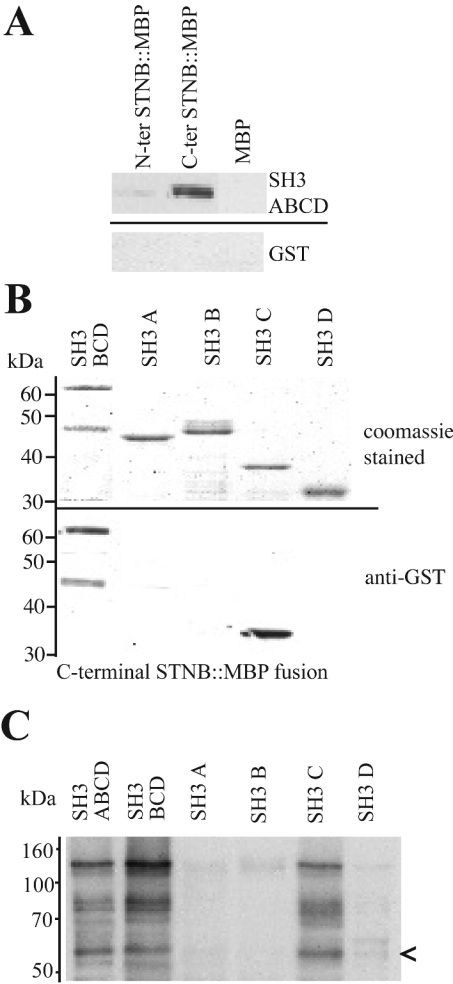
Figure 4. DAP-160 SH3 domains interact with recombinant STNB.
(A) Approximately molar equivalent amounts of MBP alone (20 μg) and MBP-fusion protein N- and C-terminal STNB fragments (50 μg) of STNB were bound to amylose resin and mixed with 50 μg of purified GST–DAP-160 SH3 domain protein containing all four SH3 domains. The resulting material, after washing, was eluted, and one tenth of the eluate was run on SDS/PAGE, Western blotted and probed with anti-GST antibodies. The C-terminal fragment of STNB preferentially interacts with the DAP-160 SH3 domains. (B) An experiment, as outlined in (A), using the MBP–C-terminal STNB fragment (50 μg) bound to amylose resin, but with separate GST–DAP-160 fusion proteins consisting of GST–SH3BCD, GST–SH3A, GST–SH3B, GST–SH3C and GST–SH3D (50 μg of each/assay). The upper panel shows the Coomassie Blue stained gel of the applied fusion proteins (10 μg of each), and the lower panel shows the resulting Western blot, one fifth of the total elution from the resin, probed with anti-GST antibodies. The SH3C domain specifically interacts with STNB. (C) The reverse of the experiment shown in (B) where each of the GST–SH3 domain fusion proteins (50 μg of each) are bound to glutathione resin and used to pull down STNB MBP–STNB(C-terminus) from an E. coli lysate. The blot is probed with anti-MBP antibodies. The arrow indicates a proteolytic product of 58 kDa that is still able to bind to the SH3C domain.