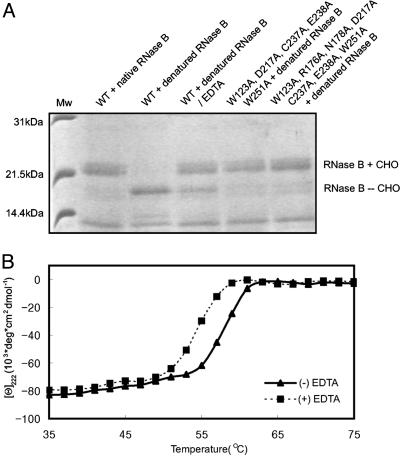
Fig. 3.
Effects of the metal chelating agent EDTA on the deglycosylation activity and stability of yPNGase. (A) Deglycosylation activities of the wild-type (WT) and mutant yPNGase proteins. Lane 1, standard molecular mass; lane 2, WT yPNGase and native RNase B; lane 3, WT yPNGase and the denatured RNase B; lane 4, WT yPNGase and the denatured RNase B in the presence of EDTA; lane 5, five residues were mutated simultaneous in yPNGasemis5; lane 6, seven residues were mutated simultaneous in yPNGasemis7. RNaseB + CHO and RNaseB - CHO represent glycosylated and deglycosylated RNase B, respectively. (B) Thermal melting curves of yPNGase without and with EDTA are determined by circular dichroism.