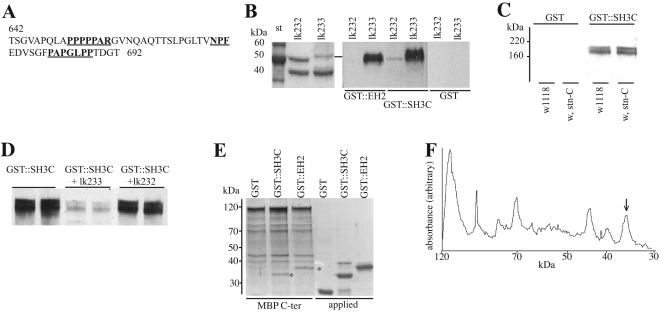
Figure 5. Further delineation of the interaction of the DAP-160 SH3C domain with STNB.
(A) Sequence of part of the C-terminal fragment of STNB indicating the possible SH3 binding sites (underlined). The MBP–STNB fusion construct starts at amino acid residue 642 of STNB. (B) Two MBP fusion constructs lk232 and lk233 contained an out-of-frame nonsense amino acid sequence or residues 642–692 respectively. These fusion proteins were purified, and both showed the expected fusion protein (upper band), as well as free MBP (lower band). The panel on the left-hand side is the Coomassie Blue stained gel. The blots containing these two fusion proteins were overlayed with GST–DAP-160(EH2) fusion protein, with GST–DAP-160(SH3C) fusion protein or with GST alone, and then developed with anti-GST antibodies. The fragment from lk233 binds both GST fusions, but not GST. (C) Overlay of the GST–SH3C fusion on Drosophila head fractions. The experimental method is identical to that described for (B), but the blot contains crude extracts from the heads of wild-type and stnC flies showing that the overlay is not identifying STNB on these blots. These proteins have been tentatively identified as clathrin [22]. (D) Blots of head fractions as described in (C) were overlayed either with GST–SH3 alone or with GST–SH3 to which had been added 10-fold molar excess of either the lk232 or lk233 fusion proteins. This shows that the region of STNB encoded by the clone lk233 can competitively inhibit the binding of the SH3C domain to the presumptive clathrin. (E) Amylose resin (25 μl) to which 28 μg of MBP–STNB C-terminal fragment is bound was incubated with 50 μg of GST, GST–SH3C or GST–EH2 at 4 °C for 4 h, eluted with maltose and then loaded on to SDS/PAGE along with 12.5 μg of GST and each of the GST fusion proteins as standards. The gel was stained using Coomassie Blue. The asterisks indicate the GST-fusion proteins bound to the MBP-fusion protein. (F) Densitometric scan of the GST–EH2 lane from (E). The arrow indicates the peak corresponding to the GST–EH2 protein. Integration under this curve, and making allowances for the change in molecular mass associated with the proteolytic products of the MBP–STNB fusion, gives an approximate molar ratio of 1:4 for GST–EH2/MBP–STNB. A similar value is calculated for the binding of the GST–SH3C domain.