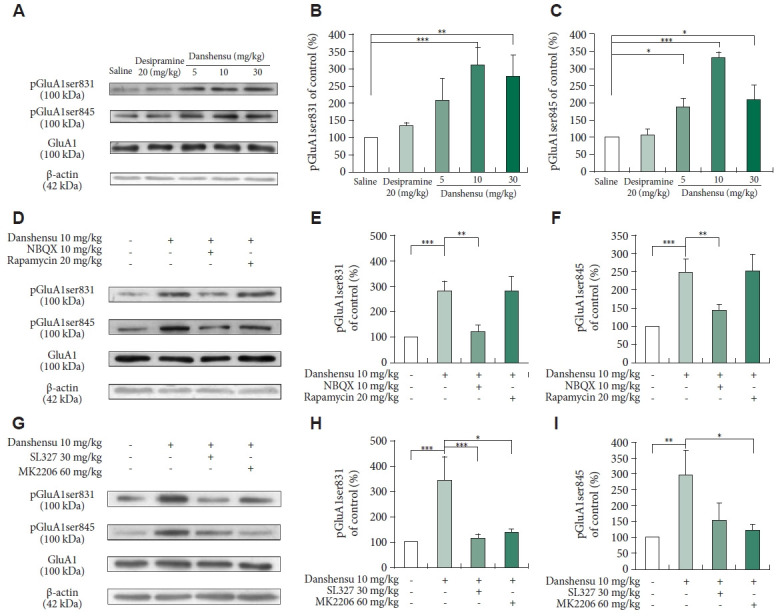
Post-treatment changes in the levels of phosphorylated GluA1ser845 and GluA1ser831. We evaluated the activation of GluA1ser845 and GluA1ser831 in the hippocampus of mice treated with saline, desipramine (20 mg/kg), or danshensu (5, 10, or 30 mg/kg). Western blotting was performed to measure the levels of phosphorylated GluA1ser845 and GluA1ser831 in mice treated with 10 mg/kg danshensu (A), those pretreated with NBQX (10 mg/kg) or rapamycin (20 mg/kg) (D), and those pretreated with SL327 (30 mg/kg) or MK2206 (60 mg/kg) (G). Treatment with 10 and 30 mg/kg danshensu significantly promoted the activation of GluA1ser831 (B) and GluA1ser845 (C). Densitometric analyses of the blots (normalized to β-actin levels) revealed that the levels of phosphorylated pGluA1ser831 (E) and pGluA1ser845 (F) were increased in mice treated with danshensu; these increases were blocked by pretreatment with NBQX but not rapamycin (D-F). Notably, pretreatment with SL327 or MK2206 block the danshensu-induced increase in the level of phosphorylated GluA1ser845 or GluA1ser831 (G-I). N=4 per group. Between-group comparisons were performed using the nonparametric Kruskal–Wallis test, followed by the Conover–Iman post hoc test. Data are presented in terms of the mean±standard error of the mean values. *p<0.05; **p<0.01; ***p<0.001. GluA1, glutamate A1 subunit of AMPAR; AMPAR, α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor; NBQX, 2,3-dioxo-6-nitro- 7-sulfamoyl-benzo(f)quinoxaline.
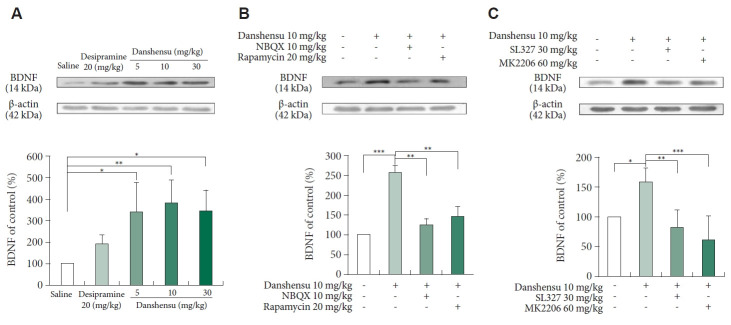
Effects of danshensu on BDNF expression
BDNF, a neurotrophin, plays pivotal roles in neuronal development, differentiation, and survival, as well as emotional regulation [39]. ERK signaling–induced BDNF release triggers a rapid antidepressant response [40]. To evaluate the effects of danshensu on BDNF expression, we measured the level of BDNF in the mice subjected to various treatments. Treatment with danshensu (10 and 30 mg/kg), but not desipramine, significantly upregulated BDNF expression in the mouse hippocampus (Figure 6A). The observed trend was consistent with the antidepressant-like effects of danshensu and the danshensu-induced activation of mTOR. Notably, pretreatment with NBQX, rapamycin, SL327, and MK2206 prevented the danshensu-induced increase in the BDNF level (Figure 6B and C).
Figure 6.
Post-treatment changes in the expression level of BDNF. We evaluated the expression of BDNF in the hippocampus of mice treated with saline, desipramine (20 mg/kg) or danshensu (5, 10, or 30 mg/kg). Western blotting was performed to measure the level of BDNF expression in mice treated with 10 mg/kg danshensu (A), those pretreated with NBQX (10 mg/kg) or rapamycin (20 mg/kg) (B), and those pretreated with SL327 (30 mg/kg) or MK2206 (60 mg/kg) (C). Treatment with 10 and 30 mg/kg danshensu significantly upregulated the expression of BDNF (A). Densitometric analyses of the blots (normalized to β-actin levels) revealed that the level of BDNF expression (B) was increased in mice treated with danshensu; these increases were blocked by pretreatment with NBQX and rapamycin (B). Furthermore, pretreatment with SL327 and MK2206 prevented the danshensu-induced upregulation of BDNF expression (C). N=4 per group. Between-group comparisons were performed using the nonparametric Kruskal–Wallis test, followed by the Conover–Iman post hoc test. Data are presented in terms of the mean±standard error of the mean values. *p<0.05; **p<0.01; ***p<0.001. BDNF, brain-derived neurotrophic factor; NBQX, 2,3-dioxo-6-nitro-7-sulfamoyl-benzo(f)quinoxaline.
DISCUSSION
In this study, we investigated whether danshensu exerts antidepressant-like effects by promoting Akt/ERK signaling downstream of mTOR signaling in the hippocampus. For this, we evaluated the effects of the compound on the Akt/ERK pathway in the brain, which has been demonstrated to be promoted by danshensu [21-25]. In the present study, treatment with 10 and 30 mg/kg danshensu promoted the Akt/ERK pathway downstream of mTOR activation. By using pharmacological inhibitors, we confirmed that the antidepressant effects of danshensu were dependent on the activation of the AMPAR-Akt/ERK–mTOR pathway (Figure 7); the underlying mechanism is similar to the mechanism involved in mediating the effects of ketamine or echinacoside [8,12]. Danshensu facilitated the insertion of AMPAR into the cell membrane and upregulated BDNF expression. These findings suggest that danshensu can be used for the rapid treatment of depression.
Figure 7.
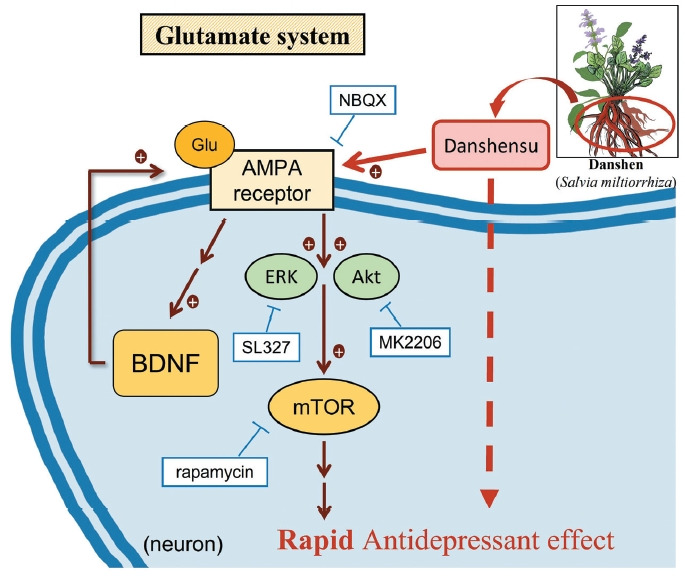
The cellular mechanisms responsible for the antidepressant- like effects of danshensu in postsynaptic glutamatergic neurons. Danshensu, an active compound derived from danshen, exerts antidepressant-like effects by activation of the AMPAR and promoting Akt/ERK signaling downstream of mTOR signaling in the postsynaptic neuronal membrane. Through the use of pharmacological inhibitors such as NBQX (an AMPAR inhibitor), rapamycin (an mTOR inhibitor), MK2206 (an Akt inhibitor), and SL327 (an ERK inhibitor), it was established that the antidepressant-like effects of danshensu depend on the activation of the AMPAR–Akt/ERK–mTOR pathway. In essence, the increase in glutamate binding to the AMPAR in the postsynaptic neuronal membrane triggers the activation of downstream factors, Akt, ERK, mTOR, and the release of BDNF, ultimately producing a positive regulatory effect and leading to the antidepressant-like outcome. NBQX, 2,3-dioxo-6-nitro-7-sulfamoyl-benzo(f)quinoxaline; AMPA, α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid; ERK, extracellular signal-regulated kinase; Akt, protein kinase B; BDNF, brain-derived neurotrophic factor; mTOR, mammalian target of rapamycin; AMPAR, α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor.
Danshensu can cross the blood-brain barrier [41]. Administering single or repeated doses of danshensu does not induce toxicity [42]. Danshensu has antioxidative, anti-inflammatory, and neuroprotective properties, which make it a promising therapeutic agent for neurodegenerative conditions such as Alzheimer’s disease and Parkinson’s disease [21-25,43,44]. Given the strong correlations between oxidative stress, inflammation, and depression, danshensu may also exert antidepressant-like effects by alleviating oxidative stress and inflammation [45,46]. Danshensu can activate the Akt/ERK pathway in the brain [24,25], which then induces downstream mTOR signaling. This convergence of signaling cascades is closely associated with the pathophysiology of depression and has been implicated for mediating the rapid antidepressant effects of certain drugs [8,47-49]. Thus, in this study, we evaluated the potential effects of danshensu against depression and explored the underlying mechanisms. Treatment with 10 and 30 mg/kg danshensu considerably reduced the immobility duration in the FST but exerted no effects on locomotor activity, as noted in the OFT. These findings confirm the antidepressant-like properties of danshensu. The reduction in the immobility duration was more prominent after treatment with 10 and 30 mg/kg danshensu than after treatment with 5 mg/kg danshensu. Treatment with 10 mg/kg danshensu substantially increased the levels of BDNF and phosphorylated Akt, ERK, and mTOR. Danshensu exerted nonlinear dose-response effects on the performance of the mice in the FST, which is consistent with the changes in the levels of BDNF and phosphorylated mTOR.
We elucidated the mechanisms underlying the antidepressant-like effects of danshensu. To investigate whether these effects are mediated directly by mTOR activation, we treated the mice with rapamycin before danshensu treatment. The findings revealed that rapamycin blocked the antidepressant effects of danshensu by inhibiting mTOR, confirming that these effects are mediated by mTOR activation. In vitro and in vivo studies have explored the effects of danshensu on mTOR signaling after cerebral and cardiac ischemia/reperfusion injuries [50-56]. Some of the aforementioned studies have indicated that danshensu reduces autophagy during cardiac ischemia/reperfusion injury by enhancing mTOR signaling and suppressing autophagosome formation [51,56]. In rat models of cerebral ischemia/reperfusion injury, treatment with danshensu (7.5, 15, or 30 mg/kg) significantly reduced necrosis and apoptosis by increasing the levels of phosphorylated AKT1 and its downstream regulator (phosphorylated mTOR) [25,52]. Furthermore, danshensu enhanced bEnd.3 cell proliferation and angiogenesis by upregulating VEGF expression through the PI3K/Akt/mTOR pathway; simultaneously, it mitigated behavioral deficits in mice affected by cerebral ischemic injury [54]. Although evidence suggests that danshensu promotes the PI3K/Akt/mTOR pathway in the brain of mice affected by cerebral ischemic injury, no in vitro or in vivo study has investigated the antidepressant-like effects of danshensu on Akt/ERK signaling downstream of mTOR activation in the brain. Thus, our study is the first in vivo investigation to demonstrate that the antidepressant-like effects of danshensu significantly promote the activation of the hippocampal Akt/ERK pathway downstream of mTOR signaling. In our study, rapamycin blocked the danshensu-induced activation of mTOR, confirming the integral role of hippocampal mTOR activation in mediating the antidepressant effects of danshensu. These findings are consistent with those of studies indicating the role of mTOR activation in mediating the effects of antidepressants [8,47-49]. The pioneering study of Duman’s research team identified the crucial role of mTOR activation in the brain in the rapid antidepressant effects of ketamine [9]. Since this discovery, mTOR has been a key topic in studies on depression and antidepressants. Subsequent study has validated the pivotal function of mTOR in the pathophysiology of depression. Conventional antidepressants such as escitalopram, fluoxetine, and paroxetine promote mTOR activation [57]. Our findings highlight the rapid and beneficial effects of danshensu against depression.
In the present study, pretreatment with NBQX prevented the danshensu-mediated increases in the levels of phosphorylated mTOR, Akt, and ERK. Notably, Akt and ERK are upstream kinases that regulate the activation of mTOR. Thus, the AMPAR-induced activation of mTOR signaling may mediate the antidepressant-like effects of danshensu. Furthermore, treatment with danshensu increased the hippocampal levels of Akt and ERK. These findings corroborate those of other studies [25,52,54]. The inhibition of Akt or ERK expression suppressed the antidepressant-like effects of danshensu; therefore, these effects are mediated through Akt and ERK. In the mice treated with danshensu, Akt/ERK signaling occurred downstream of AMPAR activation and upstream of mTOR signaling. Collectively, our findings suggest that the antidepressant effects of danshensu are mediated by the activation of the AMPAR-Akt/ERK–mTOR pathway. These findings are consistent with the mechanisms underlying the effects of fast-acting antidepressants [8,9,12,49].
AMPAR, a subtype of ionotropic glutamate receptor, consists of the GluA2 subunit along with either GluA1, GluA3, or GluA4 subunit [58]. GluA1 plays a vital role in synaptic plasticity [59]. The trafficking of AMPAR is essential for antidepressant and neuroprotective effects [11,31-33,60]. To elucidate the effects of danshensu on AMPAR trafficking, we examined the phosphorylation status of AMPAR subunits, particularly GluA1ser831 and GluA1ser845. These phosphorylation events serve as reliable markers of the insertion of GluA1 into the cell membrane [34-38]. We found that treatment with danshensu increased the levels of phosphorylated GluA1ser845 and GluA1ser831. Therefore, danshensu can promote the integration of AMPAR into synaptic sites, thereby increasing postsynaptic AMPAR levels. This, in turn, increases the AMPAR/NMDAR ratio. The increase in AMPAR throughput induces the activation of mTOR, thus mediating the antidepressant-like effects of danshensu. These findings confirm the antidepressant properties of danshensu and suggest the similar mechanism for danshensu and ketamine, which also promotes the insertion of GluA1 into the postsynaptic membrane [10,61]. The increase in AMPAR throughput after treatment with danshensu is also noted after treatment with many other antidepressants; thus, this increase may be a common convergent response in antidepressants [62,63].
Danshensu confers neuroprotective benefits against Parkinson’s disease and Alzheimer’s disease [24,25]. The beneficial effects are mediated through several pathways, including the PI3K/Akt/Nrf2 pathway and the Nrf2-induced upregulation of heme oxygenase 1 and glutathione cysteine ligase (regulatory and modifier subunits) [24,25]. However, the precise mechanisms underlying these benefits remain unclear. Our study is the first to demonstrate that danshensu induces the insertion of AMPAR into the cell membrane. These findings indicate an alternative mechanism through which danshensu exerts its neuroprotective effects. After facilitating AMPAR insertion, danshensu regulates the expression of synaptic function-related proteins, thereby conferring protection against neurosynaptic deficits and cellular damage. Hence, danshensu may confer neuroprotection against various diseases, particularly those characterized by abnormal synaptic plasticity.
A study reported that danshensu upregulates the expression of both VEGF and BDNF, thereby promoting poststroke neurogenesis and angiogenesis in the brain [26]. Beyond its neuroprotective role, BDNF holds a pivotal position in depression and the therapeutic mechanisms of antidepressant [64]. To investigate whether the antidepressant-like effects of danshensu are mediated through BDNF, we measured BDNF expression. Our findings revealed that danshensu effectively upregulated BDNF expression aligns with prior research findings and bearing relevance to its antidepressant effect. Notably, the effects of danshensu on BDNF expression are similar to those of ketamine. Treatment with the inhibitors of AMPAR, Akt, ERK, and mTOR effectively blocked the danshensu-induced upregulation of BDNF expression. Therefore, in the danshensu-treated mice, the upregulation of BDNF expression occurred downstream of the activation of the AMPAR-Akt/ERK–mTOR pathway. This mechanism differs from that underlying the effects of ketamine. Ketamine promotes glutamate release while inhibiting NMDAR activation, which is responsible for inducing AMPAR—another glutamate receptor. The results leads to BNDF release, which activates mTOR [64]. Our findings indicate that BDNF plays a key role in mediating the antidepressant-like effects of danshensu and the danshensu-induced activation of the AMPAR-Akt/ERK–mTOR pathway. Nonetheless, further mechanistic studies are needed to fully understand the indicated interactions.
The depression-like phenotype involves not only the hippocampus but also the prefrontal cortex, nucleus accumbens, and amygdala [65-69]. One limitation of our study is that we only investigated the effects of danshensu treatment on mTOR signaling and BDNF release in the hippocampus. Further research is necessary to explore the impact of danshensu treatment on mTOR signaling in other brain regions.
In conclusion, our findings confirm that danshensu exerts antidepressant-like effects are achieved via the activation of the AMPAR-Akt/ERK–mTOR pathway, which mirrors the mechanism observed with ketamine. Danshensu also promotes the insertion of AMPAR into the cell membrane and upregulates BDNF expression, thereby conferring neuroprotective effects (Figure 7). Our findings suggest that danshensu may modulate the hippocampal glutamatergic pathway and can exert rapid antidepressant-like effects.
Acknowledgments
The authors would like to thank all the participants of this study.
Footnotes
Availability of Data and Material
All data generated or analyzed during the study are included in this published article.
Conflicts of Interest
The authors have no potential conflicts of interest to disclose.
Author Contributions
Conceptualization: I-Hua Wei, Han-Wen Chuang, Chih-Chia Huang. Data curation: I-Hua Wei, Han-Wen Chuang, Chih-Chia Huang. Formal analysis: Chih-Chia Huang, Yen-Yu Kuo. Funding acquisition: I-Hua Wei, Chih-Chia Huang. Methodology: Kuang-Ti Chen, Yen-Yu Kuo, Jou-Hua Ren, Tse-Yen Wang, Mang-Hung Tsai, Po-Ting Chen. Project administration: I-Hua Wei, Han-Wen Chuang. Writing—original draft: I-Hua Wei, Han-Wen Chuang, Chih-Chia Huang. Writing—review & editing: I-Hua Wei, Han-Wen Chuang, Chih-Chia Huang.
Funding Statement
This work was supported by the Ministry of Science and Technology, Taiwan (MOST 110-2320-B-039-037, MOST 111-2320-B-039-037, and MOST 111-2314-B-894-001), National Science and Technology Council, Taiwan (NSTC 112-2320-B-039-038-MY3), and China Medical University, Taiwan (CMU110-MF-87, CMU111-MF-48, CMU112-MF-40). The funding organizations had no role in the study design, data collection, data analysis, or interpretation, nor authorship of the manuscript.
REFERENCES
- 1.Ménard C, Hodes GE, Russo SJ. Pathogenesis of depression: insights from human and rodent studies. Neuroscience. 2016;321:138–162. doi: 10.1016/j.neuroscience.2015.05.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Filatova EV, Shadrina MI, Slominsky PA. Major depression: one brain, one disease, one set of intertwined processes. Cells. 2021;10:1283. doi: 10.3390/cells10061283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Allahyari E. Differential item functioning analysis on the Geriatric Depression Scale-15: An iterative hybrid ordinal logistic regression. Biomedicine (Taipei) 2021;11:23–34. doi: 10.37796/2211-8039.1098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Keller MB, Lavori PW, Mueller TI, Endicott J, Coryell W, Hirschfeld RM, et al. Time to recovery, chronicity, and levels of psychopathology in major depression. A 5-year prospective follow-up of 431 subjects. Arch Gen Psychiatry. 1992;49:809–816. doi: 10.1001/archpsyc.1992.01820100053010. [DOI] [PubMed] [Google Scholar]
- 5.Krystal JH, Karper LP, Seibyl JP, Freeman GK, Delaney R, Bremner JD, et al. Subanesthetic effects of the noncompetitive NMDA antagonist, ketamine, in humans. Psychotomimetic, perceptual, cognitive, and neuroendocrine responses. Arch Gen Psychiatry. 1994;51:199–214. doi: 10.1001/archpsyc.1994.03950030035004. [DOI] [PubMed] [Google Scholar]
- 6.Zarate CA, Jr, Singh JB, Carlson PJ, Brutsche NE, Ameli R, Luckenbaugh DA, et al. A randomized trial of an N-methyl-D-aspartate antagonist in treatment-resistant major depression. Arch Gen Psychiatry. 2006;63:856–864. doi: 10.1001/archpsyc.63.8.856. [DOI] [PubMed] [Google Scholar]
- 7.Gündüz D, Troidl C, Tanislav C, Rohrbach S, Hamm C, Aslam M. Role of PI3K/Akt and MEK/ERK signalling in cAMP/Epac-mediated endothelial barrier stabilisation. Front Physiol. 2019;10:1387. doi: 10.3389/fphys.2019.01387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Li N, Lee B, Liu RJ, Banasr M, Dwyer JM, Iwata M, et al. mTOR-dependent synapse formation underlies the rapid antidepressant effects of NMDA antagonists. Science. 2010;329:959–964. doi: 10.1126/science.1190287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Duman RS, Shinohara R, Fogaça MV, Hare B. Neurobiology of rapidacting antidepressants: convergent effects on GluA1-synaptic function. Mol Psychiatry. 2019;24:1816–1832. doi: 10.1038/s41380-019-0400-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wei IH, Chen KT, Tsai MH, Wu CH, Lane HY, Huang CC. Acute amino acid d-serine administration, similar to ketamine, produces antidepressant-like effects through identical mechanisms. J Agric Food Chem. 2017;65:10792–10803. doi: 10.1021/acs.jafc.7b04217. [DOI] [PubMed] [Google Scholar]
- 11.Chen KT, Tsai MH, Wu CH, Jou MJ, Wei IH, Huang CC. AMPA receptor-mTOR activation is required for the antidepressant-like effects of sarcosine during the forced swim test in rats: insertion of AMPA receptor may play a role. Front Behav Neurosci. 2015;9:162. doi: 10.3389/fnbeh.2015.00162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chuang HW, Wang TY, Huang CC, Wei IH. Echinacoside exhibits antidepressant-like effects through AMPAR-Akt/ERK-mTOR pathway stimulation and BDNF expression in mice. Chin Med. 2022;17:9. doi: 10.1186/s13020-021-00549-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chuang HW, Wei IH, Lin FY, Li CT, Chen KT, Tsai MH, et al. Roles of Akt and ERK in mTOR-dependent antidepressant effects of vanillic acid. ACS Omega. 2020;5:3709–3716. doi: 10.1021/acsomega.9b04271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Huang CC, Tsai MH, Wu YC, Chen KT, Chuang HW, Chen Y, et al. Activity dependent mammalian target of rapamycin pathway and brain derived neurotrophic factor release is required for the rapid antidepressant effects of puerarin. Am J Chin Med. 2018;46:1519–1534. doi: 10.1142/S0192415X18500787. [DOI] [PubMed] [Google Scholar]
- 15.Yin Y, Guan Y, Duan J, Wei G, Zhu Y, Quan W, et al. Cardioprotective effect of Danshensu against myocardial ischemia/reperfusion injury and inhibits apoptosis of H9c2 cardiomyocytes via Akt and ERK1/2 phosphorylation. Eur J Pharmacol. 2013;699:219–226. doi: 10.1016/j.ejphar.2012.11.005. [DOI] [PubMed] [Google Scholar]
- 16.Zhou L, Zuo Z, Chow MS. Danshen: an overview of its chemistry, pharmacology, pharmacokinetics, and clinical use. J Clin Pharmacol. 2005;45:1345–1359. doi: 10.1177/0091270005282630. [DOI] [PubMed] [Google Scholar]
- 17.Chan K, Chui SH, Wong DY, Ha WY, Chan CL, Wong RN. Protective effects of Danshensu from the aqueous extract of Salvia miltiorrhiza (Danshen) against homocysteine-induced endothelial dysfunction. Life Sci. 2004;75:3157–3171. doi: 10.1016/j.lfs.2004.06.010. [DOI] [PubMed] [Google Scholar]
- 18.Han JY, Horie Y, Fan JY, Sun K, Guo J, Miura S, et al. Potential of 3,4-dihydroxy-phenyl lactic acid for ameliorating ischemia-reperfusion-induced microvascular disturbance in rat mesentery. Am J Physiol Gastrointest Liver Physiol. 2009;296:G36–G44. doi: 10.1152/ajpgi.90284.2008. [DOI] [PubMed] [Google Scholar]
- 19.Yang GD, Zhang H, Lin R, Wang WR, Shi XL, Liu Y, et al. Down-regulation of CD40 gene expression and inhibition of apoptosis with Danshensu in endothelial cells. Basic Clin Pharmacol Toxicol. 2009;104:87–92. doi: 10.1111/j.1742-7843.2008.00342.x. [DOI] [PubMed] [Google Scholar]
- 20.Wang F, Liu YY, Liu LY, Zeng QJ, Wang CS, Sun K, et al. The attenuation effect of 3,4-dihydroxy-phenyl lactic acid and salvianolic acid B on venular thrombosis induced in rat mesentery by photochemical reaction. Clin Hemorheol Microcirc. 2009;42:7–18. doi: 10.3233/CH-2009-1180. [DOI] [PubMed] [Google Scholar]
- 21.Guo C, Yin Y, Duan J, Zhu Y, Yan J, Wei G, et al. Neuroprotective effect and underlying mechanism of sodium danshensu [3-(3,4-dihydroxyphenyl) lactic acid from Radix and Rhizoma Salviae miltiorrhizae = Danshen] against cerebral ischemia and reperfusion injury in rats. Phytomedicine. 2015;22:283–289. doi: 10.1016/j.phymed.2014.12.001. [DOI] [PubMed] [Google Scholar]
- 22.Xu H, Liu W, Liu T, Su N, Guo C, Feng X, et al. Synergistic neuroprotective effects of Danshensu and hydroxysafflor yellow A on cerebral ischemia-reperfusion injury in rats. Oncotarget. 2017;8:115434–115443. doi: 10.18632/oncotarget.23272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yu J, Wang L, Akinyi M, Li Y, Duan Z, Zhu Y, et al. Danshensu protects isolated heart against ischemia reperfusion injury through activation of Akt/ERK1/2/Nrf2 signaling. Int J Clin Exp Med. 2015;8:14793–14804. [PMC free article] [PubMed] [Google Scholar]
- 24.Chong CM, Zhou ZY, Razmovski-Naumovski V, Cui GZ, Zhang LQ, Sa F, et al. Danshensu protects against 6-hydroxydopamine-induced damage of PC12 cells in vitro and dopaminergic neurons in zebrafish. Neurosci Lett. 2013;543:121–125. doi: 10.1016/j.neulet.2013.02.069. [DOI] [PubMed] [Google Scholar]
- 25.Wang T, Li C, Han B, Wang Z, Meng X, Zhang L, et al. Neuroprotective effects of Danshensu on rotenone-induced Parkinson’s disease models in vitro and in vivo. BMC Complement Med Ther. 2020;20:20. doi: 10.1186/s12906-019-2738-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wei ZZ, Chen D, Liu LP, Gu X, Zhong W, Zhang YB, et al. Enhanced neurogenesis and collaterogenesis by sodium Danshensu treatment after focal cerebral ischemia in mice. Cell Transplant. 2018;27:622–636. doi: 10.1177/0963689718771889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wang T, Fu F, Han B, Zhang L, Zhang X. Danshensu ameliorates the cognitive decline in streptozotocin-induced diabetic mice by attenuating advanced glycation end product-mediated neuroinflammation. J Neuroimmunol. 2012;245:79–86. doi: 10.1016/j.jneuroim.2012.02.008. [DOI] [PubMed] [Google Scholar]
- 28.Kwon G, Kim HJ, Park SJ, Lee HE, Woo H, Ahn YJ, et al. Anxiolytic-like effect of danshensu [(3-(3,4-dihydroxyphenyl)-lactic acid)] in mice. Life Sci. 2014;101:73–78. doi: 10.1016/j.lfs.2014.02.011. [DOI] [PubMed] [Google Scholar]
- 29.Porsolt RD, Le Pichon M, Jalfre M. Depression: a new animal model sensitive to antidepressant treatments. Nature. 1977;266:730–732. doi: 10.1038/266730a0. [DOI] [PubMed] [Google Scholar]
- 30.Porsolt RD, Anton G, Blavet N, Jalfre M. Behavioural despair in rats: a new model sensitive to antidepressant treatments. Eur J Pharmacol. 1978;47:379–391. doi: 10.1016/0014-2999(78)90118-8. [DOI] [PubMed] [Google Scholar]
- 31.Du J, Machado-Vieira R, Maeng S, Martinowich K, Manji HK, Zarate CA., Jr Enhancing AMPA to NMDA throughput as a convergent mechanism for antidepressant action. Drug Discov Today Ther Strateg. 2006;3:519–526. doi: 10.1016/j.ddstr.2006.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Du J, Suzuki K, Wei Y, Wang Y, Blumenthal R, Chen Z, et al. The anticonvulsants lamotrigine, riluzole, and valproate differentially regulate AMPA receptor membrane localization: relationship to clinical effects in mood disorders. Neuropsychopharmacology. 2007;32:793–802. doi: 10.1038/sj.npp.1301178. [DOI] [PubMed] [Google Scholar]
- 33.Réus GZ, Stringari RB, Ribeiro KF, Ferraro AK, Vitto MF, Cesconetto P, et al. Ketamine plus imipramine treatment induces antidepressant-like behavior and increases CREB and BDNF protein levels and PKA and PKC phosphorylation in rat brain. Behav Brain Res. 2011;221:166–171. doi: 10.1016/j.bbr.2011.02.024. [DOI] [PubMed] [Google Scholar]
- 34.Banke TG, Bowie D, Lee H, Huganir RL, Schousboe A, Traynelis SF. Control of GluR1 AMPA receptor function by cAMP-dependent protein kinase. J Neurosci. 2000;20:89–102. doi: 10.1523/JNEUROSCI.20-01-00089.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Esteban JA, Shi SH, Wilson C, Nuriya M, Huganir RL, Malinow R. PKA phosphorylation of AMPA receptor subunits controls synaptic trafficking underlying plasticity. Nat Neurosci. 2003;6:136–143. doi: 10.1038/nn997. [DOI] [PubMed] [Google Scholar]
- 36.Smith KE, Gibson ES, Dell’Acqua ML. cAMP-dependent protein kinase postsynaptic localization regulated by NMDA receptor activation through translocation of an A-kinase anchoring protein scaffold protein. J Neurosci. 2006;26:2391–2402. doi: 10.1523/JNEUROSCI.3092-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Barria A, Muller D, Derkach V, Griffith LC, Soderling TR. Regulatory phosphorylation of AMPA-type glutamate receptors by CaM-KII during long-term potentiation. Science. 1997;276:2042–2045. doi: 10.1126/science.276.5321.2042. [DOI] [PubMed] [Google Scholar]
- 38.Tavalin SJ. AKAP79 selectively enhances protein kinase C regulation of GluR1 at a Ca2+-calmodulin-dependent protein kinase II/protein kinase C site. J Biol Chem. 2008;283:11445–11452. doi: 10.1074/jbc.M709253200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Duman RS, Monteggia LM. A neurotrophic model for stress-related mood disorders. Biol Psychiatry. 2006;59:1116–1127. doi: 10.1016/j.biopsych.2006.02.013. [DOI] [PubMed] [Google Scholar]
- 40.Lepack AE, Bang E, Lee B, Dwyer JM, Duman RS. Fast-acting antidepressants rapidly stimulate ERK signaling and BDNF release in primary neuronal cultures. Neuropharmacology. 2016;111:242–252. doi: 10.1016/j.neuropharm.2016.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Yu PF, Wang WY, Eerdun G, Wang T, Zhang LM, Li C, et al. The role of P-glycoprotein in transport of danshensu across the blood-brain barrier. Evid Based Complement Alternat Med. 2011;2011:713523. doi: 10.1155/2011/713523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Gao Y, Liu Z, Li G, Li C, Li M, Li B. Acute and subchronic toxicity of danshensu in mice and rats. Toxicol Mech Methods. 2009;19:363–368. doi: 10.1080/15376510902810672. [DOI] [PubMed] [Google Scholar]
- 43.Ren R, Wang T, Jiang N, Liu T, Du Y, Li C, et al. Protective effects of Danshensu on liver injury induced by omethoate in rats. Toxicol Mech Methods. 2010;20:510–514. doi: 10.3109/15376516.2010.514963. [DOI] [PubMed] [Google Scholar]
- 44.Tang Y, Wang M, Le X, Meng J, Huang L, Yu P, et al. Antioxidant and cardioprotective effects of Danshensu (3-(3, 4-dihydroxyphenyl)-2-hydroxy-propanoic acid from Salvia miltiorrhiza) on isoproterenol-induced myocardial hypertrophy in rats. Phytomedicine. 2011;18:1024–1030. doi: 10.1016/j.phymed.2011.05.007. [DOI] [PubMed] [Google Scholar]
- 45.Bakunina N, Pariante CM, Zunszain PA. Immune mechanisms linked to depression via oxidative stress and neuroprogression. Immunology. 2015;144:365–373. doi: 10.1111/imm.12443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Galts CPC, Bettio LEB, Jewett DC, Yang CC, Brocardo PS, Rodrigues ALS, et al. Depression in neurodegenerative diseases: common mechanisms and current treatment options. Neurosci Biobehav Rev. 2019;102:56–84. doi: 10.1016/j.neubiorev.2019.04.002. [DOI] [PubMed] [Google Scholar]
- 47.Jernigan CS, Goswami DB, Austin MC, Iyo AH, Chandran A, Stockmeier CA, et al. The mTOR signaling pathway in the prefrontal cortex is compromised in major depressive disorder. Prog Neuropsychopharmacol Biol Psychiatry. 2011;35:1774–1779. doi: 10.1016/j.pnpbp.2011.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ota KT, Liu RJ, Voleti B, Maldonado-Aviles JG, Duric V, Iwata M, et al. REDD1 is essential for stress-induced synaptic loss and depressive behavior. Nat Med. 2014;20:531–535. doi: 10.1038/nm.3513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Voleti B, Navarria A, Liu RJ, Banasr M, Li N, Terwilliger R, et al. Scopolamine rapidly increases mammalian target of rapamycin complex 1 signaling, synaptogenesis, and antidepressant behavioral responses. Biol Psychiatry. 2013;74:742–749. doi: 10.1016/j.biopsych.2013.04.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Chen J, Yuan S, Zhou J, Huang X, Wu W, Cao Y, et al. Danshen injection induces autophagy in podocytes to alleviate nephrotic syndrome via the PI3K/AKT/mTOR pathway. Phytomedicine. 2022;107:154477. doi: 10.1016/j.phymed.2022.154477. [DOI] [PubMed] [Google Scholar]
- 51.Fan G, Yu J, Asare PF, Wang L, Zhang H, Zhang B, et al. Danshensu alleviates cardiac ischaemia/reperfusion injury by inhibiting autophagy and apoptosis via activation of mTOR signalling. J Cell Mol Med. 2016;20:1908–1919. doi: 10.1111/jcmm.12883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gao Q, Deng H, Yang Z, Yang Q, Zhang Y, Yuan X, et al. Sodium danshensu attenuates cerebral ischemia-reperfusion injury by targeting AKT1. Front Pharmacol. 2022;13:946668. doi: 10.3389/fphar.2022.946668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Hu S, Chen Y, Huang S, Liu M, Liu Y, Huang S. Sodium Danshensu protects against oxygen glucose deprivation/reoxygenation-induced astrocytes injury through regulating NOD-like receptor pyrin domain containing 3 (NLRP3) inflammasome and tuberous sclerosis complex-2 (TSC2)/mammalian target of rapamycin (mTOR) pathways. Ann Transl Med. 2022;10:1097. doi: 10.21037/atm-22-2143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Jia H, Qi X, Wu H, Wang J. Danshensu enhances cerebral angiogenesis in mice by regulating the PI3K/Akt/mTOR/VEGF signaling axis. CNS Neurol Disord Drug Targets. 2023;22:607–613. doi: 10.2174/1871527321666220329144538. [DOI] [PubMed] [Google Scholar]
- 55.Tang F, Zhou X, Wang L, Shan L, Li C, Zhou H, et al. A novel compound DT-010 protects against doxorubicin-induced cardiotoxicity in zebrafish and H9c2 cells by inhibiting reactive oxygen species-mediated apoptotic and autophagic pathways. Eur J Pharmacol. 2018;820:86–96. doi: 10.1016/j.ejphar.2017.12.021. [DOI] [PubMed] [Google Scholar]
- 56.Xie C, Luo J, Hu H, Wang L, Yu P, Xu L, et al. A novel danshensu/tetramethypyrazine derivative attenuates oxidative stress-induced autophagy injury via the AMPK-mTOR-Ulk1 signaling pathway in cardiomyocytes. Exp Ther Med. 2021;21:118. doi: 10.3892/etm.2020.9550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Park SW, Lee JG, Seo MK, Lee CH, Cho HY, Lee BJ, et al. Differential effects of antidepressant drugs on mTOR signalling in rat hippocampal neurons. Int J Neuropsychopharmacol. 2014;17:1831–1846. doi: 10.1017/S1461145714000534. [DOI] [PubMed] [Google Scholar]
- 58.Lu W, Shi Y, Jackson AC, Bjorgan K, During MJ, Sprengel R, et al. Subunit composition of synaptic AMPA receptors revealed by a single-cell genetic approach. Neuron. 2009;62:254–268. doi: 10.1016/j.neuron.2009.02.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Diering GH, Huganir RL. The AMPA receptor code of synaptic plasticity. Neuron. 2018;100:314–329. doi: 10.1016/j.neuron.2018.10.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Beattie MS, Ferguson AR, Bresnahan JC. AMPA-receptor trafficking and injury-induced cell death. Eur J Neurosci. 2010;32:290–297. doi: 10.1111/j.1460-9568.2010.07343.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Logan SM, Sarkar SN, Zhang Z, Simpkins JW. Estrogen-induced signaling attenuates soluble Aβ peptide-mediated dysfunction of pathways in synaptic plasticity. Brain Res. 2011;1383:1–12. doi: 10.1016/j.brainres.2011.01.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Schloesser RJ, Huang J, Klein PS, Manji HK. Cellular plasticity cascades in the pathophysiology and treatment of bipolar disorder. Neuropsychopharmacology. 2008;33:110–133. doi: 10.1038/sj.npp.1301575. [DOI] [PubMed] [Google Scholar]
- 63.Zarate CA, Jr, Singh J, Manji HK. Cellular plasticity cascades: targets for the development of novel therapeutics for bipolar disorder. Biol Psychiatry. 2006;59:1006–1020. doi: 10.1016/j.biopsych.2005.10.021. [DOI] [PubMed] [Google Scholar]
- 64.Castrén E, Monteggia LM. Brain-derived neurotrophic factor signaling in depression and antidepressant action. Biol Psychiatry. 2021;90:128–136. doi: 10.1016/j.biopsych.2021.05.008. [DOI] [PubMed] [Google Scholar]
- 65.Chandran A, Iyo AH, Jernigan CS, Legutko B, Austin MC, Karolewicz B. Reduced phosphorylation of the mTOR signaling pathway components in the amygdala of rats exposed to chronic stress. Prog Neuropsychopharmacol Biol Psychiatry. 2013;40:240–245. doi: 10.1016/j.pnpbp.2012.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Duman RS, Aghajanian GK. Synaptic dysfunction in depression: potential therapeutic targets. Science. 2012;338:68–72. doi: 10.1126/science.1222939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.liu S, Li T, Liu H, Wang X, Bo S, Xie Y, et al. Resveratrol exerts antidepressant properties in the chronic unpredictable mild stress model through the regulation of oxidative stress and mTOR pathway in the rat hippocampus and prefrontal cortex. Behav Brain Res. 2016;302:191–199. doi: 10.1016/j.bbr.2016.01.037. [DOI] [PubMed] [Google Scholar]
- 68.Nestler EJ. Role of the brain’s reward circuitry in depression: transcriptional mechanisms. Int Rev Neurobiol. 2015;124:151–170. doi: 10.1016/bs.irn.2015.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Shirayama Y, Yang C, Zhang JC, Ren Q, Yao W, Hashimoto K. Alterations in brain-derived neurotrophic factor (BDNF) and its precursor proBDNF in the brain regions of a learned helplessness rat model and the antidepressant effects of a TrkB agonist and antagonist. Eur Neuropsychopharmacol. 2015;25:2449–2458. doi: 10.1016/j.euroneuro.2015.09.002. [DOI] [PubMed] [Google Scholar]