Abstract
1. Intracellular recording techniques were used to study synaptic potentials in CA3 pyramidal cells elicited with mossy fibre stimulation in partially disinhibited hippocampal slice cultures. Two experimental protocols were used: (1) high concentrations (20-40 microM) of the A-type gamma-aminobutyric acid (GABAA) receptor antagonist bicuculline plus low concentrations (2-4 microM) of the glutamate receptor antagonist 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX), or (2) low concentrations (1-2.5 microM) of bicuculline alone. 2. Under the first condition, stimulation of mossy fibre afferents evoked epileptic bursts alternating with a response consisting of an excitatory postsynaptic potential (EPSP) followed by an unusually large and long-lasting hyperpolarizing potential with a maximal amplitude in the range of -30 mV from the resting membrane potential. 3. This paroxysmal inhibitory potential (PIP) had a reversal potential near that of potassium. The amplitude of the PIP was not dependent on action potentials superimposed on the preceding EPSP, and was present in cells recorded with microelectrodes containing the Ca2+ chelator EGTA. These data suggest that the PIP is not a Ca(2+)-activated K+ potential. 4. The PIP was prolonged by the GABA-uptake blocker nipecotic acid, was reduced by hyperpolarizing interneurons with the opioid agonist FK 33-824, and was abolished by the GABAB-receptor antagonist CGP 35 348. These data indicate that the PIP is mediated by the activation of GABAB receptors following GABA release from interneurons. 5. The NMDA-receptor antagonist D-2-amino-5-phosphonovalerate (D-APV) strongly reduced the amplitude of the PIP, but had no effect on the GABAB receptor-mediated inhibitory postsynaptic potential (IPSP) under control conditions. 6. Under the first condition, regular stimulation elicited a cyclical pattern of evoked responses. There was either an alternation between an epileptic burst and a PIP or, at shorter interstimulus intervals, a sequence of gradually increasing PIPs followed by an epileptic burst, which then reset the cycle. 7. Under the second condition, in low concentrations of bicuculline alone, the early GABAA-mediated IPSP was little affected, but the late GABAB-mediated IPSP was greatly enhanced. These enhanced late IPSPs were comparable in amplitude and duration to the PIPs seen under the first conditions, could exhibit cyclical behaviour, and were reduced by D-APV. 8. Application of CGP 35 348 abolished the late IPSP under control conditions, but had no effect on hippocampal excitability. In contrast, CGP 35 348 blocked the PIP elicited in low bicuculline, and consequently led to intense epileptic discharge.(ABSTRACT TRUNCATED AT 400 WORDS)
Full text
PDF




















Selected References
These references are in PubMed. This may not be the complete list of references from this article.
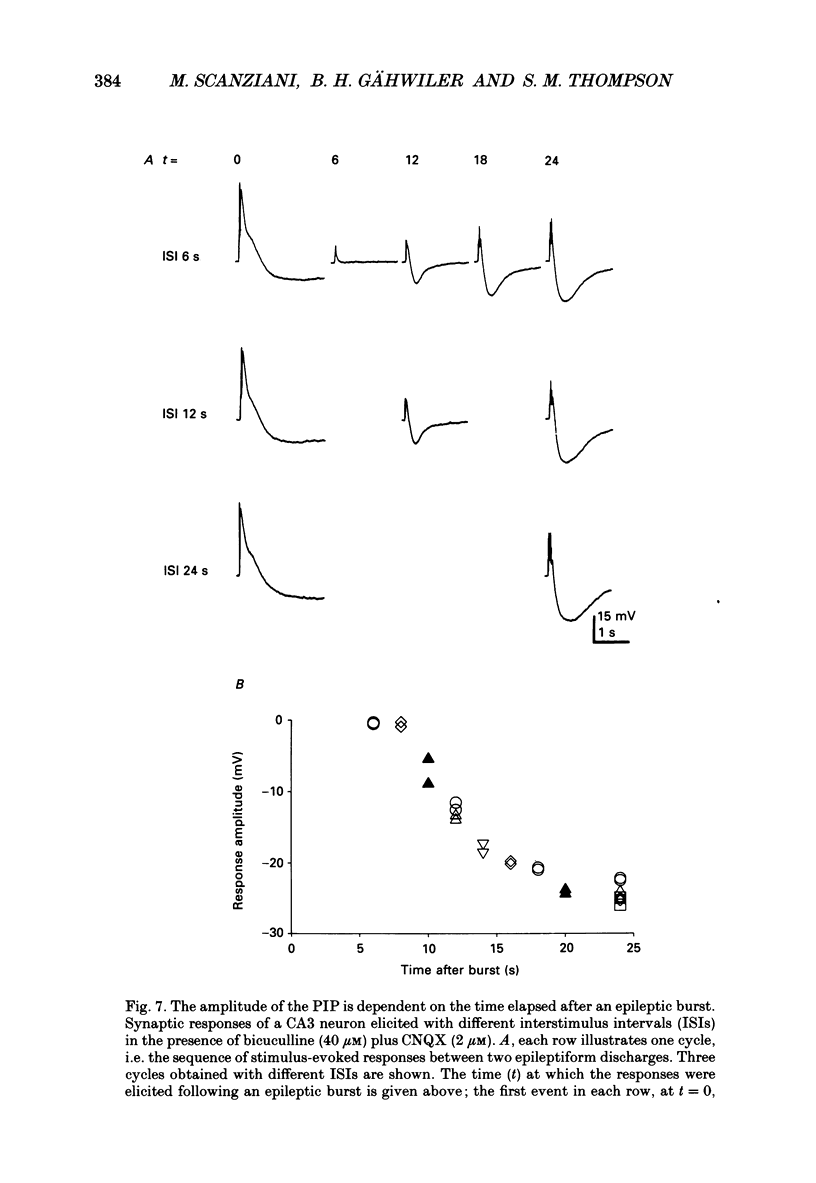
- ANDERSEN P., ECCLES J. C., LOYNING Y. PATHWAY OF POSTSYNAPTIC INHIBITION IN THE HIPPOCAMPUS. J Neurophysiol. 1964 Jul;27:608–619. doi: 10.1152/jn.1964.27.4.608. [DOI] [PubMed] [Google Scholar]
- Andreasen M., Lambert J. D., Jensen M. S. Effects of new non-N-methyl-D-aspartate antagonists on synaptic transmission in the in vitro rat hippocampus. J Physiol. 1989 Jul;414:317–336. doi: 10.1113/jphysiol.1989.sp017690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avoli M. Epileptiform discharges and a synchronous GABAergic potential induced by 4-aminopyridine in the rat immature hippocampus. Neurosci Lett. 1990 Sep 4;117(1-2):93–98. doi: 10.1016/0304-3940(90)90125-s. [DOI] [PubMed] [Google Scholar]
- Babb T. L., Pretorius J. K., Kupfer W. R., Crandall P. H. Glutamate decarboxylase-immunoreactive neurons are preserved in human epileptic hippocampus. J Neurosci. 1989 Jul;9(7):2562–2574. doi: 10.1523/JNEUROSCI.09-07-02562.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bingmann D., Speckmann E. J. Actions of pentylenetetrazol (PTZ) on CA3 neurons in hippocampal slices of guinea pigs. Exp Brain Res. 1986;64(1):94–104. doi: 10.1007/BF00238204. [DOI] [PubMed] [Google Scholar]
- Chamberlin N. L., Dingledine R. Control of epileptiform burst rate by CA3 hippocampal cell afterhyperpolarizations in vitro. Brain Res. 1989 Jul 17;492(1-2):337–346. doi: 10.1016/0006-8993(89)90917-7. [DOI] [PubMed] [Google Scholar]
- Chamberlin N. L., Traub R. D., Dingledine R. Role of EPSPs in initiation of spontaneous synchronized burst firing in rat hippocampal neurons bathed in high potassium. J Neurophysiol. 1990 Sep;64(3):1000–1008. doi: 10.1152/jn.1990.64.3.1000. [DOI] [PubMed] [Google Scholar]
- Collingridge G. L., Herron C. E., Lester R. A. Synaptic activation of N-methyl-D-aspartate receptors in the Schaffer collateral-commissural pathway of rat hippocampus. J Physiol. 1988 May;399:283–300. doi: 10.1113/jphysiol.1988.sp017080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dichter M., Spencer W. A. Penicillin-induced interictal discharges from the cat hippocampus. I. Characteristics and topographical features. J Neurophysiol. 1969 Sep;32(5):649–662. doi: 10.1152/jn.1969.32.5.649. [DOI] [PubMed] [Google Scholar]
- Dingledine R., Korn S. J. Gamma-aminobutyric acid uptake and the termination of inhibitory synaptic potentials in the rat hippocampal slice. J Physiol. 1985 Sep;366:387–409. doi: 10.1113/jphysiol.1985.sp015804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drejer J., Honoré T. New quinoxalinediones show potent antagonism of quisqualate responses in cultured mouse cortical neurons. Neurosci Lett. 1988 Apr 22;87(1-2):104–108. doi: 10.1016/0304-3940(88)90153-x. [DOI] [PubMed] [Google Scholar]
- Dutar P., Nicoll R. A. A physiological role for GABAB receptors in the central nervous system. Nature. 1988 Mar 10;332(6160):156–158. doi: 10.1038/332156a0. [DOI] [PubMed] [Google Scholar]
- Eccles J., Nicoll R. A., Oshima T., Rubia F. J. The anionic permeability of the inhibitory postsynaptic membrane of hippocampal pyramidal cells. Proc R Soc Lond B Biol Sci. 1977 Sep 19;198(1133):345–361. doi: 10.1098/rspb.1977.0102. [DOI] [PubMed] [Google Scholar]
- Gähwiler B. H., Brown D. A. GABAB-receptor-activated K+ current in voltage-clamped CA3 pyramidal cells in hippocampal cultures. Proc Natl Acad Sci U S A. 1985 Mar;82(5):1558–1562. doi: 10.1073/pnas.82.5.1558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gähwiler B. H. Development of the hippocampus in vitro: cell types, synapses and receptors. Neuroscience. 1984 Apr;11(4):751–760. doi: 10.1016/0306-4522(84)90192-1. [DOI] [PubMed] [Google Scholar]
- Gähwiler B. H. Excitatory action of opioid peptides and opiates on cultured hippocampal pyramidal cells. Brain Res. 1980 Jul 21;194(1):193–203. doi: 10.1016/0006-8993(80)91328-1. [DOI] [PubMed] [Google Scholar]
- Hablitz J. J. Picrotoxin-induced epileptiform activity in hippocampus: role of endogenous versus synaptic factors. J Neurophysiol. 1984 May;51(5):1011–1027. doi: 10.1152/jn.1984.51.5.1011. [DOI] [PubMed] [Google Scholar]
- Herron C. E., Williamson R., Collingridge G. L. A selective N-methyl-D-aspartate antagonist depresses epileptiform activity in rat hippocampal slices. Neurosci Lett. 1985 Nov 11;61(3):255–260. doi: 10.1016/0304-3940(85)90473-2. [DOI] [PubMed] [Google Scholar]
- Hill D. R., Bowery N. G. 3H-baclofen and 3H-GABA bind to bicuculline-insensitive GABA B sites in rat brain. Nature. 1981 Mar 12;290(5802):149–152. doi: 10.1038/290149a0. [DOI] [PubMed] [Google Scholar]
- Isokawa-Akesson M., Wilson C. L., Babb T. L. Inhibition in synchronously firing human hippocampal neurons. Epilepsy Res. 1989 May-Jun;3(3):236–247. doi: 10.1016/0920-1211(89)90030-2. [DOI] [PubMed] [Google Scholar]
- Johnston D., Brown T. H. Giant synaptic potential hypothesis for epileptiform activity. Science. 1981 Jan 16;211(4479):294–297. doi: 10.1126/science.7444469. [DOI] [PubMed] [Google Scholar]
- Karlsson G., Olpe H. R. Inhibitory processes in normal and epileptic-like rat hippocampal slices: the role of GABAB receptors. Eur J Pharmacol. 1989 Apr 25;163(2-3):285–290. doi: 10.1016/0014-2999(89)90197-0. [DOI] [PubMed] [Google Scholar]
- Knowles W. D., Schneiderman J. H., Wheal H. V., Stafstrom C. E., Schwartzkroin P. A. Hyperpolarizing potentials in guinea pig hippocampal CA3 neurons. Cell Mol Neurobiol. 1984 Sep;4(3):207–230. doi: 10.1007/BF00733586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krogsgaard-Larsen P., Johnston G. A. Inhibition of GABA uptake in rat brain slices by nipecotic acid, various isoxazoles and related compounds. J Neurochem. 1975 Dec;25(6):797–802. doi: 10.1111/j.1471-4159.1975.tb04410.x. [DOI] [PubMed] [Google Scholar]
- Kunkel D. D., Hendrickson A. E., Wu J. Y., Schwartzkroin P. A. Glutamic acid decarboxylase (GAD) immunocytochemistry of developing rabbit hippocampus. J Neurosci. 1986 Feb;6(2):541–552. doi: 10.1523/JNEUROSCI.06-02-00541.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lloyd K. G., Bossi L., Morselli P. L., Munari C., Rougier M., Loiseau H. Alterations of GABA-mediated synaptic transmission in human epilepsy. Adv Neurol. 1986;44:1033–1044. [PubMed] [Google Scholar]
- Madison D. V., Nicoll R. A. Enkephalin hyperpolarizes interneurones in the rat hippocampus. J Physiol. 1988 Apr;398:123–130. doi: 10.1113/jphysiol.1988.sp017033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malouf A. T., Robbins C. A., Schwartzkroin P. A. Phaclofen inhibition of the slow inhibitory postsynaptic potential in hippocampal slice cultures: a possible role for the GABAB-mediated inhibitory postsynaptic potential. Neuroscience. 1990;35(1):53–61. doi: 10.1016/0306-4522(90)90119-o. [DOI] [PubMed] [Google Scholar]
- Masukawa L. M., Prince D. A. Enkephalin inhibition of inhibitory input to CA1 and CA3 pyramidal neurons in the hippocampus. Brain Res. 1982 Oct 14;249(2):271–280. doi: 10.1016/0006-8993(82)90061-0. [DOI] [PubMed] [Google Scholar]
- Mayer M. L., Westbrook G. L. The physiology of excitatory amino acids in the vertebrate central nervous system. Prog Neurobiol. 1987;28(3):197–276. doi: 10.1016/0301-0082(87)90011-6. [DOI] [PubMed] [Google Scholar]
- McCormick D. A., Connors B. W., Lighthall J. W., Prince D. A. Comparative electrophysiology of pyramidal and sparsely spiny stellate neurons of the neocortex. J Neurophysiol. 1985 Oct;54(4):782–806. doi: 10.1152/jn.1985.54.4.782. [DOI] [PubMed] [Google Scholar]
- McCormick D. A. GABA as an inhibitory neurotransmitter in human cerebral cortex. J Neurophysiol. 1989 Nov;62(5):1018–1027. doi: 10.1152/jn.1989.62.5.1018. [DOI] [PubMed] [Google Scholar]
- Miles R. Synaptic excitation of inhibitory cells by single CA3 hippocampal pyramidal cells of the guinea-pig in vitro. J Physiol. 1990 Sep;428:61–77. doi: 10.1113/jphysiol.1990.sp018200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miles R. Variation in strength of inhibitory synapses in the CA3 region of guinea-pig hippocampus in vitro. J Physiol. 1990 Dec;431:659–676. doi: 10.1113/jphysiol.1990.sp018353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miles R., Wong R. K. Excitatory synaptic interactions between CA3 neurones in the guinea-pig hippocampus. J Physiol. 1986 Apr;373:397–418. doi: 10.1113/jphysiol.1986.sp016055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miles R., Wong R. K. Inhibitory control of local excitatory circuits in the guinea-pig hippocampus. J Physiol. 1987 Jul;388:611–629. doi: 10.1113/jphysiol.1987.sp016634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miles R., Wong R. K., Traub R. D. Synchronized afterdischarges in the hippocampus: contribution of local synaptic interactions. Neuroscience. 1984 Aug;12(4):1179–1189. doi: 10.1016/0306-4522(84)90012-5. [DOI] [PubMed] [Google Scholar]
- Müller W., Misgeld U. Inhibitory role of dentate hilus neurons in guinea pig hippocampal slice. J Neurophysiol. 1990 Jul;64(1):46–56. doi: 10.1152/jn.1990.64.1.46. [DOI] [PubMed] [Google Scholar]
- Neuman R. S., Ben-Ari Y., Cherubini E. Antagonism of spontaneous and evoked bursts by 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX) in the CA3 region of the in vitro hippocampus. Brain Res. 1988 Nov 22;474(1):201–203. doi: 10.1016/0006-8993(88)90686-5. [DOI] [PubMed] [Google Scholar]
- Newberry N. R., Nicoll R. A. A bicuculline-resistant inhibitory post-synaptic potential in rat hippocampal pyramidal cells in vitro. J Physiol. 1984 Mar;348:239–254. doi: 10.1113/jphysiol.1984.sp015107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newberry N. R., Nicoll R. A. Comparison of the action of baclofen with gamma-aminobutyric acid on rat hippocampal pyramidal cells in vitro. J Physiol. 1985 Mar;360:161–185. doi: 10.1113/jphysiol.1985.sp015610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olpe H. R., Karlsson G., Pozza M. F., Brugger F., Steinmann M., Van Riezen H., Fagg G., Hall R. G., Froestl W., Bittiger H. CGP 35348: a centrally active blocker of GABAB receptors. Eur J Pharmacol. 1990 Oct 2;187(1):27–38. doi: 10.1016/0014-2999(90)90337-6. [DOI] [PubMed] [Google Scholar]
- Rutecki P. A., Lebeda F. J., Johnston D. 4-Aminopyridine produces epileptiform activity in hippocampus and enhances synaptic excitation and inhibition. J Neurophysiol. 1987 Jun;57(6):1911–1924. doi: 10.1152/jn.1987.57.6.1911. [DOI] [PubMed] [Google Scholar]
- Rutecki P. A., Lebeda F. J., Johnston D. Epileptiform activity induced by changes in extracellular potassium in hippocampus. J Neurophysiol. 1985 Nov;54(5):1363–1374. doi: 10.1152/jn.1985.54.5.1363. [DOI] [PubMed] [Google Scholar]
- Sah P., Hestrin S., Nicoll R. A. Properties of excitatory postsynaptic currents recorded in vitro from rat hippocampal interneurones. J Physiol. 1990 Nov;430:605–616. doi: 10.1113/jphysiol.1990.sp018310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartzkroin P. A., Mathers L. H. Physiological and morphological identification of a nonpyramidal hippocampal cell type. Brain Res. 1978 Nov 17;157(1):1–10. doi: 10.1016/0006-8993(78)90991-5. [DOI] [PubMed] [Google Scholar]
- Schwartzkroin P. A., Stafstrom C. E. Effects of EGTA on the calcium-activated afterhyperpolarization in hippocampal CA3 pyramidal cells. Science. 1980 Dec 5;210(4474):1125–1126. doi: 10.1126/science.6777871. [DOI] [PubMed] [Google Scholar]
- Segal M. Repetitive inhibitory postsynaptic potentials evoked by 4-aminopyridine in hippocampal neurons in vitro. Brain Res. 1987 Jun 30;414(2):285–293. doi: 10.1016/0006-8993(87)90008-4. [DOI] [PubMed] [Google Scholar]
- Sherwin A. L., van Gelder N. M. Amino acid and catecholamine markers of metabolic abnormalities in human focal epilepsy. Adv Neurol. 1986;44:1011–1032. [PubMed] [Google Scholar]
- Soltesz I., Lightowler S., Leresche N., Crunelli V. On the properties and origin of the GABAB inhibitory postsynaptic potential recorded in morphologically identified projection cells of the cat dorsal lateral geniculate nucleus. Neuroscience. 1989;33(1):23–33. doi: 10.1016/0306-4522(89)90307-2. [DOI] [PubMed] [Google Scholar]
- Soltesz I., Lightowler S., Leresche N., Crunelli V. Optic tract stimulation evokes GABAA but not GABAB IPSPs in the rat ventral lateral geniculate nucleus. Brain Res. 1989 Feb 6;479(1):49–55. doi: 10.1016/0006-8993(89)91334-6. [DOI] [PubMed] [Google Scholar]
- Thompson S. M., Gähwiler B. H. Activity-dependent disinhibition. I. Repetitive stimulation reduces IPSP driving force and conductance in the hippocampus in vitro. J Neurophysiol. 1989 Mar;61(3):501–511. doi: 10.1152/jn.1989.61.3.501. [DOI] [PubMed] [Google Scholar]
- Traub R. D., Dingledine R. Model of synchronized epileptiform bursts induced by high potassium in CA3 region of rat hippocampal slice. Role of spontaneous EPSPs in initiation. J Neurophysiol. 1990 Sep;64(3):1009–1018. doi: 10.1152/jn.1990.64.3.1009. [DOI] [PubMed] [Google Scholar]
- Traub R. D., Miles R., Wong R. K. Model of the origin of rhythmic population oscillations in the hippocampal slice. Science. 1989 Mar 10;243(4896):1319–1325. doi: 10.1126/science.2646715. [DOI] [PubMed] [Google Scholar]
- Traub R. D., Wong R. K. Cellular mechanism of neuronal synchronization in epilepsy. Science. 1982 May 14;216(4547):745–747. doi: 10.1126/science.7079735. [DOI] [PubMed] [Google Scholar]
- Yamada K. A., Dubinsky J. M., Rothman S. M. Quantitative physiological characterization of a quinoxalinedione non-NMDA receptor antagonist. J Neurosci. 1989 Sep;9(9):3230–3236. doi: 10.1523/JNEUROSCI.09-09-03230.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]


