Abstract
The central nervous system and adipose tissue interact through complex communication. This bidirectional signaling regulates metabolic functions. The hypothalamus, a key homeostatic brain region, integrates exteroceptive and interoceptive signals to control appetite, energy expenditure, glucose, and lipid metabolism. This regulation is partly achieved via the nervous modulation of white (WAT) and brown (BAT) adipose tissue. In this review, we highlight the roles of sympathetic and parasympathetic innervation in regulating WAT and BAT activities, such as lipolysis and thermogenesis. Adipose tissue, in turn, plays a dual role as an energy reservoir and an endocrine organ, secreting hormones that influence brain function and metabolic health. In addition, this review focuses on recently uncovered communication pathways, including extracellular vesicles and neuro-mesenchymal units, which add new layers of regulation and complexity to the brain–adipose tissue interaction. Finally, we also examine the consequences of disrupted communication between the brain and adipose tissue in metabolic disorders like obesity and type-2 diabetes, emphasizing the potential for new therapeutic strategies targeting these pathways to improve metabolic health.
Keywords: Brain, Brown Adipose Tissue, White Adipose Tissue, Sympathetic and Parasympathetic Innervation, Adipokines
Subject terms: Metabolism, Neuroscience
This review discusses the crosstalk between the central nervous system and the adipose tissue, focusing on how this bidirectional signaling regulates metabolic functions.

Introduction
The central nervous system (CNS) can be regarded as a sophisticated interface computer. It senses and integrates a variety of signals from the body’s internal milieu and the environment, executing multiple biological programs to maintain optimal health and ensure survival. This is achieved by modulating essential functions such as appetite, energy expenditure, metabolism, and behavior. While these functions are modulated by complex and distributed neurocircuits across multiple brain regions, the hypothalamus (particularly the arcuate nucleus; ARC) critically contributes to these biological processes. This occurs in part through the coordinated action of the melanocortin system, specifically pro-opiomelanocortin (POMC) and agouti-related peptide (AgRP)-expressing neurons (Brüning and Fenselau, 2023). Therefore, adequate organismal homeostasis relies on intricate multiway communication routes between organs, with the brain acting as a central processing unit.
Adipose tissue is a fundamental and dynamic organ found across a wide range of the animal kingdom, serving as a buffer for excess calories, a metabolic hub, and an endocrine platform. Adipose tissue comprises two types of depots: white adipose tissue (WAT) and brown adipose tissue (BAT), each exhibiting unique anatomical, functional, and metabolic characteristics crucial for energy balance and physiological regulation. WAT is the predominant type of adipose tissue, functioning as an insulator, energy reservoir (in the form of unilocular storage of triglycerides), and endocrine organ by the release of adipokines (e.g., leptin, adiponectin, resistin) and other signaling molecules (e.g., cytokines, fatty acids, exosomes). In contrast, BAT is selectively located in specific regions, exhibits abundant mitochondria, and specializes in thermogenesis via the distinct expression of uncoupling protein 1 (UCP1). Therefore, precise control of adipose tissue function is key for adapting to the ever-changing metabolic demands and maintaining metabolic health.
In this review, we delve into the bidirectional communication between the CNS and adipose tissue. We examine the anatomical nature, functions, and integration features of efferent (from brain to adipose tissue) and afferent (from adipose tissue to brain) signals upon adipose tissue physiology and systemic metabolism. We also explore novel modes of communication between the brain and adipose tissue, beyond classic nervous innervation, with implications for metabolic control. Finally, we also highlight the pathophysiological consequences of brain–adipose tissue axis dysfunction in the context of prevalent metabolic conditions such as obesity and type-2 diabetes (T2D).
Brain → white adipose tissue crosstalk
Efferent innervation
Anatomical and functional aspects of sympathetic innervation to the white adipose tissue
WAT is directly innervated by sympathetic endings of the autonomic nervous system (Fig. 1). In response to norepinephrine (NE) binding to β-adrenergic receptors (βARs) on adipocytes, a signaling cascade is triggered that enhances lipolysis and releases free fatty acids (FFA) and glycerol from white adipocytes (Lass et al, 2011) (Fig. 1). Sympathetic nervous system (SNS) innervation of WAT has been demonstrated by different means, including electrophysiology, surgical sympathectomy, and electrical stimulation of sympathetic nerve endings (Nguyen et al, 2014b). Bamshad et al defined the origins of the sympathetic innervation of WAT using trans-synaptic retrograde neural tracer pseudorabies virus (PRV) in a model of Siberian hamsters. Injection of this virus into the WAT-identified neurons localized in the spinal cord, brainstem, midbrain, and forebrain, in addition to different hypothalamic nuclei (Bamshad et al, 1998). Later studies in rats using neurotrophic viruses defined that WAT projections from the ARC were mainly derived from POMC neurons, while projections from AgRP were absent (Adler et al, 2012). Importantly, WAT is comprised of different fat pads (i.e., subcutaneous versus visceral). Each one of these fat pads is characterized by a distinct innervation density, specific receptor levels, and sympathetic nerve activity (Willows et al, 2023a; Vaughan et al, 2014). Interestingly, sexual dimorphic anatomical differences were also reported in sympathetic WAT innervation (Adler et al, 2012), indicative of a sex-dependent metabolic regulation of WAT. This observation was also confirmed by advanced WAT clearing techniques, which allowed imaging of entire fat pads, revealing that almost 99% of presynaptic fibers within WAT are likely sympathetic (tyrosine hydroxylase (TH) and synaptophysin positive) (Jiang et al, 2017).
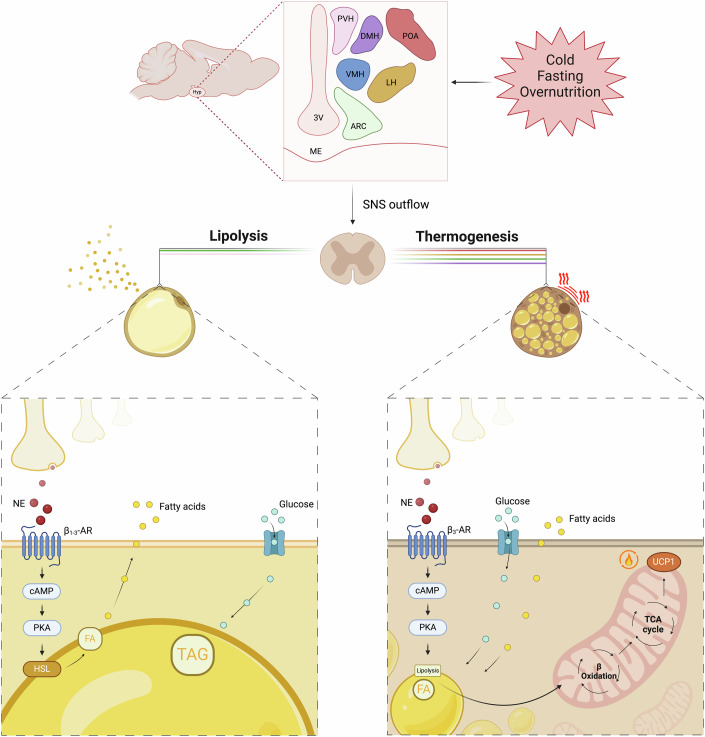
Figure 1. Neural regulation of adipocyte function by the sympathetic nervous system.
The role of the sympathetic nervous system (SNS) in regulating adipose tissue function in response to metabolic challenges is illustrated. The upper panel illustrates the brain regions involved in this process in different colors, including the paraventricular nucleus (PVH), dorsomedial nucleus (DMH), ventromedial nucleus (VMH), lateral hypothalamus (LH), arcuate nucleus (ARC), and pre-optical area (POA). The lower panels detail the specific actions of SNS outflow on white adipose tissue (WAT) (left) and brown adipose tissue (BAT) (right) referring to the color of the different hypothalamic nuclei related to lipolysis and thermogenesis. In WAT, norepinephrine (NE) binds to β3-adrenergic receptors (β3-AR), triggering a signaling cascade involving cyclic AMP (cAMP) and protein kinase A (PKA) that leads to the activation of hormone-sensitive lipase (HSL) and subsequent lipolysis, releasing fatty acids (FA). In BAT, NE binds to β3-AR, promoting the uptake of glucose and fatty acids. These substrates undergo β-oxidation in the mitochondria, driving thermogenesis through the action of uncoupling protein 1 (UCP1). This thermogenic response helps to dissipate energy as heat, contributing to the maintenance of energy balance.
A crucial function of sympathetic innervation of WAT is controlling lipolysis. Pioneering in vitro studies performed in the sixties by James W. Correll demonstrated that the concentration of FFA in the bathing medium increased when the nerves of a dissected epididymal fat pad were electrically stimulated (Correll, 1963). This observation was confirmed in different animal models, concluding that surgical or chemical WAT denervation blocked lipid mobilization (Harris, 2018). Importantly, sympathetic innervation is also involved in the development and maintenance of WAT by controlling the entire lifecycle of pre-adipocytes, from their proliferation and differentiation to apoptosis (Cousin et al, 1993; Ruschke et al, 2009; Bowers et al, 2004).
Anatomical and functional aspects of parasympathetic innervation to the white adipose tissue
The paraventricular nucleus of the hypothalamus (PVH) receives inputs from various hypothalamic nuclei, including the suprachiasmatic nucleus (SCN), which serves as the brain’s circadian clock, and the ARC. The PVH sends autonomic projections to the dorsal motor nucleus of the vagus (DMV), where parasympathetic motor neurons that innervate WAT are located. In addition, the PVH projects to the intermediolateral column, the source of sympathetic innervation to WAT.
In contrast to the well-established sympathetic innervation of WAT, which has been confirmed by diverse research groups and study models, the functional outcome of parasympathetic nervous system (PNS) innervation of WAT remains controversial, with different studies reporting conflicting results. Indeed, Giordano et al did not detect neurochemical markers of parasympathetic innervation in various WAT depots using models such as Sprague–Dawley rats, C57Bl/6 mice, and leptin-deficient ob/ob mice (Giordano et al, 2006; Bartness et al, 2014). Nonetheless, Kreier et al, in rats, showed that PRV inoculation of intra-abdominal WAT labeled the DMV nerve, providing evidence of parasympathetic innervation of WAT (Kreier et al, 2002). However, this parasympathetic innervation does not appear to be involved in lipogenesis, which would have been expected as the opposing effect to the lipolytic function of the SNS. Instead, Kreier et al showed that parasympathetic innervation of WAT is implicated in the production of leptin and resistin as well as in insulin sensitivity, as evidenced by the decrease in insulin-dependent glucose uptake and FFA uptake following vagotomy (Kreier et al, 2002).
Metabolic effects via efferent innervation
As previously mentioned, the SNS exerts significant influence over WAT lipolysis primarily through NE-mediated activation of βARs (subtypes β1-3). Activation of βAR β1-3 engages the GTP-binding protein Gsα, promoting cAMP production via adenylyl cyclase. cAMP enhances protein kinase A (PKA) activity, phosphorylating and activating two crucial proteins for lipolysis: the hormone-sensitive lipase (HSL) and perilipin A (Lass et al, 2011) (Fig. 1). During stress or exercise, the release of NE from sympathetic nerve terminals increases thereby stimulating βARs and promoting FFA release (Santos et al, 2022). βAR levels can be controlled by the CNS as central melanin-concentrating hormone (MCH) infusion, acting on ARC neurons expressing the MCH receptor, diminishes the expression of βARs in WAT (Imbernon et al, 2013).
POMC and AgRP neurons influence WAT lipolysis. Specific activation of AgRP neurons results in reduced circulating fatty acids and activation of HSL in WAT via the SNS (Cavalcanti-de-Albuquerque et al, 2019). Chemogenetic manipulation of POMC neurons curtails the molecular lipolytic program and decreases fasting-induced lipid mobilization (Gómez-Valadés et al, 2021). Leptin, a key adipokine crucially implicated in energy balance control, also exerts a lipolytic effect by activating sympathetic neurons that innervate WAT adipocytes. This effect is partially mediated by the activation of βARs (Zeng et al, 2015). Leptin, by acting on its receptors in AgRP and POMC neurons in the ARC, controls the sympathetic innervation of adipose tissue via a top-down neural pathway that depends on the generation of Brain-Derived Neurotrophic Factor (BDNF) produced by neurons in the PVH (Wang et al, 2020a). Nonetheless, leptin also exerts a concomitant anti-lipogenesis action via hypothalamic phosphoinositide 3-kinase signaling and the endocannabinoid anandamide in WAT (Buettner et al, 2008). Conversely to leptin, insulin infusion into the mediobasal hypothalamus (MBH) dampens SNS activity and enhances WAT lipogenic protein expression, inhibiting HSL and, therefore, lipolysis (Scherer et al, 2011).
Interestingly, it has been reported that the brain controls leptin gene expression in the WAT through sympathetic innervation (Kalsbeek et al, 2001). Indeed, lesions in the SCN, which abolish the biological clock, eliminate the daily rhythm of leptin release (Kalsbeek et al, 2001). Furthermore, SNS innervation also controls the lifecycle (proliferation) of pre-adipocytes. This is evidenced by models of denervation or studies in transgenic mice lacking the neuron-specific Nscl-2 (Nescient Helix–Loop–Helix 2) transcription factor, which exhibit adulthood obesity due to an increased number of pre-adipocytes (Cousin et al, 1993; Ruschke et al, 2009). In Nscl-2 mutant mice, the reduced innervation also leads to a significant decrease in the microvasculature, which might contribute to the observed alterations in WAT (Ruschke et al, 2009). Altogether, these studies reveal the diverse functions of the SNS, ranging from its classical role in the induction of lipolysis to its lesser-studied involvement in hormone production and adipogenesis (Box 1).
In response to external stimuli, such as cold exposure, adrenaline-mediated SNS activation promotes the “browning” of white adipocytes. During this process, white fat cells acquire a brown fat-like phenotype with the primary function of dissipating energy as heat via thermogenesis (Schirinzi et al, 2023). White fat browning is a complex process that is activated by different signaling pathways, which culminates with the expression of UCP1, together with other brown-tissue specific genes (e.g., PPARγ, PRDM-16, and PGC-1α) (Seale et al, 2007). The excitation of the SNS and activation of the β3-AR signaling pathway induced by PRDM-16 is the classic mechanism driving the browning of white adipocytes (Qiang et al, 2012; Seale et al, 2011). Recent studies also indicate that thyroid hormones play a significant role in promoting the browning of WAT, nonetheless this process does not necessarily depend on the SNS (Johann et al, 2019). Taken together, these studies emphasize the importance of innervation in maintaining adipose metabolic health across various levels, including functional, developmental, and lineage aspects.
Box 1: In need of answers.
Does the PNS innervate WAT? And, if so, what is its functional outcome?
How do sensory nerves and their secreted neuropeptides exert their effects to counteract sympathetic signaling in adipose tissues?
What is the precise role of leptin in thermogenesis?
How do sensory pathways mechanistically interact with the SNS?
Could mechanical forces in WAT or BAT control adipokine/batokine release and represent a new mechanism of adipose tissue →brain communication?
How do BAT-derived factors influence brain function and overall energy homeostasis?
What is the relevance of bidirectional brain–adipose tissue communication in humans in the context of metabolic control and health?
Brain → brown adipose tissue crosstalk
BAT is the primary source of nonshivering thermogenesis, capable of increasing energy expenditure by approximately 5-10%, oxidizing intracellular triglycerides, and enhancing glucose uptake upon activation (Celi et al, 2010; Yoneshiro et al, 2011; Orava et al, 2011; Chen et al, 2013; Ouellet et al, 2012). This makes BAT an attractive target to counteract metabolic disturbances. Brown adipocytes, characterized by their abundant mitochondria and multilocular lipid droplets, play a crucial role in adaptive thermogenesis through the expression of UCP1. The interaction between the brain and BAT enables an effective response to challenges such as fasting, cold exposure, and energy balance deregulation.
Efferent innervation
The communication between the brain and BAT primarily occurs through the SNS (Fig. 1). The intermediolateral nucleus (IML), located between the dorsal and ventral horns of the spinal cord and extending from segment C8 to L1, contains preganglionic sympathetic neurons. This branch is the principal efferent innervation to the BAT (Morrison and Nakamura, 2019) and, like WAT, is predominantly composed of TH cells (Murano et al, 2009). Nervous activation of BAT occurs in response to various stimuli, such as cold exposure, thyroid hormones or energy balance deregulation, activating the SNS. Cold exposure and energy balance deregulation are sensed and integrated by the brain, triggering the release of NE from TH cells leading to the activation of β3-AR. These receptors are abundantly expressed in BAT and play crucial roles in adaptive thermogenesis (Bartness et al, 2010), and their sensitivity is controlled, among other factors, by thyroid hormones. Indeed, central triiodothyronine (T3) stimulates lipid oxidation and therefore a thermogenic program in BAT by acting on AMP-activated protein kinase (AMPK) (Martínez-Sánchez et al, 2017).
In addition, at the intracellular level, and similar to WAT, the binding of NE to β3-AR increases cAMP levels. This second messenger activates PKA, which in turn regulates the activity of HSL. Consequently, this process releases fatty acids, serving as oxidative substrates that stimulate the activation of mitochondrial UCP1 (Cannon and Nedergaard, 2004; Matthias et al, 2000) (Fig. 1). UCP1, embedded in the inner mitochondrial membrane of brown adipocytes, uncouples oxidative phosphorylation from ATP synthesis, allowing protons to flow back into the mitochondrial matrix without generating ATP. Increased UCP1 expression requires T3, and T3-dependent thyroid hormone receptor (TR) β activation (Ribeiro et al, 2010; Bianco and Silva, 1987). UCP1 uncoupling, which characterizes BAT, results in the dissipation of energy as heat that is crucial for maintaining body temperature and energy balance homeostasis (Cannon and Nedergaard, 2004).
The SNS activity involved in BAT thermogenesis originates from the hypothalamus (Fig. 1). Within this region, various nuclei converge to orchestrate the regulation of body temperature and energy balance, aligning with the pivotal role of thermoregulation in metabolic homeostasis. Among the brain regions implicated in the regulation of core body temperature, also known as the “thermoregulatory pathway”, the preoptic area (POA) stands out as a crucial nucleus (Morrison and Nakamura, 2019). The POA coordinates a sophisticated circuit that, in a simplified view, responds to low temperatures by receiving inputs from skin thermoreceptors (Nakamura and Morrison, 2008). Subsequently, it generates outputs to other hypothalamic nuclei, such as the dorsomedial hypothalamus (DMH), thus triggering heat production by BAT and contributing to temperature homeostasis (Contreras et al, 2017). Conversely, DMH cholinergic neurons are active in warm temperatures, and their activity is related to the regulation of BAT thermogenesis (Jeong et al, 2015). Lesions in the ventromedial hypothalamus (VMH) have been shown to impair the ability to respond to external cooling or direct stimulation of the POA to induce BAT thermogenesis (Hogan et al, 1982; Preston et al, 1989). These findings underscore the critical role of complex circuits between the POA, VMH, and DMH in body thermoregulation. Among the extra-hypothalamic regions involved in efferent signaling to BAT, the dorsal raphe nucleus (DRN) has been reported as a key player. Electrophysiological recordings have identified certain GABAergic DRN neurons whose firing rates are influenced by fluctuations in local or ambient temperature (Weiss and Aghajanian, 1971; Hale et al, 2011). Moreover, electrical stimulation or pharmacological manipulation of these neurons can alter BAT thermogenesis and concomitantly core temperature (Dib et al, 1994; Higgins et al, 1988). Recently, it has been reported that GABAergic neurons in the DRN are activated by ambient heat and regulate energy expenditure bidirectionally through changes in both thermogenesis and locomotion (Schneeberger et al, 2019). Optogenetic studies revealed that these neurons influence thermogenesis via direct innervation of the BAT through to the raphe pallidus (RPa) and indirect ascending pathways to the hypothalamus and extended amygdala (Schneeberger et al, 2019). Additional extra-hypothalamic areas have been proposed in thermoregulatory control, such as the inferior olivary nucleus (Uno and Shibata, 2001), highlighting the great complexity of thermoregulation mechanisms.
Metabolic effects via efferent innervation
During fluctuations in energy balance, as a mechanism to adjust to metabolic demands and maintain overall homeostasis, several hypothalamic nuclei (including the ARC, VMH, and PVH) play key roles in modulating thermogenesis through BAT activation (Contreras et al, 2017). Nutrient levels and hormonal flux, which inform on metabolic status, influence neuronal activity within these nuclei. Importantly, their thermogenic actions can occur independently of “thermoregulatory pathway” circuits (Zheng et al, 1995; Contreras et al, 2017), emphasizing the significance of BAT thermogenic activity in energy homeostasis.
Melanocortin neurons predominantly project to the PVH (Wang et al, 2015), where the release of the POMC-derived neuropeptide alpha-melanocyte-stimulating hormone (α-MSH) binds and activates melanocortin receptors (MCR; mainly MC4-R). This activation is counteracted by the inverse agonist AgRP, which inhibits MCRs and opposes the effects of α-MSH (Nijenhuis et al, 2001). These antagonistic interactions form a critical component of the melanocortin system (De Solis et al, 2024). Through transneuronal retrograde tracing studies using PRVs, it has been demonstrated that SNS connections to the BAT originate from brain areas with notable MC4-R expression (Song et al, 2008). Central pharmacologic interventions with a MCR agonist, such as Melanotan II (MTII), directly into the PVH induces BAT thermogenesis (Monge-Roffarello et al, 2014). Consistent with these findings, several mouse mutants of components of the melanocortin system or direct manipulations of POMC and/or AgRP neurons through optogenetic or chemogenetic activation result in BAT thermogenesis or modulation of BAT glucose uptake, profoundly affecting energy homeostasis (Yu et al, 2020; Schneeberger et al, 2013; Quiñones et al, 2021; Steculorum et al, 2016; Han et al, 2021).
Hypothalamic lipid sensing also modulates BAT activity, with central ceramides emerging as key lipid species influencing sympathetic outflow to BAT. This regulatory mechanism attenuates thermogenesis and promotes a positive energy balance, unveiling a pathway linking lipid signaling in the brain with systemic energy homeostasis (Contreras et al, 2014).
The effects of leptin on BAT thermogenesis are not entirely clear. Several studies using intracerebroventricular (ICV) injections of leptin have shown an increase in BAT and body temperature (Rodríguez-Rodríguez et al, 2019; Pelleymounter et al, 1995), an increase in NE release (Satoh et al, 1999), increase in BAT glucose uptake (Haque et al, 1999), and elevated Ucp1 mRNA expression in wild-type mice (Rodríguez-Rodríguez et al, 2019; Mistry et al, 1997; Scarpace and Matheny, 1998; Commins et al, 1999). However, peripheral administration of leptin elicits marginal or no thermogenic response in wild-type or leptin-deficient ob/ob mice (Harris et al, 1998). These discrepancies seem to arise from variations in the administration routes, animal models, methods of measuring thermogenesis, and differences between exogenous and endogenous leptin in its physiological effects (Ottaway et al, 2015). Importantly, evidence suggests that leptin may play a role in maintaining body temperature without activating thermogenesis (Fischer et al, 2016). In support of this, it has been reported that sympathetic innervation to BAT is dispensable for leptin-induced weight loss (Côté et al, 2018). As the precise role of leptin in thermogenesis remains unclear, this question can be further explored elsewhere (Fischer et al, 2020).
The VMH is another hypothalamic nucleus that has been extensively associated with BAT thermogenesis. Specifically, neurons expressing steroidogenic factor 1 (SF1; a transcription factor exclusively expressed in the VMH) play a crucial role in this process. Studies deleting the SF1 gene or ablating SF1 neurons have demonstrated significant impairments in BAT thermogenesis (Majdic et al, 2002; Kim et al, 2011; Rashid et al, 2023). From the mechanistic perspective, accumulating evidence indicates that the energy-sensor AMPK within the VMH may act as a canonical mediator of BAT thermogenesis by integrating diverse homeostatic signals (Seoane-Collazo et al, 2018; Martínez-Sánchez et al, 2017; Beiroa et al, 2014; Martínez de Morentin et al, 2014).
Other systems crucial for the regulation of energy balance, such as the endocannabinoid system, have also been implicated in BAT thermogenesis. Through its interaction with cannabinoid receptors 1 and 2 (CB1R and CB2R), this system has been proposed as a target for several obesity treatments (Miralpeix et al, 2021). Direct administration of the cannabinoid receptor agonist delta-9-tetrahydrocannabinol (D9-THC) has been shown to reduce UCP1 on BAT (Verty et al, 2011), while CB1 antagonist rimonabant increases thermogenesis (Verty et al, 2009). Genetic manipulation of cannabinoid receptors also affects BAT activity (Cardinal et al, 2012).
Most of the information discussed in this section was derived from preclinical models; however, it is important to note that the presence of functional BAT in adult humans was confirmed about 15 years ago. In humans, higher BAT activity positively correlates with a lower body mass index (BMI), younger age, colder outdoor temperatures, female sex, reduced glucose levels, and improved cardiometabolic health (Cohade et al, 2003; van Marken Lichtenbelt et al, 2009; Virtanen et al, 2009; Cypess et al, 2009). However, the brain→BAT crosstalk in maintaining metabolic homeostasis in humans remains largely unknown.
White adipose tissue → brain crosstalk
Sensory innervation
The sensory innervation pathway is a sophisticated network that facilitates communication between peripheral fat depots and the CNS (Fig. 2). Sensory neurons, which are pseudo-unipolar and reside in the dorsal root ganglia (DRG) of the spinal cord, have axons extending peripherally toward tissues and centrally toward the brain via the spinal cord. DRG neurons, which are clusters of sensory neuron cell bodies located adjacent to the spinal cord, serve as a processing hub, relaying sensory information to the brain. The specialized structural and functional adaptations of sensory neurons at their termini allow them to detect a wide range of stimuli through various types of receptors: mechanoreceptors, thermoreceptors, nociceptors, electromagnetic receptors, and chemoreceptors (Valencia-Montoya et al, 2024). Although their function and innervation patterns are still largely unknown, reports have demonstrated sensory afferent pathways originating from the WAT to the brain. These pathways are crucial for regulating lipid mobilization and body fat stores as they transmit information about the metabolic state of fat through hormones secreted by the adipose tissue (Willows et al, 2021; Zhang et al, 2018).
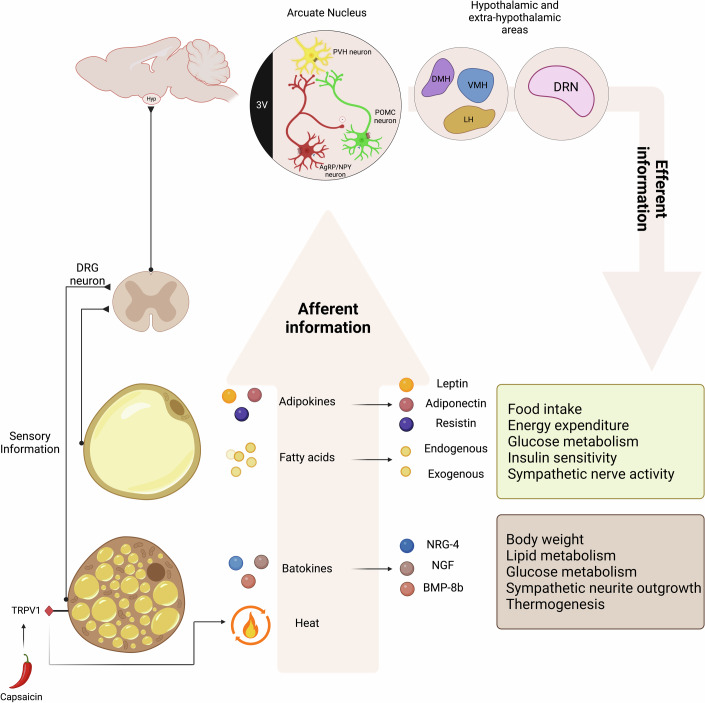
Figure 2. Afferent neural pathways and adipose tissue-derived signals in energy homeostasis.
The afferent neural pathways and the signals derived from adipose tissue that contribute to the regulation of energy homeostasis are illustrated. The central panel shows the arcuate nucleus (ARC) in the hypothalamus, which houses first-order neurons, including pro-opiomelanocortin (POMC) and agouti-related peptide/neuropeptide Y (AgRP/NPY) neurons. These neurons detect fluctuations in energy status (afferent information) and, in response, interact with other hypothalamic nuclei, including the dorsomedial hypothalamus (DMH), ventromedial hypothalamus (VMH), lateral hypothalamus (LH), and extra-hypothalamic regions such as the dorsal raphe nucleus (DRN). Collectively, these structures modulate energy balance and metabolic processes (efferent information) to maintain systemic energy homeostasis. Sensory information from adipose tissues is carried by dorsal root ganglion (DRG) neurons to deliver information to the central nervous system. The lower panels detail the specific signals emanating from white adipose tissue (WAT) and brown adipose tissue (BAT). WAT releases adipokines (e.g., leptin, adiponectin, resistin) and fatty acids, which modulate various physiological processes such as food intake, energy expenditure, glucose metabolism, insulin sensitivity, and sympathetic nerve activity. BAT produces batokines (e.g., NRG-4, NGF, BMP-8b) and heat through thermogenesis. These signals influence body weight, lipid metabolism, glucose metabolism, sympathetic neurite outgrowth, and energy balance. The transient receptor potential vanilloid 1 (TRPV1) channels, activated by capsaicin, play a role in transmitting thermal and metabolic information from BAT, thereby integrating sensory information with energy homeostasis. This intricate system of afferent and efferent signaling pathways underscores the complexity of energy regulation mechanisms and highlights the pivotal role of both central and peripheral signals in maintaining metabolic stability.
Early immunocytochemistry studies using antibodies against the neuropeptides substance P and CGRP (calcitonin gene-related peptide) (Giordano et al, 1996) or against ubiquitous proteins characteristic of sensory neurons (advillin and Nav18) (Willows et al, 2021) identified the presence of sensory-related innervation in the WAT. This was confirmed using an anterograde transneuronal viral tract tracer, as shown by Song et al, who identified a sensory WAT pathway to the brain in the inguinal and epididymal WAT of the Siberian hamster model (Song et al, 2009). The sensory innervation is mainly composed of “C” fibers, small and non-myelinated, although myelinated fibers have also been described (Giordano et al, 1996; Willows et al, 2021, 2023b). The CNS projections of WAT afferents overlap with SNS outflow sites, creating a WAT sensory-SNS circuit involved in the control of lipid mobilization (Song et al, 2009). More recent work also reported the existence of somatosensory fibers, originating from the DRG, innervating adipose tissue in rodents (Bartness et al, 2014; Fishman and Dark, 1987). These fibers provide fast neural transmission of information from peripheral organs to the brain. It remains unclear which DRG subtypes innervate WAT and how the sensory pathways mechanistically interact with the SNS. Nevertheless, it is well-established that sensory ablation affects fatty acid and lipid metabolism. In addition, sensory ablation increases cold-induced thermogenesis driving the browning of WAT and rising body temperature (Wang et al, 2022b). Sympathetic neuron deletion increases the size of the WAT fat pad, indicating that sensory innervation also counteracts sympathetic activity (Wang et al, 2022b). As such, the afferent flow of sensory information from a WAT pad directly modulates the activity of the sympathetic efferent pathways that innervate the same pad. Altogether, these studies demonstrate a bidirectional neural communication between the brain and WAT, which is relevant for energy balance and metabolic control.
Endocrine regulation of the adipose–brain axis (adipokines)
As previously mentioned, WAT is a crucial endocrine organ that secretes hormones and other factors collectively known as adipokines. These adipokines play a major role in the regulation of organismal metabolism by informing the brain about whole-body long-term energy-storage status (Fig. 2). The best-studied adipokine is leptin (Zhang et al, 1994), which is produced by the adipose tissue in proportion to fat stores and informs the brain about energy-storage levels in adipose tissue. The leptin receptor (LepR) is most densely distributed in the ARC (Elmquist et al, 1998). Leptin acts on ARC neurons, promoting satiety and increasing energy expenditure, thus controlling energy balance (Lavoie et al, 2023). Indeed, leptin activates POMC neurons (Cowley et al, 2001) while inhibiting AgRP neurons (van den Top et al, 2004). It also activates presynaptic GABAergic neurons, thereby decreasing the inhibitory tone of postsynaptic POMC neurons (Vong et al, 2011). Recent work has identified an additional population of ARC neurons expressing LepR: prepronociceptin (PNOC)-expressing neurons (Jais et al, 2020). The activation of PNOC neurons by short-term high-fat diet (HFD) feeding is mediated by inhibitory synaptic input to POMC neurons, leading to an increase in food intake (Jais et al, 2020). Leptin inhibits a subpopulation of PNOC neurons (around 80% of neurons); nonetheless, the relevance of these neurons in mediating leptin’s metabolic effects is currently unknown. This leptin-mediated brain action plays a crucial role in regulating appetite and glucose homeostasis (Berglund et al, 2012; Campfield et al, 1995), in addition to controlling insulin sensitivity in peripheral tissues via a neurocircuit involving the vagus nerve (German et al, 2009). Importantly, leptin not only modulates the neuronal activity of hypothalamic neurons. The LepR has also been identified in astrocytes, whose activation controls synaptic remodeling and food intake (Kim et al, 2014).
Another important adipocyte-derived hormone is adiponectin (Scherer et al, 1995). Adiponectin activates its receptors, AdipoR1 and AdipoR2, expressed on various nuclei of the MBH (Guillod-Maximin et al, 2009; Kubota et al, 2007), modulating food intake (Kubota et al, 2007; Coope et al, 2008), energy metabolism and body weight (Qi et al, 2004), bone metabolism (Kajimura et al, 2013), and glucose metabolism (Koch et al, 2014). Adiponectin can positively or negatively affect food intake by acting on POMC neurons, an effect that depends on the energy state or glucose concentration in the brain (Suyama et al, 2016). Interestingly, POMC but not neuropeptide Y (NPY) neurons are sensitive to glucose concentration. Indeed, adiponectin also enhances inhibitory postsynaptic currents onto NPY neurons, attenuating their firing rate independently of glucose levels (Suyama et al, 2017). Adiponectin also acts on oxytocin neurons by inhibiting their excitability, suggesting a potential role in influencing energy homeostasis through the control of pituitary hormone secretion (Hoyda et al, 2007). In addition, adiponectin influences hypothalamic-controlled circadian rhythms via AdipoR1-mediated upregulation of the core clock gene Bmal1, controlling orexigenic neuropeptide expression and food intake (Tsang et al, 2020). Similarly to leptin, adiponectin also triggers catabolic processes in astrocytes, having a positive impact on nutrient availability in the hypothalamus (Song et al, 2021). These findings suggest that adiponectin plays a crucial role in modulating hypothalamic activity and energy homeostasis through multiple mechanisms modulating the function of neurons and non-neuronal populations. Nonetheless, adiponectin not only acts on the hypothalamus in the brain. Indeed, AdipoR1 and AdipoR2 have also been identified in the nucleus of the solitary tract, where they decrease blood pressure by modulating the excitability of NPY neurons, thus influencing central autonomic processing (Hoyda et al, 2009).
The WAT also communicates with the brain through resistin, an adipokine that is produced in adipocytes in rodents and adipose tissue-infiltrated macrophages in humans, and whose levels correlate with adiposity. Its name derives from its action to induce resistance to insulin (Steppan et al, 2001); additionally, this adipokine influences metabolic regulation and inflammatory responses by acting on the hypothalamus. Indeed, although a specific resistin receptor has not yet been identified, TLR4 has been recognized as a binding site for resistin, where it activates a TLR4-mediated signaling pathway involved in resistin-induced inflammation and insulin resistance (Benomar et al, 2013). Resistin acts in the brain increasing sympathetic nerve activity, affecting the cardiovascular system, but inhibits sympathetic nerve activity to BAT (Steppan et al, 2001). The literature is still limited, however, crosstalk between leptin and resistin on metabolic function has been described, particularly through their action on the hypothalamus. Evidence indicates that resistin reduces the action of leptin on food intake and has opposing actions on insulin sensitivity. However, the precise mechanisms that underlie this crosstalk are still a matter of investigation.
Mounting evidence has shown that a growing list of adipokines produced by the WAT is involved in energy and metabolic homeostasis through their actions on the brain. Apelin, acting on its receptor (APJ), exerts pleiotropic effects that include glucose homeostasis, control of blood pressure, nutritional behavior, and the secretion of pituitary hormones (Mehri et al, 2023). Nonetheless, it is important to mention that apelin is produced by diverse tissues and cell types, including hypothalamic POMC neurons (Reaux-Le Goazigo et al, 2011), and as such it is difficult to determine the specific impact of WAT-derived apelin in WAT–brain communication. Additional adipokines, such as omentin-1 and retinol-binding protein 4 (RBP4), have been proposed to act on the hypothalamus. However, to date, these hypothalamic effects appear to be secondary to their peripheral actions (Brunetti et al, 2011).
Metabolic regulation of the adipose–brain axis (fatty acids)
WAT also secretes various species of lipids into the circulation to communicate with the brain (Fig. 2). Fatty acids are released from adipocytes into the bloodstream, often bound to albumin or incorporated as triglycerides within lipoproteins. As they circulate, they can cross the blood-brain barrier (BBB) through diverse transport mechanisms, including passive diffusion or facilitated transport (Pifferi et al, 2021). The BBB has specific transport proteins that help shuttle fatty acids into the brain, among these are fatty acid transport proteins 1–6 (FATP1-6), fatty acid translocase/CD36 (FAT/CD36), intracellular fatty acid binding proteins 1–9 (FABP1-9), plasma membrane fatty acid binding protein (FABPpm), and caveolin-1; for a recent review see (Cruciani-Guglielmacci et al, 2024). More recently, an additional fatty acid transport protein called Mfsd2a has been identified, which specifically binds and transports the long-chain unsaturated fatty acids docosahexaenoic acid (DHA) and α-linolenic acid (ALA), using a unique flipping mechanism (Nguyen et al, 2023; Chua et al, 2023; Nguyen et al, 2014a). Once in the brain, fatty acids can be esterified and/or incorporated into membrane phospholipids (Rapoport et al, 2001). Nonetheless, they can also function as signaling molecules by binding to membrane receptors or by functioning as intracellular messengers modulating signal transduction. One of the first and most studied metabolic functions of fatty acid sensing is the control of energy and glucose homeostasis. Specialized neurons in the hypothalamus, including AgRP and POMC neurons, detect fluctuations in fatty acid concentration and modulate their activity, thereby orchestrating behavioral and metabolic responses (Cruciani-Guglielmacci et al, 2024). More than two decades ago, the team of Rossetti showed that oleic acid, acting on the brain, inhibits glucose production and food intake (Obici et al, 2002), an effect mediated by FAT/CD36 (Le Foll et al, 2013) and acyl-CoA synthesis (Moullé et al, 2013). More recently, other fatty acid receptors involved in metabolic regulation have been identified in hypothalamic neurons: free fatty acid receptor-4 (FFAR4) and -1 (FFAR1) (Dragano et al, 2017). While FFAR4 has been mainly associated with anti-inflammatory functions (Dragano et al, 2017), FFAR1 in hypothalamic POMC neurons controls food intake and body weight by mechanisms that are still unclear (Dragano et al, 2024). Fatty acid-mediated FFAR1 activation increases intracellular Ca2+ levels in hypothalamic neurons, which could affect neuronal activity as well as different cellular processes. Indeed, palmitic acid-mediated activation of FFAR1 in hypothalamic neurons inhibits autophagy, which is considered an important factor in the control of food intake (Hernández-Cáceres et al, 2019; Quan et al, 2012).
Hypothalamic fatty acid sensing also influences the function of several hormones. Fatty acid signaling in the hypothalamus regulates insulin secretion and action, a process dependent on fatty acid β-oxidation and the enzyme carnitine palmitoyl transferase 1 (CPT1) (Migrenne et al, 2006), which regulates the entrance of fatty acids into mitochondria (Obici et al, 2003). Fatty acids in the VMH stimulate the orexigenic effects of ghrelin on food intake, which involves the inhibition of fatty acid biosynthesis induced by AMPK and concomitant activation of CPT1 (López et al, 2008). Fatty acid sensing is also involved in the hepatic secretion of triglyceride-rich lipoproteins (VLDL-TG). Infusion of oleic acid into the MBH activates a PKC-δ → KATP-channel signaling axis, which relies on the dorsal vagal complex (DVC) and hepatic innervation leading to the suppression of VLDL-TG secretion in rats (Yue et al, 2015). Although much of the literature has focused on the role of neurons in lipid sensing, mounting evidence highlights the involvement of hypothalamic astrocytes and tanycytes in fatty acid detection and metabolic actions (Hofmann et al, 2017). However, the mechanisms underlying these processes and whether this information is subsequently transmitted to specific hypothalamic neurons still require further investigation.
Immune system in the regulation of the brain–adipose axis
The innate immune system plays a critical role in adipose tissue function (Kawai et al, 2021). Among the various immune cells in WAT, macrophages are the most abundant, accounting for more than half of the leukocytes in both lean and obese animals. Their accumulation in WAT is closely associated with obesity (Weisberg et al, 2003). Evidence suggests a bidirectional communication between the CNS and resident immune cells in WAT, influencing lipid homeostasis. Consequently, the immune system may play a key role in mediating the crosstalk between the brain and WAT. This mediation is likely facilitated by the presence of adrenergic receptors on macrophages, specifically β2-AR (Petkevicius et al, 2021) and their ability to regulate sympathetic innervation of WAT (Rahman and Jun, 2022).
Stimulation with a β-adrenergic agonist induces adipose tissue remodeling, triggering a beigeing process that involves adipocyte cell death and their clearance by a coordinated network of non-inflammatory M2 macrophages (Lee et al, 2013, 2016). In addition, cold exposure can induce the secretion of molecules from macrophages that modulate WAT sympathetic innervation. The cytokine Slit3, secreted by M2 macrophages, increases adipose tissue sympathetic activity via the Slit-ROBO pathway, enhancing thermogenesis and increasing the synthesis and release of NE (Wang et al, 2021). BDNF is another molecule secreted by macrophages in response to cold exposure. Studies using various models of BDNF modulation (knockdown and overexpression) (Wang et al, 2020b; Cao et al, 2011, 2009) suggest that BDNF contributes to WAT innervation, potentially through the highly expressed TrkB receptor found in sensory and sympathetic nerves (Blaszkiewicz et al, 2020). In conclusion, while the immune system’s role in regulating the brain–adipose axis shows promising potential, our understanding remains limited. Further research is needed to fully uncover these complex interactions and their implications for metabolic regulation.
Brown adipose tissue → brain crosstalk
Sensory innervation
Sensory neurons originating in the BAT extend their axons to the DRG (Fig. 2), as anatomical studies in rodents have shown that sensory projections in C4–C8/T1–T2 DRG were traced from BAT (Ryu et al, 2015). Studies using anterograde transneuronal viral tracing have revealed that sensory nerve projections from BAT primarily reach the brainstem, forebrain, and hypothalamus, targeting nuclei that in turn regulate sympathetic output to adipose tissues (Vaughan and Bartness, 2012). WAT exhibits more extensive sensory innervation (Wang et al, 2022b), however, studies that modulate sensory activity in BAT underscored the critical role of these afferent signals. A major focus in sensory nerve research has been on transient receptor potential (TRP) channels, which include receptors from all five types. Among these, TRPV1 has been particularly investigated in a metabolic context due to its capacity to be activated by capsaicin (a compound found in hot chili peppers) (Caterina et al, 1997) (Fig. 2). TRPV1 activation in human randomized controlled trials enhances energy expenditure through BAT thermogenesis and reduces food intake, resulting in a negative energy balance and decreased adiposity (Inoue et al, 2007; Snitker et al, 2009; Yoshioka et al, 2001). In line with this, global double-knockout TRPV1/UCP1 mice show reduced BAT mass and mitochondrial respiration, leading to lower whole-body oxygen consumption and heat production resulting in an obese phenotype (Li et al, 2022). The observed phenotype was more pronounced than the single UCP1 knockout mouse, suggesting a synergistic effect (Li et al, 2022). Together, these results suggest that BAT sensory innervation could be a potential target for anti-obesity drug therapies (Ohyama et al, 2016).
Endocrine regulation (batokines)
Traditionally, BAT has been studied for its key role in thermogenesis. However, in recent years, the secretory function of BAT has gained significant attention for its impact on interorgan communication (Fig. 2). The secreted molecules, known as “brown adipokines” or “batokines,” target distant organs and tissues, including the liver, heart, skeletal muscle, WAT, and brain. They exert their effects through autocrine, paracrine, or endocrine mechanisms (Ahmad et al, 2021). Among these batokines, neuregulin-4 (NRG-4) is highly expressed in BAT compared with WAT (Chen et al, 2017; Christian, 2015) and has been shown to positively influence energy balance, lipid metabolism, and glucose metabolism (Chen et al, 2017; Wang et al, 2014). NRG-4 also promotes sympathetic neurite outgrowth (Pellegrinelli et al, 2018; Rosell et al, 2014), as is also the case for nerve growth factor (NGF) (Néchad et al, 1994; Nisoli et al, 1996).
Another batokine implicated in brain crosstalk is bone morphogenetic protein 8b (BMP-8b), which increases in response to thermogenic and nutritional factors, such as cold exposure or an HFD (Whittle et al, 2012). BMP-8b not only acts autocrinally within the BAT but also plays an endocrine role in the hypothalamus, thereby enhancing BAT thermogenesis and increasing energy expenditure through the SNS (Pellegrinelli et al, 2018; Whittle et al, 2012). BAT also secretes FGF-21, GDF-15, and IL-6, which have been associated with various effects on the brain (Ahmad et al, 2021). However, the specific contribution of these factors when derived from BAT in the context of BAT-brain communication warrants further investigation.
Collectively, these findings emphasize the secretory function of BAT, redefining our understanding of this tissue beyond its role in thermogenesis. This positions BAT as an endocrine organ that influences metabolism through inter-tissue communication. This expanded perspective highlights BAT’s potential as a target for therapeutic intervention against metabolic disorders (Ahmad et al, 2021).
New modes of communication
Emerging research has unveiled novel communication mechanisms between adipose tissue and the brain, influencing physiological processes beyond metabolism and energy balance. In this section, we delve into the nature and diversity of these means of communication, which add another layer of complexity to the intricate interorgan crosstalk.
Neuro-mesenchymal units
Neuro-mesenchymal units refer to specialized cellular networks that involve communication and interactions between nervous system components and mesenchymal cells. Recent research in adipose tissue biology has highlighted the cooperation between sympathetic neurons and immune cells in maintaining metabolism (Cardoso et al, 2021). Cardoso and collaborators discovered that sympathetic terminals control the expression of glial-derived neurotrophic factor (GDNF) and the activity of type-2 innate lymphoid cells (ILC2s) through β2-ARs in adjacent adipose mesenchymal cells. Disruption of this communication alters the generation of cytokines, thus contributing to metabolic imbalance (Cardoso et al, 2021). This neuroimmune regulation is anatomically and functionally mediated by multiple brain areas, including the PVH which plays a crucial role in systemic metabolism and selectively influences sympathetic output to adipose tissue in a depot-specific manner (Nguyen et al, 2014b). While our current understanding of neuro-mesenchymal units is still developing, this recent research underscores their relevance as a dynamic communication platform for regulating energy balance.
Extracellular vesicles
In addition to traditional soluble factors, adipose tissue also secretes extracellular vesicles (EVs). These EVs are lipid bilayer nanoparticles that function as physiological intercellular and interorgan communication vehicles, facilitating the transfer of diverse bioactive cargos (proteins, lipids, diverse RNA species, especially miRNAs, organelle parts/proteins, etc.) that contribute to a wide range of metabolic processes (Isaac et al, 2021). Given the cellular heterogeneity of adipose tissue, EVs can be released by adipocytes, macrophages, adipose stem cells, or endothelial cells (Isaac et al, 2021). The content of EVs is influenced by nutritional or pathophysiological conditions (obesity, T2D) reflecting the metabolic state of the adipose tissue and partially mediating the systemic effects observed in these conditions (Isaac et al, 2021). For example, EVs from obese or diabetic individuals often carry altered molecular signatures that can exacerbate inflammation and insulin resistance, while those from individuals in a fasted or refeed state may promote metabolic adaptations beneficial for maintaining energy homeostasis (Crewe et al, 2018). Numerous reports evidenced EV-mediated communication between the adipose tissue and several organs, such as the pancreas, skeletal muscle, liver, and heart (Crewe and Scherer, 2022). However, much less is known about their interactions with the brain. Gao et al reported that adipocytes from obese mice secrete EVs that can be taken up by POMC-like neurons in vitro (Gao et al, 2020). When these EVs were transferred to lean mice, they promoted appetite and body weight gain. These effects were believed to occur through the transfer of miRNAs and lncRNAs to POMC neurons, thereby stimulating the mTORC1 signaling pathway (Gao et al, 2020). Conversely, EVs from adipocytes of lean mice reduced appetite and mitigated weight gain in mice fed with HFD by decreasing mTORC1 signaling (Gao et al, 2020). Another study demonstrated that extracellular nicotinamide phosphoribosyltransferase (eNAMPT), an enzyme implicated in the biosynthesis of NAD+ , is transported in EVs through systemic circulation (Yoshida et al, 2019). The authors found that eNAMPT declined with age (in both mice and humans), and that genetic adipose-specific overexpression of eNAMPT increased NAD+ levels in diverse tissues including the hypothalamus. This was correlated with improved function and antiaging effects in female mice (Yoshida et al, 2019). Interestingly, a specific population of neurons located in the DMH has been shown to influence aging and lifespan via the release of adipose eNAMPT via the SNS (Tokizane et al, 2024). These results suggest a bidirectional brain–adipose tissue communication to maintain NAD+ levels and healthy aging.
Obesity and T2D are associated with cognitive decline (Biessels and Despa, 2018). Control mice treated with adipose tissue-derived EVs from HFD-fed mice or patients with diabetes exhibited a synaptic loss in the hippocampus and cognitive impairments (Wang et al, 2022a). An altered EV miRNA cargo was responsible for these alterations. Specifically, the enrichment of miR-9-3p negatively regulated the expression of BDNF in the hippocampus, which is key for synaptic function (Wang et al, 2022a). Together, these results unveil an EV-mediated communication mode between adipose tissue and the brain implicated in cognitive dysfunction in the context of metabolic disorders.
Mechanical forces
Recently, research has been focusing on signaling mechanisms beyond nervous and chemical signals in the control of the adipose–brain axis. A hot topic in this area is the importance of mechanical forces, which are currently considered a key factor for the maintenance of WAT health (De Luca et al, 2021). It is tempting to speculate that mechanical forces might control the production and release of adipokines, thereby playing a role in tissue communication.
Consequences of brain–adipose tissue axis dysfunction
Environmental factors (e.g., imbalanced diets, exposure to toxins, sedentary lifestyle, etc.) can significantly impair organ function by causing metabolic alterations, inflammation, or oxidative stress. These disruptions not only affect individual organs but also the communication routes that coordinate systemic processes, resulting in a cascade of adverse health outcomes (Fig. 3).
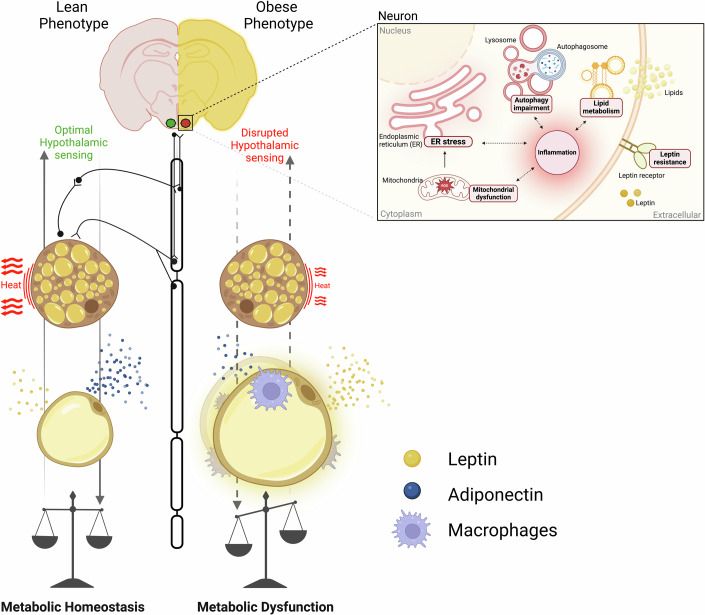
Figure 3. Brain–Adipose tissue communication in health and obesity.
The hypothalamic sensing and adipose tissue function between lean and obese phenotypes is illustrated. On the left, the lean phenotype is characterized by optimal hypothalamic sensing, efficient thermogenesis in brown adipose tissue (BAT), and healthy white adipose tissue (WAT) function, converging in systemic a metabolic homeostasis. On the right, the obese phenotype is associated with disrupted hypothalamic sensing, leading to impaired thermogenesis in BAT and dysfunctional WAT. The disrupted sensing results in reduced energy expenditure and altered adipokine secretion, including elevated leptin levels and reduced adiponectin. This condition is also characterized by increased macrophage infiltration into WAT, contributing to chronic inflammation and further metabolic dysregulation. At the neuronal level, the obese phenotype is characterized by heightened inflammation, which may lead to mitochondrial dysfunction, endoplasmic reticulum (ER) stress, disrupted lipid metabolism, and impaired autophagy. These cellular disturbances ultimately contribute to the development of leptin resistance.
WAT is particularly vulnerable to environmental aggressions. A paradigmatic example is the predominant Western lifestyle, characterized by the consumption of obesogenic diets and sedentarism. Energy excess leads to fat accumulation primarily through adipocyte hypertrophy, which is associated with adipose tissue inflammation, fibrosis, and hypoxia, ultimately contributing to poor metabolic health (Sakers et al, 2022). This dysfunctional fat also produces lower levels of adiponectin (an insulin-sensitizing adipokine) and higher levels of leptin (which is produced in proportion to fat stores) (Sakers et al, 2022). In the context of obesity, elevated leptin levels are frequently associated with leptin resistance, a condition marked by the inability to respond to the anorexigenic effects of this hormone. Evidence indicates that hyperleptinemia is required for the development of leptin resistance (Knight et al, 2010), which evolves through the overactivation of leptin signaling in key hypothalamic areas that eventually promote a negative feedback regulation via SOCS-3 (Ernst et al, 2009). This blunts LepR signaling and its appetite-suppressing effects, thus sustaining increased food intake and body weight. Other mechanisms potentially underlying leptin resistance include various molecular and functional alterations associated with structural changes to the molecule, its transport across the BBB, and the deterioration of leptin-receptor function and signaling.
Obesogenic diets also have direct effects on the hypothalamus. Excessive fat and sugar consumption may cause mitochondrial dysfunction (Gonzalez-Franquesa et al, 2022), endoplasmic reticulum (ER) stress (Zhang et al, 2008), autophagy impairment (Meng and Cai, 2011), lipid metabolism deregulation (González-García et al, 2017), and inflammation (De Souza et al, 2005) in the hypothalamus, leading to alterations in neuronal function and contributing to the development of obesity. These pathological processes are closely interrelated and can influence each other in an intricate and synergetic manner. Their combined and sustained effects promote in the hypothalamus accumulation of some types of lipids, such as palmitate or ceramides, enhancing local oxidative stress, the release of cytokines and inflammatory mediators, as well as ER malfunction (Rodriguez-Navas et al, 2016; Contreras et al, 2014). This results in defective neuropeptide expression (e.g., Npy, Agrp) and processing (e.g., POMC), the deterioration of metabolic signaling pathways (e.g., leptin and insulin), and apoptosis of certain neuronal mediators (Ozcan et al, 2009; Cakir et al, 2013; Schneeberger et al, 2013; Williams et al, 2014; Moraes et al, 2009; Yi et al, 2017). Collectively, this promotes increased appetite and altered communication with WAT and BAT, interfering with lipolysis and thermogenesis, and ultimately contributing to the development of obesity. While most of our current understanding of these processes comes from rodent models, evidence suggests that these findings may also be relevant to humans (Lei et al, 2024; Thaler et al, 2012; Kreutzer et al, 2017).
Conclusions
The bidirectional communication between the brain and adipose tissue is multifaceted and complex, encompassing multiple nervous and hormonal pathways as well as novel and unexpected routes of communication. These multiple layers of signaling enhance the system’s ability to fine-tune metabolic processes, integrate diverse metabolic signals, and respond effectively to varying physiological conditions and disruptions, thereby maintaining overall physiological stability and promoting health. While research on this topic has started to unveil a clearer picture of the interactions involved, there is still much to discover about their nature, molecular mechanisms, functions, and the consequences of their disruption. As research advances, further elucidation of these interactions may lead to novel strategies for managing metabolic diseases and enhancing overall metabolic health.
Supplementary information
Acknowledgements
This work was supported by the Departament de Recerca i Universitats de la Generalitat de Catalunya (2021-SGR-01320) and the CERCA Programme/Generalitat de Catalunya (to MC) and by the grant from Fondo Nacional de Desarrollo Científico y Tecnológico, FONDECYT 1240623 (to EM). FD-C is a recipient of a fellowship from the Agencia Nacional de Investigación y Desarrollo (ANID), Doctorado Nacional 21210611, Chile, and Scientific Exchange Grant 10594 from the European Molecular Biology Organization (EMBO), Germany. This work was carried out in part at the Esther Koplowitz Centre, Barcelona. Figures were created using BioRender (BioRender.com).
Author contributions
Francisco Díaz-Castro: Conceptualization; Visualization; Writing—original draft; Writing—review and editing. Eugenia Morselli: Conceptualization; Visualization; Writing—original draft; Writing—review and editing. Marc Claret: Conceptualization; Visualization; Writing—original draft; Writing—review and editing.
Disclosure and competing interests statement
The authors declare no competing interests.
Contributor Information
Eugenia Morselli, Email: eugenia.morselli@uss.cl.
Marc Claret, Email: mclaret@recerca.clinic.cat.
Peer review information
A peer review file is available at 10.1038/s44319-024-00321-4
References
- Adler ES, Hollis JH, Clarke IJ, Grattan DR, Oldfield BJ (2012) Neurochemical characterization and sexual dimorphism of projections from the brain to abdominal and subcutaneous white adipose tissue in the rat. J Neurosci 32:15913–15921 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahmad B, Vohra MS, Saleemi MA, Serpell CJ, Fong IL, Wong EH (2021) Brown/Beige adipose tissues and the emerging role of their secretory factors in improving metabolic health: the batokines. Biochimie 184:26–39 [DOI] [PubMed] [Google Scholar]
- Bamshad M, Aoki VT, Adkison MG, Warren WS, Bartness TJ (1998) Central nervous system origins of the sympathetic nervous system outflow to white adipose tissue. Am J Physiol 275:R291–299 [DOI] [PubMed] [Google Scholar]
- Bartness TJ, Liu Y, Shrestha YB, Ryu V (2014) Neural innervation of white adipose tissue and the control of lipolysis. Front Neuroendocrinol 35:473–493 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartness TJ, Vaughan CH, Song CK (2010) Sympathetic and sensory innervation of brown adipose tissue. Int J Obes 34(Suppl 1):S36–42 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beiroa D, Imbernon M, Gallego R, Senra A, Herranz D, Villarroya F, Serrano M, Fernø J, Salvador J, Escalada J et al (2014) GLP-1 agonism stimulates brown adipose tissue thermogenesis and browning through hypothalamic AMPK. Diabetes 63:3346–3358 [DOI] [PubMed] [Google Scholar]
- Benomar Y, Gertler A, De Lacy P, Crépin D, Ould Hamouda H, Riffault L, Taouis M (2013) Central resistin overexposure induces insulin resistance through Toll-like receptor 4. Diabetes 62:102–114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berglund ED, Vianna CR, Donato J, Kim MH, Chuang J-C, Lee CE, Lauzon DA, Lin P, Brule LJ, Scott MM et al (2012) Direct leptin action on POMC neurons regulates glucose homeostasis and hepatic insulin sensitivity in mice. J Clin Investig 122:1000–1009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bianco AC, Silva JE (1987) Intracellular conversion of thyroxine to triiodothyronine is required for the optimal thermogenic function of brown adipose tissue. J Clin Investig 79:295–300 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biessels GJ, Despa F (2018) Cognitive decline and dementia in diabetes mellitus: mechanisms and clinical implications. Nat Rev Endocrinol 14:591–604 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blaszkiewicz M, Wood E, Koizar S, Willows J, Anderson R, Tseng Y-H, Godwin J, Townsend KL (2020) The involvement of neuroimmune cells in adipose innervation. Mol Med 26:126 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bowers RR, Festuccia WTL, Song CK, Shi H, Migliorini RH, Bartness TJ (2004) Sympathetic innervation of white adipose tissue and its regulation of fat cell number. Am J Physiol Regul Integr Comp Physiol 286:R1167–1175 [DOI] [PubMed] [Google Scholar]
- Brunetti L, Di Nisio C, Recinella L, Chiavaroli A, Leone S, Ferrante C, Orlando G, Vacca M (2011) Effects of vaspin, chemerin and omentin-1 on feeding behavior and hypothalamic peptide gene expression in the rat. Peptides 32:1866–1871 [DOI] [PubMed] [Google Scholar]
- Brüning JC, Fenselau H (2023) Integrative neurocircuits that control metabolism and food intake. Science 381:eabl7398 [DOI] [PubMed] [Google Scholar]
- Buettner C, Muse ED, Cheng A, Chen L, Scherer T, Pocai A, Su K, Cheng B, Li X, Harvey-White J et al (2008) Leptin controls adipose tissue lipogenesis via central, STAT3-independent mechanisms. Nat Med 14:667–675 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cakir I, Cyr NE, Perello M, Litvinov BP, Romero A, Stuart RC, Nillni EA (2013) Obesity induces hypothalamic endoplasmic reticulum stress and impairs proopiomelanocortin (POMC) post-translational processing. J Biol Chem 288:17675–17688 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campfield LA, Smith FJ, Guisez Y, Devos R, Burn P (1995) Recombinant mouse OB protein: evidence for a peripheral signal linking adiposity and central neural networks. Science 269:546–549 [DOI] [PubMed] [Google Scholar]
- Cannon B, Nedergaard J (2004) Brown adipose tissue: function and physiological significance. Physiol Rev 84:277–359 [DOI] [PubMed] [Google Scholar]
- Cao L, Choi EY, Liu X, Martin A, Wang C, Xu X, During MJ (2011) White to brown fat phenotypic switch induced by genetic and environmental activation of a hypothalamic-adipocyte axis. Cell Metab 14:324–338 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao L, Lin E-JD, Cahill MC, Wang C, Liu X, During MJ (2009) Molecular therapy of obesity and diabetes by a physiological autoregulatory approach. Nat Med 15:447–454 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cardinal P, Bellocchio L, Clark S, Cannich A, Klugmann M, Lutz B, Marsicano G, Cota D (2012) Hypothalamic CB1 cannabinoid receptors regulate energy balance in mice. Endocrinology 153:4136–4143 [DOI] [PubMed] [Google Scholar]
- Cardoso F, Klein Wolterink RGJ, Godinho-Silva C, Domingues RG, Ribeiro H, da Silva JA, Mahú I, Domingos AI, Veiga-Fernandes H (2021) Neuro-mesenchymal units control ILC2 and obesity via a brain-adipose circuit. Nature 597:410–414 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caterina MJ, Schumacher MA, Tominaga M, Rosen TA, Levine JD, Julius D (1997) The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature 389:816–824 [DOI] [PubMed] [Google Scholar]
- Cavalcanti-de-Albuquerque JP, Bober J, Zimmer MR, Dietrich MO (2019) Regulation of substrate utilization and adiposity by Agrp neurons. Nat Commun 10:311 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Celi FS, Brychta RJ, Linderman JD, Butler PW, Alberobello AT, Smith S, Courville AB, Lai EW, Costello R, Skarulis MC et al (2010) Minimal changes in environmental temperature result in a significant increase in energy expenditure and changes in the hormonal homeostasis in healthy adults. Eur J Endocrinol 163:863–872 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen KY, Brychta RJ, Linderman JD, Smith S, Courville A, Dieckmann W, Herscovitch P, Millo CM, Remaley A, Lee P et al (2013) Brown fat activation mediates cold-induced thermogenesis in adult humans in response to a mild decrease in ambient temperature. J Clin Endocrinol Metab 98:E1218–1223 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Z, Wang G-X, Ma SL, Jung DY, Ha H, Altamimi T, Zhao X-Y, Guo L, Zhang P, Hu C-R et al (2017) Nrg4 promotes fuel oxidation and a healthy adipokine profile to ameliorate diet-induced metabolic disorders. Mol Metab 6:863–872 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christian M (2015) Transcriptional fingerprinting of ‘browning’ white fat identifies NRG4 as a novel adipokine. Adipocyte 4:50–54 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chua G-L, Tan BC, Loke RYJ, He M, Chin C-F, Wong BH, Kuk ACY, Ding M, Wenk MR, Guan L et al (2023) Mfsd2a utilizes a flippase mechanism to mediate omega-3 fatty acid lysolipid transport. Proc Natl Acad Sci USA 120:e2215290120 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohade C, Osman M, Pannu HK, Wahl RL (2003) Uptake in supraclavicular area fat (‘USA-Fat’): description on 18F-FDG PET/CT. J Nucl Med 44:170–176 [PubMed] [Google Scholar]
- Commins SP, Watson PM, Padgett MA, Dudley A, Argyropoulos G, Gettys TW (1999) Induction of uncoupling protein expression in brown and white adipose tissue by leptin. Endocrinology 140:292–300 [DOI] [PubMed] [Google Scholar]
- Contreras C, González-García I, Martínez-Sánchez N, Seoane-Collazo P, Jacas J, Morgan DA, Serra D, Gallego R, Gonzalez F, Casals N et al (2014) Central ceramide-induced hypothalamic lipotoxicity and ER stress regulate energy balance. Cell Rep 9:366–377 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Contreras C, Nogueiras R, Diéguez C, Rahmouni K, López M (2017) Traveling from the hypothalamus to the adipose tissue: the thermogenic pathway. Redox Biol 12:854–863 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coope A, Milanski M, Araújo EP, Tambascia M, Saad MJA, Geloneze B, Velloso LA (2008) AdipoR1 mediates the anorexigenic and insulin/leptin-like actions of adiponectin in the hypothalamus. FEBS Lett 582:1471–1476 [DOI] [PubMed] [Google Scholar]
- Correll JW (1963) Adipose tissue: ability to respond to nerve stimulation in vitro. Science 140:387–388 [DOI] [PubMed] [Google Scholar]
- Côté I, Sakarya Y, Green SM, Morgan D, Carter CS, Tümer N, Scarpace PJ (2018) iBAT sympathetic innervation is not required for body weight loss induced by central leptin delivery. Am J Physiol Endocrinol Metab 314:E224–E231 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cousin B, Casteilla L, Lafontan M, Ambid L, Langin D, Berthault MF, Pénicaud L (1993) Local sympathetic denervation of white adipose tissue in rats induces preadipocyte proliferation without noticeable changes in metabolism. Endocrinology 133:2255–2262 [DOI] [PubMed] [Google Scholar]
- Cowley MA, Smart JL, Rubinstein M, Cerdán MG, Diano S, Horvath TL, Cone RD, Low MJ (2001) Leptin activates anorexigenic POMC neurons through a neural network in the arcuate nucleus. Nature 411:480–484 [DOI] [PubMed] [Google Scholar]
- Crewe C, Joffin N, Rutkowski JM, Kim M, Zhang F, Towler DA, Gordillo R, Scherer PE (2018) An endothelial-to-adipocyte extracellular vesicle axis governed by metabolic state. Cell 175:695–708.e13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crewe C, Scherer PE (2022) Intercellular and interorgan crosstalk through adipocyte extracellular vesicles. Rev Endocr Metab Disord 23:61–69 [DOI] [PubMed] [Google Scholar]
- Cruciani-Guglielmacci C, Le Stunff H, Magnan C (2024) Brain lipid sensing and the neural control of energy balance. Biochimie 223:159–165 [DOI] [PubMed] [Google Scholar]
- Cypess AM, Lehman S, Williams G, Tal I, Rodman D, Goldfine AB, Kuo FC, Palmer EL, Tseng Y-H, Doria A et al (2009) Identification and importance of brown adipose tissue in adult humans. New Engl J Med 360:1509–1517 [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Luca M, Mandala M, Rose G (2021) Towards an understanding of the mechanoreciprocity process in adipocytes and its perturbation with aging. Mech Ageing Dev 197:111522 [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Solis AJ, Del Río-Martín A, Radermacher J, Chen W, Steuernagel L, Bauder CA, Eggersmann FR, Morgan DA, Cremer A-L, Sué M et al (2024) Reciprocal activity of AgRP and POMC neurons governs coordinated control of feeding and metabolism. Nat Metab 6:473–493 [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Souza CT, Araujo EP, Bordin S, Ashimine R, Zollner RL, Boschero AC, Saad MJA, Velloso LA (2005) Consumption of a fat-rich diet activates a proinflammatory response and induces insulin resistance in the hypothalamus. Endocrinology 146:4192–4199 [DOI] [PubMed] [Google Scholar]
- Dib B, Rompré PP, Amir S, Shizgal P (1994) Thermogenesis in brown adipose tissue is activated by electrical stimulation of the rat dorsal raphe nucleus. Brain Res 650:149–152 [DOI] [PubMed] [Google Scholar]
- Dragano NRV, Milbank E, Haddad-Tóvolli R, Garrido-Gil P, Nóvoa E, Fondevilla MF, Capelli V, Zanesco AM, Solon C, Morari J et al (2024) Hypothalamic free fatty acid receptor-1 regulates whole-body energy balance. Mol Metab 79:101840 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dragano NRV, Solon C, Ramalho AF, de Moura RF, Razolli DS, Christiansen E, Azevedo C, Ulven T, Velloso LA (2017) Polyunsaturated fatty acid receptors, GPR40 and GPR120, are expressed in the hypothalamus and control energy homeostasis and inflammation. J Neuroinflammation 14:91 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elmquist JK, Bjørbaek C, Ahima RS, Flier JS, Saper CB (1998) Distributions of leptin receptor mRNA isoforms in the rat brain. J Comp Neurol 395:535–547 [PubMed] [Google Scholar]
- Ernst MB, Wunderlich CM, Hess S, Paehler M, Mesaros A, Koralov SB, Kleinridders A, Husch A, Münzberg H, Hampel B et al (2009) Enhanced Stat3 activation in POMC neurons provokes negative feedback inhibition of leptin and insulin signaling in obesity. J Neurosci 29:11582–11593 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fischer AW, Cannon B, Nedergaard J (2020) Leptin: is it thermogenic? Endocr Rev 41:232–260 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fischer AW, Hoefig CS, Abreu-Vieira G, de Jong JMA, Petrovic N, Mittag J, Cannon B, Nedergaard J (2016) Leptin raises defended body temperature without activating thermogenesis. Cell Rep 14:1621–1631 [DOI] [PubMed] [Google Scholar]
- Fishman RB, Dark J (1987) Sensory innervation of white adipose tissue. Am J Physiol 253:R942–944 [DOI] [PubMed] [Google Scholar]
- Gao J, Li X, Wang Y, Cao Y, Yao D, Sun L, Qin L, Qiu H, Zhan X (2020) Adipocyte-derived extracellular vesicles modulate appetite and weight through mTOR signalling in the hypothalamus. Acta Physiol 228:e13339 [DOI] [PubMed] [Google Scholar]
- German J, Kim F, Schwartz GJ, Havel PJ, Rhodes CJ, Schwartz MW, Morton GJ (2009) Hypothalamic leptin signaling regulates hepatic insulin sensitivity via a neurocircuit involving the vagus nerve. Endocrinology 150:4502–4511 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giordano A, Morroni M, Santone G, Marchesi GF, Cinti S (1996) Tyrosine hydroxylase, neuropeptide Y, substance P, calcitonin gene-related peptide and vasoactive intestinal peptide in nerves of rat periovarian adipose tissue: an immunohistochemical and ultrastructural investigation. J Neurocytol 25:125–136 [DOI] [PubMed] [Google Scholar]
- Giordano A, Song CK, Bowers RR, Ehlen JC, Frontini A, Cinti S, Bartness TJ (2006) White adipose tissue lacks significant vagal innervation and immunohistochemical evidence of parasympathetic innervation. Am J Physiol Regul Integr Comp Physiol 291:R1243–1255 [DOI] [PubMed] [Google Scholar]
- Gómez-Valadés AG, Pozo M, Varela L, Boudjadja MB, Ramírez S, Chivite I, Eyre E, Haddad-Tóvolli R, Obri A, Milà-Guasch M et al (2021) Mitochondrial cristae-remodeling protein OPA1 in POMC neurons couples Ca2+ homeostasis with adipose tissue lipolysis. Cell Metab 33:1820–1835.e9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonzalez-Franquesa A, Gama-Perez P, Kulis M, Szczepanowska K, Dahdah N, Moreno-Gomez S, Latorre-Pellicer A, Fernández-Ruiz R, Aguilar-Mogas A, Hoffman A et al (2022) Remission of obesity and insulin resistance is not sufficient to restore mitochondrial homeostasis in visceral adipose tissue. Redox Biol 54:102353 [DOI] [PMC free article] [PubMed] [Google Scholar]
- González-García I, Fernø J, Diéguez C, Nogueiras R, López M (2017) Hypothalamic lipids: key regulators of whole body energy balance. Neuroendocrinology 104:398–411 [DOI] [PubMed] [Google Scholar]
- Guillod-Maximin E, Roy AF, Vacher CM, Aubourg A, Bailleux V, Lorsignol A, Pénicaud L, Parquet M, Taouis M (2009) Adiponectin receptors are expressed in hypothalamus and colocalized with proopiomelanocortin and neuropeptide Y in rodent arcuate neurons. J Endocrinol 200:93–105 [DOI] [PubMed] [Google Scholar]
- Hale MW, Dady KF, Evans AK, Lowry CA (2011) Evidence for in vivo thermosensitivity of serotonergic neurons in the rat dorsal raphe nucleus and raphe pallidus nucleus implicated in thermoregulatory cooling. Exp Neurol 227:264–278 [DOI] [PubMed] [Google Scholar]
- Han Y, Xia G, Srisai D, Meng F, He Y, Ran Y, He Y, Farias M, Hoang G, Tóth I et al (2021) Deciphering an AgRP-serotoninergic neural circuit in distinct control of energy metabolism from feeding. Nat Commun 12:3525 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haque MS, Minokoshi Y, Hamai M, Iwai M, Horiuchi M, Shimazu T (1999) Role of the sympathetic nervous system and insulin in enhancing glucose uptake in peripheral tissues after intrahypothalamic injection of leptin in rats. Diabetes 48:1706–1712 [DOI] [PubMed] [Google Scholar]
- Harris RB, Zhou J, Redmann SM, Smagin GN, Smith SR, Rodgers E, Zachwieja JJ (1998) A leptin dose-response study in obese (ob/ob) and lean (+/?) mice. Endocrinology 139:8–19 [DOI] [PubMed] [Google Scholar]
- Harris RBS (2018) Denervation as a tool for testing sympathetic control of white adipose tissue. Physiol Behav 190:3–10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hernández-Cáceres MP, Toledo-Valenzuela L, Díaz-Castro F, Ávalos Y, Burgos P, Narro C, Peña-Oyarzun D, Espinoza-Caicedo J, Cifuentes-Araneda F, Navarro-Aguad F et al (2019) Palmitic acid reduces the autophagic flux and insulin sensitivity through the activation of the free fatty acid receptor 1 (FFAR1) in the hypothalamic neuronal cell line N43/5. Front Endocrinol 10:176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higgins GA, Bradbury AJ, Jones BJ, Oakley NR (1988) Behavioural and biochemical consequences following activation of 5HT1-like and GABA receptors in the dorsal raphé nucleus of the rat. Neuropharmacology 27:993–1001 [DOI] [PubMed] [Google Scholar]
- Hofmann K, Lamberz C, Piotrowitz K, Offermann N, But D, Scheller A, Al-Amoudi A, Kuerschner L (2017) Tanycytes and a differential fatty acid metabolism in the hypothalamus. Glia 65:231–249 [DOI] [PubMed] [Google Scholar]
- Hogan S, Coscina DV, Himms-Hagen J (1982) Brown adipose tissue of rats with obesity-inducing ventromedial hypothalamic lesions. Am J Physiol 243:E338–344 [DOI] [PubMed] [Google Scholar]
- Hoyda TD, Fry M, Ahima RS, Ferguson AV (2007) Adiponectin selectively inhibits oxytocin neurons of the paraventricular nucleus of the hypothalamus. J Physiol 585:805–816 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoyda TD, Smith PM, Ferguson AV (2009) Adiponectin acts in the nucleus of the solitary tract to decrease blood pressure by modulating the excitability of neuropeptide Y neurons. Brain Res 1256:76–84 [DOI] [PubMed] [Google Scholar]
- Imbernon M, Beiroa D, Vázquez MJ, Morgan DA, Veyrat-Durebex C, Porteiro B, Díaz-Arteaga A, Senra A, Busquets S, Velásquez DA et al (2013) Central melanin-concentrating hormone influences liver and adipose metabolism via specific hypothalamic nuclei and efferent autonomic/JNK1 pathways. Gastroenterology 144:636–649.e6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inoue N, Matsunaga Y, Satoh H, Takahashi M (2007) Enhanced energy expenditure and fat oxidation in humans with high BMI scores by the ingestion of novel and non-pungent capsaicin analogues (capsinoids). Biosci Biotechnol Biochem 71:380–389 [DOI] [PubMed] [Google Scholar]
- Isaac R, Reis FCG, Ying W, Olefsky JM (2021) Exosomes as mediators of intercellular crosstalk in metabolism. Cell Metab 33:1744–1762 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jais A, Paeger L, Sotelo-Hitschfeld T, Bremser S, Prinzensteiner M, Klemm P, Mykytiuk V, Widdershooven PJM, Vesting AJ, Grzelka K et al (2020) PNOCARC neurons promote hyperphagia and obesity upon high-fat-diet feeding. Neuron 106:1009–1025.e10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeong JH, Lee DK, Blouet C, Ruiz HH, Buettner C, Chua S, Schwartz GJ, Jo Y-H (2015) Cholinergic neurons in the dorsomedial hypothalamus regulate mouse brown adipose tissue metabolism. Mol Metab 4:483–492 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang H, Ding X, Cao Y, Wang H, Zeng W (2017) Dense intra-adipose sympathetic arborizations are essential for cold-induced beiging of mouse white adipose tissue. Cell Metab 26:686–692.e3 [DOI] [PubMed] [Google Scholar]
- Johann K, Cremer AL, Fischer AW, Heine M, Pensado ER, Resch J, Nock S, Virtue S, Harder L, Oelkrug R et al (2019) Thyroid-hormone-induced browning of white adipose tissue does not contribute to thermogenesis and glucose consumption. Cell Rep 27:3385–3400.e3 [DOI] [PubMed] [Google Scholar]
- Kajimura D, Lee HW, Riley KJ, Arteaga-Solis E, Ferron M, Zhou B, Clarke CJ, Hannun YA, DePinho RA, Guo XE et al (2013) Adiponectin regulates bone mass via opposite central and peripheral mechanisms through FoxO1. Cell Metab 17:901–915 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalsbeek A, Fliers E, Romijn JA, La Fleur SE, Wortel J, Bakker O, Endert E, Buijs RM (2001) The suprachiasmatic nucleus generates the diurnal changes in plasma leptin levels. Endocrinology 142:2677–2685 [DOI] [PubMed] [Google Scholar]
- Kawai T, Autieri MV, Scalia R (2021) Adipose tissue inflammation and metabolic dysfunction in obesity. Am J Physiol Cell Physiol 320:C375–C391 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim JG, Suyama S, Koch M, Jin S, Argente-Arizon P, Argente J, Liu Z-W, Zimmer MR, Jeong JK, Szigeti-Buck K et al (2014) Leptin signaling in astrocytes regulates hypothalamic neuronal circuits and feeding. Nat Neurosci 17:908–910 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim KW, Zhao L, Donato J, Kohno D, Xu Y, Elias CF, Lee C, Parker KL, Elmquist JK (2011) Steroidogenic factor 1 directs programs regulating diet-induced thermogenesis and leptin action in the ventral medial hypothalamic nucleus. Proc Natl Acad Sci USA 108:10673–10678 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knight ZA, Hannan KS, Greenberg ML, Friedman JM (2010) Hyperleptinemia is required for the development of leptin resistance. PLoS ONE 5:e11376 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koch CE, Lowe C, Legler K, Benzler J, Boucsein A, Böttiger G, Grattan DR, Williams LM, Tups A (2014) Central adiponectin acutely improves glucose tolerance in male mice. Endocrinology 155:1806–1816 [DOI] [PubMed] [Google Scholar]
- Kreier F, Fliers E, Voshol PJ, Van Eden CG, Havekes LM, Kalsbeek A, Van Heijningen CL, Sluiter AA, Mettenleiter TC, Romijn JA et al (2002) Selective parasympathetic innervation of subcutaneous and intra-abdominal fat-functional implications. J Clin Investig 110:1243–1250 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kreutzer C, Peters S, Schulte DM, Fangmann D, Türk K, Wolff S, van Eimeren T, Ahrens M, Beckmann J, Schafmayer C et al (2017) Hypothalamic inflammation in human obesity is mediated by environmental and genetic factors. Diabetes 66:2407–2415 [DOI] [PubMed] [Google Scholar]
- Kubota N, Yano W, Kubota T, Yamauchi T, Itoh S, Kumagai H, Kozono H, Takamoto I, Okamoto S, Shiuchi T et al (2007) Adiponectin stimulates AMP-activated protein kinase in the hypothalamus and increases food intake. Cell Metab 6:55–68 [DOI] [PubMed] [Google Scholar]
- Lass A, Zimmermann R, Oberer M, Zechner R (2011) Lipolysis—a highly regulated multi-enzyme complex mediates the catabolism of cellular fat stores. Prog Lipid Res 50:14–27 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lavoie O, Michael NJ, Caron A (2023) A critical update on the leptin-melanocortin system. J Neurochem 165:467–486 [DOI] [PubMed] [Google Scholar]
- Le Foll C, Dunn-Meynell A, Musatov S, Magnan C, Levin BE (2013) FAT/CD36: a major regulator of neuronal fatty acid sensing and energy homeostasis in rats and mice. Diabetes 62:2709–2716 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee Y-H, Kim S-N, Kwon H-J, Maddipati KR, Granneman JG (2016) Adipogenic role of alternatively activated macrophages in β-adrenergic remodeling of white adipose tissue. Am J Physiol Regul Integr Comp Physiol 310:R55–65 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee Y-H, Petkova AP, Granneman JG (2013) Identification of an adipogenic niche for adipose tissue remodeling and restoration. Cell Metab 18:355–367 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lei Y, Liang X, Sun Y, Yao T, Gong H, Chen Z, Gao Y, Wang H, Wang R, Huang Y et al (2024) Region-specific transcriptomic responses to obesity and diabetes in macaque hypothalamus. Cell Metab 36:438–453.e6 [DOI] [PubMed] [Google Scholar]
- Li L, Ma L, Luo Z, Wei X, Zhao Y, Zhou C, Mou A, Lu Z, You M, He C et al (2022) Lack of TRPV1 aggravates obesity-associated hypertension through the disturbance of mitochondrial Ca2+ homeostasis in brown adipose tissue. Hypertens Res 45:789–801 [DOI] [PMC free article] [PubMed] [Google Scholar]
- López M, Lage R, Saha AK, Pérez-Tilve D, Vázquez MJ, Varela L, Sangiao-Alvarellos S, Tovar S, Raghay K, Rodríguez-Cuenca S et al (2008) Hypothalamic fatty acid metabolism mediates the orexigenic action of ghrelin. Cell Metab 7:389–399 [DOI] [PubMed] [Google Scholar]
- Majdic G, Young M, Gomez-Sanchez E, Anderson P, Szczepaniak LS, Dobbins RL, McGarry JD, Parker KL (2002) Knockout mice lacking steroidogenic factor 1 are a novel genetic model of hypothalamic obesity. Endocrinology 143:607–614 [DOI] [PubMed] [Google Scholar]
- Martínez de Morentin PB, González-García I, Martins L, Lage R, Fernández-Mallo D, Martínez-Sánchez N, Ruíz-Pino F, Liu J, Morgan DA, Pinilla L et al (2014) Estradiol regulates brown adipose tissue thermogenesis via hypothalamic AMPK. Cell Metab 20:41–53 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martínez-Sánchez N, Seoane-Collazo P, Contreras C, Varela L, Villarroya J, Rial-Pensado E, Buqué X, Aurrekoetxea I, Delgado TC, Vázquez-Martínez R et al (2017) Hypothalamic AMPK-ER stress-JNK1 axis mediates the central actions of thyroid hormones on energy balance. Cell Metab 26:212–229.e12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matthias A, Ohlson KB, Fredriksson JM, Jacobsson A, Nedergaard J, Cannon B (2000) Thermogenic responses in brown fat cells are fully UCP1-dependent. UCP2 or UCP3 do not substitute for UCP1 in adrenergically or fatty scid-induced thermogenesis. J Biol Chem 275:25073–25081 [DOI] [PubMed] [Google Scholar]
- Mehri K, Hamidian G, Zavvari Oskuye Z, Nayebirad S, Farajdokht F (2023) The role of apelinergic system in metabolism and reproductive system in normal and pathological conditions: an overview. Front Endocrinol 14:1193150 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meng Q, Cai D (2011) Defective hypothalamic autophagy directs the central pathogenesis of obesity via the IkappaB kinase beta (IKKbeta)/NF-kappaB pathway. J Biol Chem 286:32324–32332 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Migrenne S, Cruciani-Guglielmacci C, Kang L, Wang R, Rouch C, Lefèvre A-L, Ktorza A, Routh VH, Levin BE, Magnan C (2006) Fatty acid signaling in the hypothalamus and the neural control of insulin secretion. Diabetes 55:S139–S144 [Google Scholar]
- Miralpeix C, Reguera AC, Fosch A, Zagmutt S, Casals N, Cota D, Rodríguez-Rodríguez R (2021) Hypothalamic endocannabinoids in obesity: an old story with new challenges. Cell Mol Life Sci 78:7469–7490 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mistry AM, Swick AG, Romsos DR (1997) Leptin rapidly lowers food intake and elevates metabolic rates in lean and ob/ob mice. J Nutr 127:2065–2072 [DOI] [PubMed] [Google Scholar]
- Monge-Roffarello B, Labbe SM, Roy M-C, Lemay M-L, Coneggo E, Samson P, Lanfray D, Richard D (2014) The PVH as a site of CB1-mediated stimulation of thermogenesis by MC4R agonism in male rats. Endocrinology 155:3448–3458 [DOI] [PubMed] [Google Scholar]
- Moraes JC, Coope A, Morari J, Cintra DE, Roman EA, Pauli JR, Romanatto T, Carvalheira JB, Oliveira ALR, Saad MJ et al (2009) High-fat diet induces apoptosis of hypothalamic neurons. PLoS ONE 4:e5045 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morrison SF, Nakamura K (2019) Central mechanisms for thermoregulation. Annu Rev Physiol 81:285–308 [DOI] [PubMed] [Google Scholar]
- Moullé VS, Le Foll C, Philippe E, Kassis N, Rouch C, Marsollier N, Bui L-C, Guissard C, Dairou J, Lorsignol A et al (2013) Fatty acid transporter CD36 mediates hypothalamic effect of fatty acids on food intake in rats. PLoS ONE 8:e74021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murano I, Barbatelli G, Giordano A, Cinti S (2009) Noradrenergic parenchymal nerve fiber branching after cold acclimatisation correlates with brown adipocyte density in mouse adipose organ. J Anat 214:171–178 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakamura K, Morrison SF (2008) Preoptic mechanism for cold-defensive responses to skin cooling. J Physiol 586:2611–2620 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Néchad M, Ruka E, Thibault J (1994) Production of nerve growth factor by brown fat in culture: relation with the in vivo developmental stage of the tissue. Comp Biochem Physiol Comp Physiol 107:381–388 [DOI] [PubMed] [Google Scholar]
- Nguyen C, Lei H-T, Lai LTF, Gallenito MJ, Mu X, Matthies D, Gonen T (2023) Lipid flipping in the omega-3 fatty-acid transporter. Nat Commun 14:2571 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nguyen LN, Ma D, Shui G, Wong P, Cazenave-Gassiot A, Zhang X, Wenk MR, Goh ELK, Silver DL (2014a) Mfsd2a is a transporter for the essential omega-3 fatty acid docosahexaenoic acid. Nature 509:503–506 [DOI] [PubMed] [Google Scholar]
- Nguyen NLT, Randall J, Banfield BW, Bartness TJ (2014b) Central sympathetic innervations to visceral and subcutaneous white adipose tissue. Am J Physiol Regul Integr Comp Physiol 306:R375–386 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nijenhuis WA, Oosterom J, Adan RA (2001) AgRP(83-132) acts as an inverse agonist on the human-melanocortin-4 receptor. Mol Endocrinol 15:164–171 [DOI] [PubMed] [Google Scholar]
- Nisoli E, Tonello C, Benarese M, Liberini P, Carruba MO (1996) Expression of nerve growth factor in brown adipose tissue: implications for thermogenesis and obesity. Endocrinology 137:495–503 [DOI] [PubMed] [Google Scholar]
- Obici S, Feng Z, Arduini A, Conti R, Rossetti L (2003) Inhibition of hypothalamic carnitine palmitoyltransferase-1 decreases food intake and glucose production. Nat Med 9:756–761 [DOI] [PubMed] [Google Scholar]
- Obici S, Feng Z, Morgan K, Stein D, Karkanias G, Rossetti L (2002) Central administration of oleic acid inhibits glucose production and food intake. Diabetes 51:271–275 [DOI] [PubMed] [Google Scholar]
- Ohyama K, Nogusa Y, Shinoda K, Suzuki K, Bannai M, Kajimura S (2016) A synergistic antiobesity effect by a combination of capsinoids and cold temperature through promoting beige adipocyte biogenesis. Diabetes 65:1410–1423 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orava J, Nuutila P, Lidell ME, Oikonen V, Noponen T, Viljanen T, Scheinin M, Taittonen M, Niemi T, Enerbäck S et al (2011) Different metabolic responses of human brown adipose tissue to activation by cold and insulin. Cell Metab 14:272–279 [DOI] [PubMed] [Google Scholar]
- Ottaway N, Mahbod P, Rivero B, Norman LA, Gertler A, D’Alessio DA, Perez-Tilve D (2015) Diet-induced obese mice retain endogenous leptin action. Cell Metab 21:877–882 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ouellet V, Labbé SM, Blondin DP, Phoenix S, Guérin B, Haman F, Turcotte EE, Richard D, Carpentier AC (2012) Brown adipose tissue oxidative metabolism contributes to energy expenditure during acute cold exposure in humans. J Clin Invest 122:545–552 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozcan L, Ergin AS, Lu A, Chung J, Sarkar S, Nie D, Myers MG, Ozcan U (2009) Endoplasmic reticulum stress plays a central role in development of leptin resistance. Cell Metab 9:35–51 [DOI] [PubMed] [Google Scholar]
- Pellegrinelli V, Peirce VJ, Howard L, Virtue S, Türei D, Senzacqua M, Frontini A, Dalley JW, Horton AR, Bidault G et al (2018) Adipocyte-secreted BMP8b mediates adrenergic-induced remodeling of the neuro-vascular network in adipose tissue. Nat Commun 9:4974 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelleymounter MA, Cullen MJ, Baker MB, Hecht R, Winters D, Boone T, Collins F (1995) Effects of the obese gene product on body weight regulation in ob/ob mice. Science 269:540–543 [DOI] [PubMed] [Google Scholar]
- Petkevicius K, Bidault G, Virtue S, Newland SA, Dale M, Dugourd A, Saez-Rodriguez J, Mallat Z, Vidal-Puig A (2021) Macrophage beta2-adrenergic receptor is dispensable for the adipose tissue inflammation and function. Mol Metab 48:101220 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pifferi F, Laurent B, Plourde M (2021) Lipid transport and metabolism at the blood-brain interface: implications in health and disease. Front Physiol 12:645646 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Preston E, Triandafillou J, Haas N (1989) Colchicine lesions of ventromedial hypothalamus: effects on regulatory thermogenesis in the rat. Pharmacol Biochem Behav 32:301–307 [DOI] [PubMed] [Google Scholar]
- Qi Y, Takahashi N, Hileman SM, Patel HR, Berg AH, Pajvani UB, Scherer PE, Ahima RS (2004) Adiponectin acts in the brain to decrease body weight. Nat Med 10:524–529 [DOI] [PubMed] [Google Scholar]
- Qiang L, Wang L, Kon N, Zhao W, Lee S, Zhang Y, Rosenbaum M, Zhao Y, Gu W, Farmer SR et al (2012) Brown remodeling of white adipose tissue by SirT1-dependent deacetylation of Pparγ. Cell 150:620–632 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quan W, Kim H-K, Moon E-Y, Kim SS, Choi CS, Komatsu M, Jeong YT, Lee M-K, Kim K-W, Kim M-S et al (2012) Role of hypothalamic proopiomelanocortin neuron autophagy in the control of appetite and leptin response. Endocrinology 153:1817–1826 [DOI] [PubMed] [Google Scholar]
- Quiñones M, Hernández-Bautista R, Beiroa D, Heras V, Torres-Leal FL, Lam BYH, Senra A, Fernø J, Gómez-Valadés AG, Schwaninger M et al (2021) Sirt3 in POMC neurons controls energy balance in a sex- and diet-dependent manner. Redox Biol 41:101945 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rahman MS, Jun H (2022) The adipose tissue macrophages central to adaptive thermoregulation. Front Immunol 13:884126 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rapoport SI, Chang MC, Spector AA (2001) Delivery and turnover of plasma-derived essential PUFAs in mammalian brain. J Lipid Res 42:678–685 [PubMed] [Google Scholar]
- Rashid M, Kondoh K, Palfalvi G, Nakajima K-I, Minokoshi Y (2023) Inhibition of high-fat diet-induced inflammatory responses in adipose tissue by SF1-expressing neurons of the ventromedial hypothalamus. Cell Rep 42:112627 [DOI] [PubMed] [Google Scholar]
- Reaux-Le Goazigo A, Bodineau L, De Mota N, Jeandel L, Chartrel N, Knauf C, Raad C, Valet P, Llorens-Cortes C (2011) Apelin and the proopiomelanocortin system: a new regulatory pathway of hypothalamic α-MSH release. Am J Physiol Endocrinol Metab 301:E955–966 [DOI] [PubMed] [Google Scholar]
- Ribeiro MO, Bianco SDC, Kaneshige M, Schultz JJ, Cheng S, Bianco AC, Brent GA (2010) Expression of uncoupling protein 1 in mouse brown adipose tissue is thyroid hormone receptor-beta isoform specific and required for adaptive thermogenesis. Endocrinology 151:432–440 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez-Navas C, Morselli E, Clegg DJ (2016) Sexually dimorphic brain fatty acid composition in low and high fat diet-fed mice. Mol Metab 5:680 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodríguez-Rodríguez R, Miralpeix C, Fosch A, Pozo M, Calderón-Domínguez M, Perpinyà X, Vellvehí M, López M, Herrero L, Serra D et al (2019) CPT1C in the ventromedial nucleus of the hypothalamus is necessary for brown fat thermogenesis activation in obesity. Mol Metab 19:75–85 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosell M, Kaforou M, Frontini A, Okolo A, Chan Y-W, Nikolopoulou E, Millership S, Fenech ME, MacIntyre D, Turner JO et al (2014) Brown and white adipose tissues: intrinsic differences in gene expression and response to cold exposure in mice. Am J Physiol Endocrinol Metab 306:E945–E964 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruschke K, Ebelt H, Klöting N, Boettger T, Raum K, Blüher M, Braun T (2009) Defective peripheral nerve development is linked to abnormal architecture and metabolic activity of adipose tissue in Nscl-2 mutant mice. PLoS ONE 4:e5516 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryu V, Garretson JT, Liu Y, Vaughan CH, Bartness TJ (2015) Brown adipose tissue has sympathetic-sensory feedback circuits. J Neurosci 35:2181–2190 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sakers A, De Siqueira MK, Seale P, Villanueva CJ (2022) Adipose-tissue plasticity in health and disease. Cell 185:419–446 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santos GSP, Costa AC, Picoli CC, Rocha BGS, Sulaiman SO, Radicchi DC, Pinto MCX, Batista ML, Amorim JH, Azevedo VAC et al (2022) Sympathetic nerve-adipocyte interactions in response to acute stress. J Mol Med 100:151–165 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Satoh N, Ogawa Y, Katsuura G, Numata Y, Tsuji T, Hayase M, Ebihara K, Masuzaki H, Hosoda K, Yoshimasa Y et al (1999) Sympathetic activation of leptin via the ventromedial hypothalamus: leptin-induced increase in catecholamine secretion. Diabetes 48:1787–1793 [DOI] [PubMed] [Google Scholar]
- Scarpace PJ, Matheny M (1998) Leptin induction of UCP1 gene expression is dependent on sympathetic innervation. Am J Physiol 275:E259–264 [DOI] [PubMed] [Google Scholar]
- Scherer PE, Williams S, Fogliano M, Baldini G, Lodish HF (1995) A novel serum protein similar to C1q, produced exclusively in adipocytes. J Biol Chem 270:26746–26749 [DOI] [PubMed] [Google Scholar]
- Scherer T, O’Hare J, Diggs-Andrews K, Schweiger M, Cheng B, Lindtner C, Zielinski E, Vempati P, Su K, Dighe S et al (2011) Brain insulin controls adipose tissue lipolysis and lipogenesis. Cell Metab 13:183–194 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schirinzi V, Poli C, Berteotti C, Leone A (2023) Browning of adipocytes: a potential therapeutic approach to obesity. Nutrients 15:2229 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneeberger M, Dietrich MO, Sebastián D, Imbernón M, Castaño C, Garcia A, Esteban Y, Gonzalez-Franquesa A, Rodríguez IC, Bortolozzi A et al (2013) Mitofusin 2 in POMC neurons connects ER stress with leptin resistance and energy imbalance. Cell 155:172–187 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneeberger M, Parolari L, Das Banerjee T, Bhave V, Wang P, Patel B, Topilko T, Wu Z, Choi CHJ, Yu X et al (2019) Regulation of energy expenditure by brainstem GABA neurons. Cell 178:672–685.e12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seale P, Conroe HM, Estall J, Kajimura S, Frontini A, Ishibashi J, Cohen P, Cinti S, Spiegelman BM (2011) Prdm16 determines the thermogenic program of subcutaneous white adipose tissue in mice. J Clin Investig 121:96–105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seale P, Kajimura S, Yang W, Chin S, Rohas LM, Uldry M, Tavernier G, Langin D, Spiegelman BM (2007) Transcriptional control of brown fat determination by PRDM16. Cell Metab 6:38–54 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seoane-Collazo P, Roa J, Rial-Pensado E, Liñares-Pose L, Beiroa D, Ruíz-Pino F, López-González T, Morgan DA, Pardavila JÁ, Sánchez-Tapia MJ et al (2018) SF1-specific AMPKα1 deletion protects against diet-induced obesity. Diabetes 67:2213–2226 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snitker S, Fujishima Y, Shen H, Ott S, Pi-Sunyer X, Furuhata Y, Sato H, Takahashi M (2009) Effects of novel capsinoid treatment on fatness and energy metabolism in humans: possible pharmacogenetic implications. Am J Clin Nutr 89:45–50 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song CK, Schwartz GJ, Bartness TJ (2009) Anterograde transneuronal viral tract tracing reveals central sensory circuits from white adipose tissue. Am J Physiol Regul Integr Comp Physiol 296:R501–511 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song CK, Vaughan CH, Keen-Rhinehart E, Harris RBS, Richard D, Bartness TJ (2008) Melanocortin-4 receptor mRNA expressed in sympathetic outflow neurons to brown adipose tissue: neuroanatomical and functional evidence. Am J Physiol Regul Integr Comp Physiol 295:R417–428 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song N, Jeong DY, Tu TH, Park BS, Yang HR, Kim YJ, Kim JK, Park JT, Yeh J-Y, Yang S et al (2021) Adiponectin Controls Nutrient Availability in Hypothalamic Astrocytes. Int J Mol Sci 22:1587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steculorum SM, Ruud J, Karakasilioti I, Backes H, Engström Ruud L, Timper K, Hess ME, Tsaousidou E, Mauer J, Vogt MC et al (2016) AgRP neurons control systemic insulin sensitivity via myostatin expression in brown adipose tissue. Cell 165:125–138 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steppan CM, Bailey ST, Bhat S, Brown EJ, Banerjee RR, Wright CM, Patel HR, Ahima RS, Lazar MA (2001) The hormone resistin links obesity to diabetes. Nature 409:307–312 [DOI] [PubMed] [Google Scholar]
- Suyama S, Lei W, Kubota N, Kadowaki T, Yada T (2017) Adiponectin at physiological level glucose-independently enhances inhibitory postsynaptic current onto NPY neurons in the hypothalamic arcuate nucleus. Neuropeptides 65:1–9 [DOI] [PubMed] [Google Scholar]
- Suyama S, Maekawa F, Maejima Y, Kubota N, Kadowaki T, Yada T (2016) Glucose level determines excitatory or inhibitory effects of adiponectin on arcuate POMC neuron activity and feeding. Sci Rep 6:30796 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thaler JP, Yi C-X, Schur EA, Guyenet SJ, Hwang BH, Dietrich MO, Zhao X, Sarruf DA, Izgur V, Maravilla KR et al (2012) Obesity is associated with hypothalamic injury in rodents and humans. J Clin Investig 122:153–162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tokizane K, Brace CS, Imai S-I (2024) DMHPpp1r17 neurons regulate aging and lifespan in mice through hypothalamic-adipose inter-tissue communication. Cell Metab 36:377–392.e11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsang AH, Koch CE, Kiehn J-T, Schmidt CX, Oster H (2020) An adipokine feedback regulating diurnal food intake rhythms in mice. eLife 9:e55388 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uno T, Shibata M (2001) Role of inferior olive and thoracic IML neurons in nonshivering thermogenesis in rats. Am J Physiol Regul Integr Comp Physiol 280:R536–546 [DOI] [PubMed] [Google Scholar]
- Valencia-Montoya WA, Pierce NE, Bellono NW (2024) Evolution of sensory receptors. Annu Rev Cell Dev Biol 40:353–379 [DOI] [PMC free article] [PubMed] [Google Scholar]
- van den Top M, Lee K, Whyment AD, Blanks AM, Spanswick D (2004) Orexigen-sensitive NPY/AgRP pacemaker neurons in the hypothalamic arcuate nucleus. Nat Neurosci 7:493–494 [DOI] [PubMed] [Google Scholar]
- van Marken Lichtenbelt WD, Vanhommerig JW, Smulders NM, Drossaerts JMAFL, Kemerink GJ, Bouvy ND, Schrauwen P, Teule GJJ (2009) Cold-activated brown adipose tissue in healthy men. N Engl J Med 360:1500–1508 [DOI] [PubMed] [Google Scholar]
- Vaughan CH, Bartness TJ (2012) Anterograde transneuronal viral tract tracing reveals central sensory circuits from brown fat and sensory denervation alters its thermogenic responses. Am J Physiol Regul Integr Comp Physiol 302:R1049–1058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaughan CH, Zarebidaki E, Ehlen JC, Bartness TJ (2014) Analysis and measurement of the sympathetic and sensory innervation of white and brown adipose tissue. Methods Enzymol 537:199–225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verty AN, Evetts MJ, Crouch GJ, McGregor IS, Stefanidis A, Oldfield BJ (2011) The cannabinoid receptor agonist THC attenuates weight loss in a rodent model of activity-based anorexia. Neuropsychopharmacol 36:1349–1358 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verty ANA, Allen AM, Oldfield BJ (2009) The effects of rimonabant on brown adipose tissue in rat: implications for energy expenditure. Obesity 17:254–261 [DOI] [PubMed] [Google Scholar]
- Virtanen KA, Lidell ME, Orava J, Heglind M, Westergren R, Niemi T, Taittonen M, Laine J, Savisto N-J, Enerbäck S et al (2009) Functional brown adipose tissue in healthy adults. New Engl J Med 360:1518–1525 [DOI] [PubMed] [Google Scholar]
- Vong L, Ye C, Yang Z, Choi B, Chua S, Lowell BB (2011) Leptin action on GABAergic neurons prevents obesity and reduces inhibitory tone to POMC neurons. Neuron 71:142–154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang D, He X, Zhao Z, Feng Q, Lin R, Sun Y, Ding T, Xu F, Luo M, Zhan C (2015) Whole-brain mapping of the direct inputs and axonal projections of POMC and AgRP neurons. Front Neuroanat 9:40 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang G-X, Zhao X-Y, Meng Z-X, Kern M, Dietrich A, Chen Z, Cozacov Z, Zhou D, Okunade AL, Su X et al (2014) The brown fat-enriched secreted factor Nrg4 preserves metabolic homeostasis through attenuation of hepatic lipogenesis. Nat Med 20:1436–1443 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J, Li L, Zhang Z, Zhang X, Zhu Y, Zhang C, Bi Y (2022a) Extracellular vesicles mediate the communication of adipose tissue with brain and promote cognitive impairment associated with insulin resistance. Cell Metab 34:1264–1279.e8 [DOI] [PubMed] [Google Scholar]
- Wang P, Loh KH, Wu M, Morgan DA, Schneeberger M, Yu X, Chi J, Kosse C, Kim D, Rahmouni K et al (2020a) A leptin-BDNF pathway regulating sympathetic innervation of adipose tissue. Nature 583:839–844 [DOI] [PubMed] [Google Scholar]
- Wang P, Loh KH, Wu M, Morgan DA, Schneeberger M, Yu X, Chi J, Kosse C, Kim D, Rahmouni K et al (2020b) A leptin-BDNF pathway regulating sympathetic innervation of adipose tissue. Nature 583:839–844 [DOI] [PubMed] [Google Scholar]
- Wang Y, Leung VH, Zhang Y, Nudell VS, Loud M, Servin-Vences MR, Yang D, Wang K, Moya-Garzon MD, Li VL et al (2022b) The role of somatosensory innervation of adipose tissues. Nature 609:569–574 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y-N, Tang Y, He Z, Ma H, Wang L, Liu Y, Yang Q, Pan D, Zhu C, Qian S et al (2021) Slit3 secreted from M2-like macrophages increases sympathetic activity and thermogenesis in adipose tissue. Nat Metab 3:1536–1551 [DOI] [PubMed] [Google Scholar]
- Weisberg SP, McCann D, Desai M, Rosenbaum M, Leibel RL, Ferrante AW (2003) Obesity is associated with macrophage accumulation in adipose tissue. J Clin Investig 112:1796–1808 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss BL, Aghajanian GK (1971) Activation of brain serotonin metabolism by heat: Role of midbrain raphe neurons. Brain Res 26:37–48 [Google Scholar]
- Whittle AJ, Carobbio S, Martins L, Slawik M, Hondares E, Vázquez MJ, Morgan D, Csikasz RI, Gallego R, Rodriguez-Cuenca S et al (2012) BMP8B increases brown adipose tissue thermogenesis through both central and peripheral actions. Cell 149:871–885 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams KW, Liu T, Kong X, Fukuda M, Deng Y, Berglund ED, Deng Z, Gao Y, Liu T, Sohn J-W et al (2014) Xbp1s in Pomc neurons connects ER stress with energy balance and glucose homeostasis. Cell Metab 20:471–482 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willows JW, Blaszkiewicz M, Lamore A, Borer S, Dubois AL, Garner E, Breeding WP, Tilbury KB, Khalil A, Townsend KL (2021) Visualization and analysis of whole depot adipose tissue neural innervation. iScience 24:103127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willows JW, Blaszkiewicz M, Townsend KL (2023a) The sympathetic innervation of adipose tissues: regulation, functions, and plasticity. Compr Physiol 13:4985–5021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willows JW, Gunsch G, Paradie E, Blaszkiewicz M, Tonniges JR, Pino MF, Smith SR, Sparks LM, Townsend KL (2023b) Schwann cells contribute to demyelinating diabetic neuropathy and nerve terminal structures in white adipose tissue. iScience 26:106189 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yi C-X, Walter M, Gao Y, Pitra S, Legutko B, Kälin S, Layritz C, García-Cáceres C, Bielohuby M, Bidlingmaier M et al (2017) TNFα drives mitochondrial stress in POMC neurons in obesity. Nat Commun 8:15143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoneshiro T, Aita S, Matsushita M, Okamatsu-Ogura Y, Kameya T, Kawai Y, Miyagawa M, Tsujisaki M, Saito M (2011) Age-related decrease in cold-activated brown adipose tissue and accumulation of body fat in healthy humans. Obesity 19:1755–1760 [DOI] [PubMed] [Google Scholar]
- Yoshida M, Satoh A, Lin JB, Mills KF, Sasaki Y, Rensing N, Wong M, Apte RS, Imai S-I (2019) Extracellular vesicle-contained eNAMPT delays aging and extends lifespan in mice. Cell Metab 30:329–342.e5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshioka M, Doucet E, Drapeau V, Dionne I, Tremblay A (2001) Combined effects of red pepper and caffeine consumption on 24 h energy balance in subjects given free access to foods. Br J Nutr 85:203–211 [DOI] [PubMed] [Google Scholar]
- Yu H, Chhabra KH, Thompson Z, Jones GL, Kiran S, Shangguan G, Low MJ (2020) Hypothalamic POMC deficiency increases circulating adiponectin despite obesity. Mol Metab 35:100957 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yue JTY, Abraham MA, LaPierre MP, Mighiu PI, Light PE, Filippi BM, Lam TKT (2015) A fatty acid-dependent hypothalamic-DVC neurocircuitry that regulates hepatic secretion of triglyceride-rich lipoproteins. Nat Commun 6:5970 [DOI] [PubMed] [Google Scholar]
- Zeng W, Pirzgalska RM, Pereira MMA, Kubasova N, Barateiro A, Seixas E, Lu Y-H, Kozlova A, Voss H, Martins GG et al (2015) Sympathetic neuro-adipose connections mediate leptin-driven lipolysis. Cell 163:84–94 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang F, Hao G, Shao M, Nham K, An Y, Wang Q, Zhu Y, Kusminski CM, Hassan G, Gupta RK et al (2018) An adipose tissue atlas: an image-guided identification of human-like BAT and beige depots in rodents. Cell Metab 27:252–262.e3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X, Zhang G, Zhang H, Karin M, Bai H, Cai D (2008) Hypothalamic IKKbeta/NF-kappaB and ER stress link overnutrition to energy imbalance and obesity. Cell 135:61–73 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y, Proenca R, Maffei M, Barone M, Leopold L, Friedman JM (1994) Positional cloning of the mouse obese gene and its human homologue. Nature 372:425–432 [DOI] [PubMed] [Google Scholar]
- Zheng JQ, Seki M, Hayakawa T, Ito H, Zyo K (1995) Descending projections from the paraventricular hypothalamic nucleus to the spinal cord: anterograde tracing study in the rat. Okajimas Folia Anat Jpn 72:119–135 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.