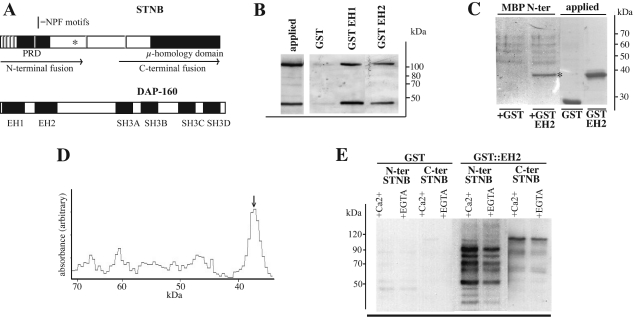
Figure 3. DAP-160 EH domains interact with recombinant STNB.
(A) Representation of the STNB and DAP-160 proteins, indicating the position of the NPF tripeptide sequences, the proline-rich domain (PRD) and the μ-HD of STNB. The EH and SH3 domains of DAP-160 are shown. Also indicated are the regions included in the two fragments of STNB that are expressed as MBP fusions. The asterisk denotes the position of the stop codon in the stnC mutation. (B) Purified MBP–STNB(N-terminus) fusion protein yielded a protein in excess of 100 kDa, as well as a proteolytic product of 50 kDa, as determined by Coomassie Blue staining of an SDS gel (applied sample). Glutathione resin (25 μl) to which GST (25 μg) or GST–DAP-160(EH1) (40 μg) or GST–DAP-160(EH2) fusion protein (28 μg) was bound, was incubated in TBS with 10 μg of the purified MBP–STNB fusion protein. The bound material was eluted with glutathione, and one fifth of the sample was subjected to Western blot analysis using anti-MBP antibodies as the primary antibody. The relative affinity of each EH domain was seen to be approximately equivalent when normalized for the GST–EH domain protein bound to the resin. (C) Amylose resin (25 μl) to which 8 μg of the MBP–STNB N-terminal fragment is bound was incubated with 25 μg of either GST alone or GST–EH2 fusion protein for 4 h at 4 °C. The MBP was eluted with maltose and the mixture run on SDS/PAGE and the gel stained with Coomassie Blue. Again the MBP–STNB fusion expressed in E. coli was seen to fragment. GST and GST-fusion proteins (12.5 μg of each) were run as controls. The asterisk indicates the GST–EH2 protein eluted with maltose. (D) Densitometric scan of the +GST–EH2 lane from the gel in (C). The arrow indicates the GST–EH2 fusion protein. Integration under these curves along with average estimates of the molecular mass of the MBP–STNB N-terminal fragments suggests an approximately equimolar level of the two protein species. (E) Glutathione resin to which either GST (25 μg) or the molar equivalent amount of the GST–DAP-160(EH2) fusion protein (40 μg) was bound, were incubated with E. coli lysates expressing equivalent amounts (as estimated by the signal generated with anti-STNB antibodies on Western blots) of either the N-terminal or C-terminal fragments of STNB as MBP-fusion proteins. The binding was carried out in TBS with either 100 μM Ca2+ or 1 mM EGTA. The bound material was eluted with glutathione and subjected to Western blot analysis using anti-MBP antibodies as the primary antibody. Exposure times for the N-terminal- and C-terminal-containing lysates were 10 s and 1 min respectively. This indicates the relative binding capacity of the STNB N- and C-terminal fragments.