Abstract
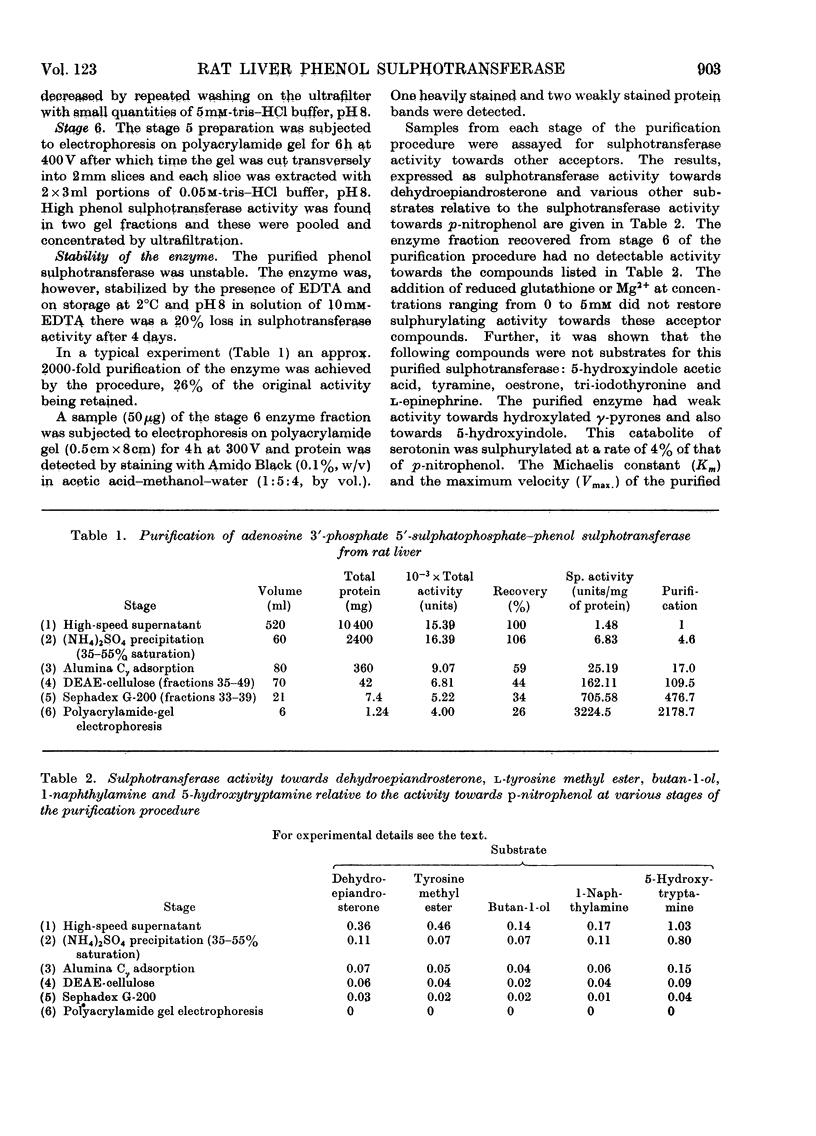
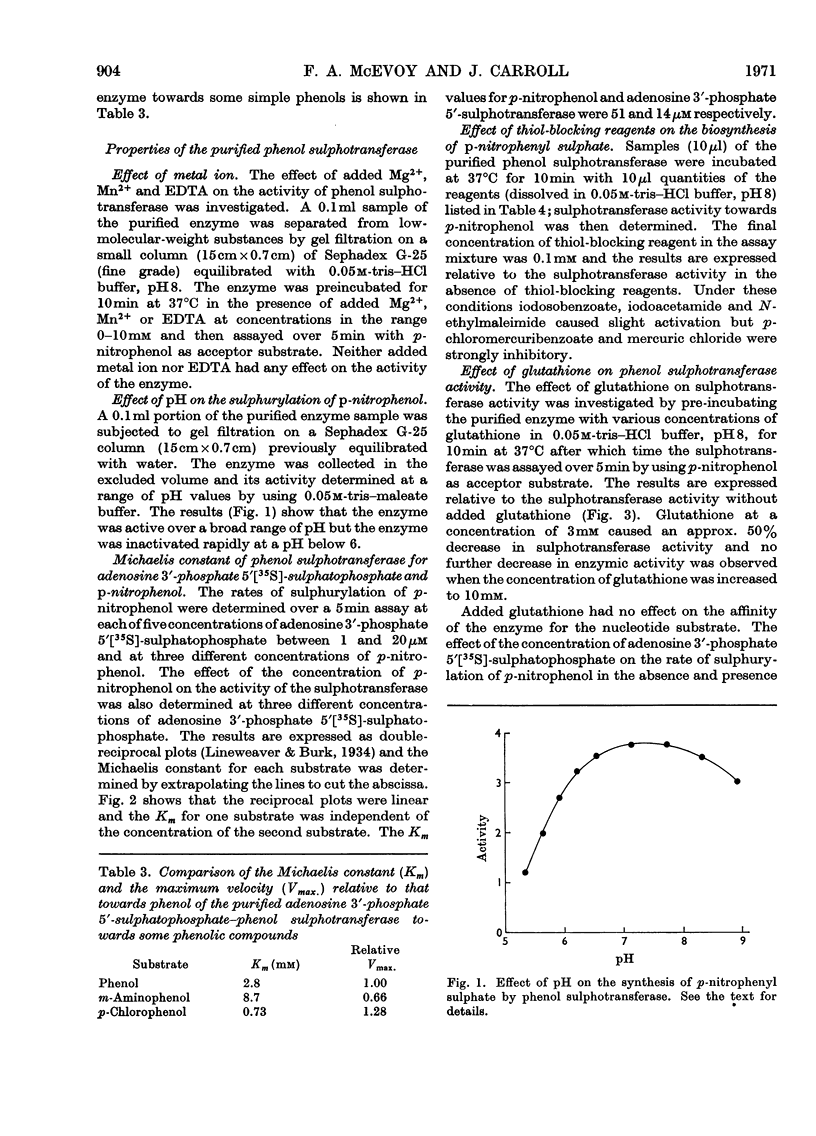
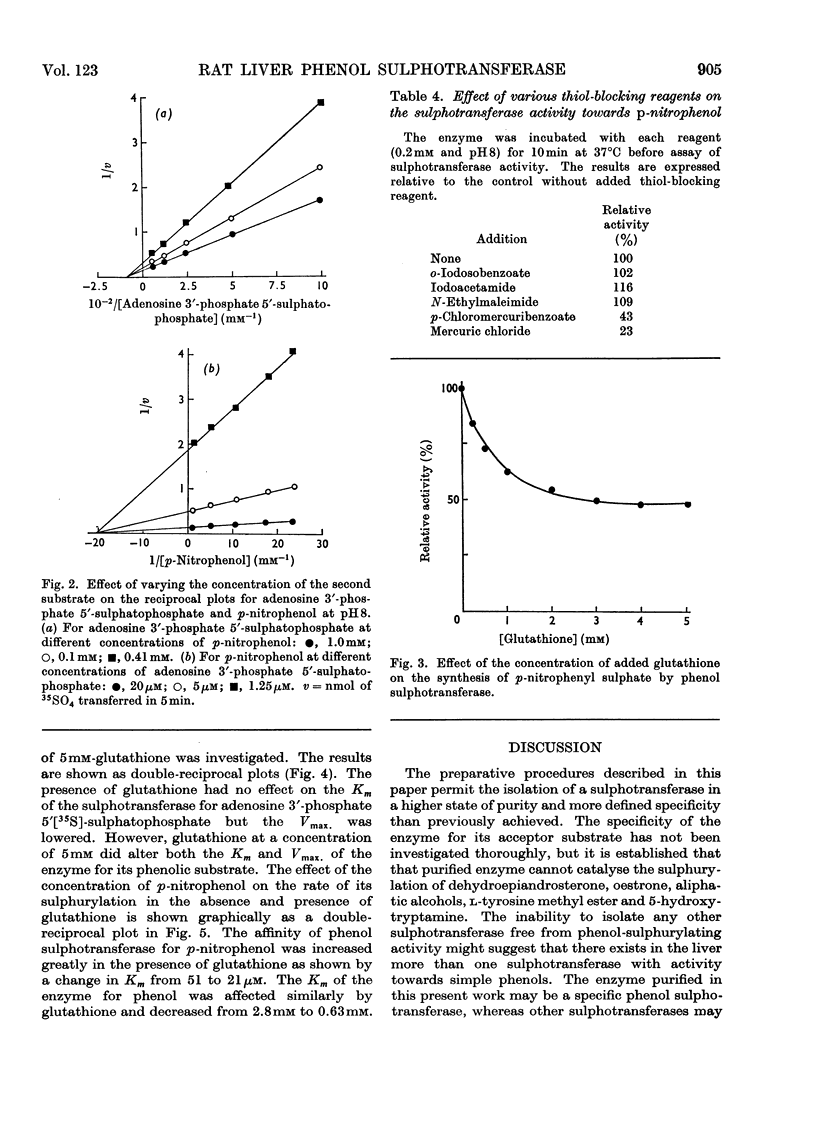
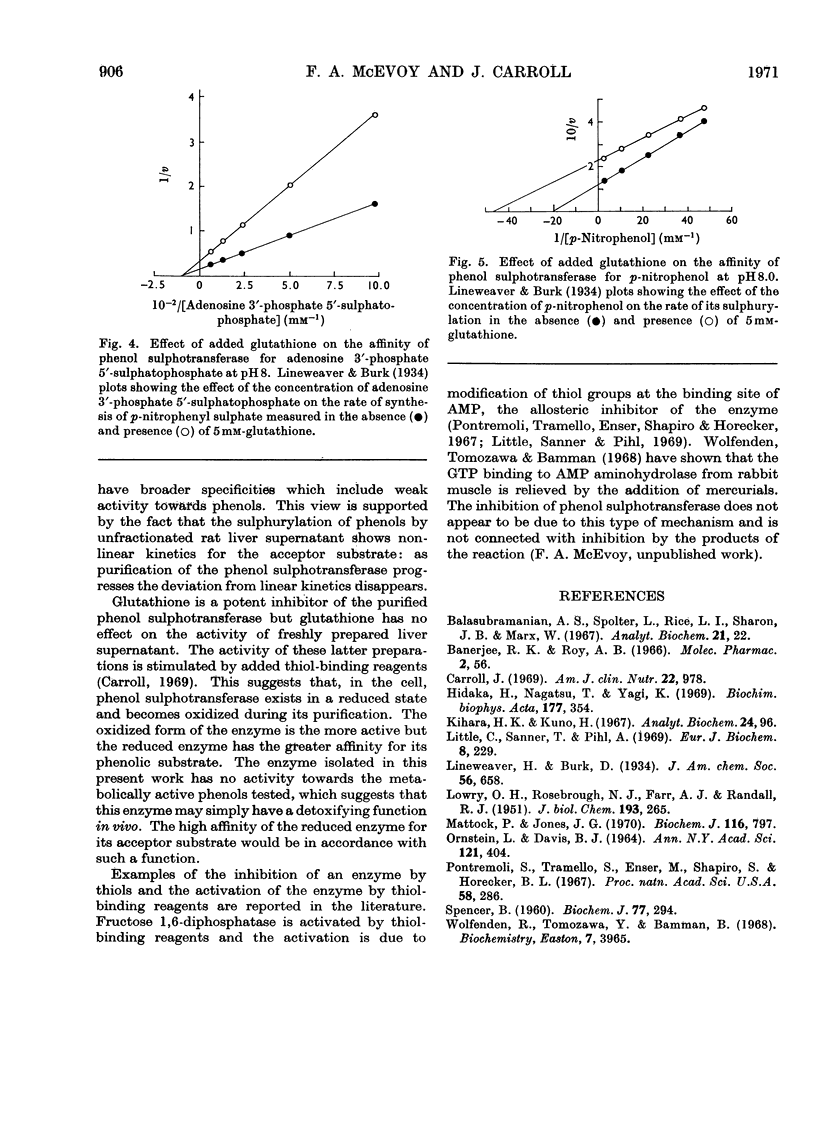
1. An enzyme (EC 2.8.2.1) that catalyses the transfer of sulphate from adenosine 3′-phosphate 5′-sulphatophosphate to phenols was purified approx. 2000-fold from male rat livers. 2. The purified preparation did not catalyse the sulphurylation of dehydroepiandrosterone, butan-1-ol, l-tyrosine methyl ester, 1-naphthylamine or serotonin. 3. At pH8.0 and 37°C the Km values of the enzyme for p-nitrophenol and adenosine 3′-phosphate 5′-sulphatophosphate are 51 and 14μm respectively. The Km value for either substrate is independent of the concentration of the other. 4. The sulphurylation of phenol is inhibited by thiol compounds and glutathione at a concentration of 3mm caused an approx. 50% decrease in enzyme activity. 5. The Km of the enzyme for adenosine 3′-phosphate 5′-sulphatophosphate is unaffected by the presence of added glutathione but at a concentration of 5mm-glutathione the Km of the enzyme for its phenolic substrate is decreased.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Balasubramanian A. S., Spolter L., Rice L. I., Sharon J. B., Marx W. Preparation of 3'-phosphoadenylyl sulfate in substrate quantities using mastocytoma enzymes. Anal Biochem. 1967 Oct;21(1):22–33. doi: 10.1016/0003-2697(67)90078-4. [DOI] [PubMed] [Google Scholar]
- Banerjee R. K., Roy A. B. The sulfotransferases of guinea pig liver. Mol Pharmacol. 1966 Jan;2(1):56–66. [PubMed] [Google Scholar]
- Carroll J. Phenolsulfotransferase in the developing rat. Am J Clin Nutr. 1969 Aug;22(8):978–985. doi: 10.1093/ajcn/22.8.978. [DOI] [PubMed] [Google Scholar]
- DAVIS B. J. DISC ELECTROPHORESIS. II. METHOD AND APPLICATION TO HUMAN SERUM PROTEINS. Ann N Y Acad Sci. 1964 Dec 28;121:404–427. doi: 10.1111/j.1749-6632.1964.tb14213.x. [DOI] [PubMed] [Google Scholar]
- Hidaka H., Nagatsu T., Yagi K. Formation of serotonin O-sulfate by sulfotransferase of rabbit liver. Biochim Biophys Acta. 1969 Apr 1;177(2):354–357. doi: 10.1016/0304-4165(69)90149-4. [DOI] [PubMed] [Google Scholar]
- Kihara H. K., Kuno H. Microassay of protein with nitrocellulose membrane filters. Anal Biochem. 1968 Jul;24(1):96–105. doi: 10.1016/0003-2697(68)90063-8. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Little C., Sanner T., Pihl A. Stimulation of fructose 1,6-diphosphatase by sulfhydryl reagents and interaction between the different enzymic sites in the molecule. Eur J Biochem. 1969 Mar;8(2):229–236. doi: 10.1111/j.1432-1033.1969.tb00519.x. [DOI] [PubMed] [Google Scholar]
- Mattock P., Jones J. G. Partial purification and properties of an enzyme from rat liver that catalyses the sulphation of L-tyrosyl derivatives. Biochem J. 1970 Mar;116(5):797–803. doi: 10.1042/bj1160797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pontremoli S., Traniello S., Enser M., Shapiro S., Horecker B. L. Regulation of fructose diphosphatase activity by disulfide exchange. Proc Natl Acad Sci U S A. 1967 Jul;58(1):286–293. doi: 10.1073/pnas.58.1.286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spencer B. Endogenous sulphate acceptors in rat liver. Biochem J. 1960 Nov;77(2):294–304. doi: 10.1042/bj0770294. [DOI] [PMC free article] [PubMed] [Google Scholar]


