Abstract
Background
People living with HIV (PLWH) in sub-Saharan Africa are vulnerable to end organ dysfunction such as albuminuria, which is associated with an increased risk of cardiovascular and renal events. However, the prevalence of persistent albuminuria among PLWH in Africa is unclear. This observational prospective study assessed for persistence of albuminuria in a cohort of PLWH on longterm antiretroviral therapy (ART) across various HIV service platforms in Gaborone, Botswana.
Methods
A subgroup of ART treated PLWH (n = 867) from a larger cross-sectional study (n = 1537) assessing prevalence of albuminuria among PLWH at a referral hospital HIV clinic and satellite HIV clinics in Gaborone, were invited to participate in a 12-month long albuminuria prospective cohort study between January 2020 and March 2022. During three planned study visits, albumin-creatinine (ACR) was computed using urine albumin measured using immunoturbidimetric assay and urine creatinine using colorimetric assay (Jaffe method), at the Botswana Harvard HIV Reference Laboratory. ACR trajectory groups were identified using growth mixture models, and factors associated with ACR trajectory were analyzed using modified Poisson regression.
Results
Among the 623 adults with complete data for all 3 study visits, their baseline median age was 50 (42–57) years with a median HIV disease duration of 13.1 (8.7–16.7) years, and 266 (42.7%) of them were female. Study participants were categorized into two ACR trajectory groups: low increasing ACR, N = 290 (46.5%) and moderate increasing ACR, N = 333 (53.5%) groups. ACR increased by 4.7 mg/g in the slow increasing ACR groups versus 11.5 mg/g in the moderate increasing ACR trajectory group by end of follow-up. Active use of Tenofovir, aRR 1.27 [95% CI 1.02–1.60], p = 0.036, or ACEi/ARB, aRR 1.31 [95% CI 1.07–1.61], p = 0.008, and an elevated baseline ACR, aRR 1.34 [95% CI 1.28–1.41], p < 0.001, were all associated with being in the moderate increasing ACR trajectory group.
Conclusion
Chronic, treated HIV infection was associated with more than 5 fold increase in ACR over 12 months. Future studies should explore the extent to which modifiable risk factors and HIV specific factors for progressive increase in albuminuria could be aggressively controlled among PLWH.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12879-025-10721-z.
Keywords: Human immuno-deficiency virus, Botswana, Africa, Inflammation, Albuminuria
Significance
What is already known on this topic- Prevalence of albuminuria varies globally across different global people living with HIV. However, little is known about persistent albuminuria among PLWH in sub-Saharan Africa.
What this study adds- HIV is associated with progressive albuminuria among PLWH despite longterm ART. Both modifiable cardiovascular risk factors, and HIV related factors, are linked to progressive albuminuria among PLWH.
How this study might affect research, practice or policy- Future studies investigating effect of traditional cardiovascular risk factors and use of ACEi/ARB among PLWH, on progression of albuminuria will provide additional insights on persistent albuminuria and its associated clinical complications.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12879-025-10721-z.
Background
Albuminuria is an early manifestation and an independent predictor of cardiovascular events in the general population, even in the absence of diabetes or hypertension [1]. Albuminuria is found more frequently and occurs earlier in people living with HIV (PLWH) [2, 3, 4] and carries a high mortality risk [5]. This may be due to direct kidney injury from Anti-Retroviral Therapy (ART) such as Tenofovir [6, 7], or through inflammation from HIV [3], as well as environmental factors such as smoking [8]. Detecting albuminuria may therefore identify PLWH at heightened risk for future cardiovascular disease and mortality.
In the treatment of hypertension in HIV a few small studies have demonstrated improvement in albuminuria with the use of an Angiotensin Receptor Blocker (Telmisartan) [9, 10]. However, it is unknown whether this translates into benefit in the population of PWLH in sub-Saharan Africa with albuminuria irrespective of whether they have hypertension.
Botswana has a generalized hyper-endemic HIV epidemic and in 2021 had the third highest prevalence of HIV among adults in the world (21%), behind Lesotho and Eswatini [11]. With a comprehensive and progressive strategy utilizing universal access to ART for PLWH, Botswana has surpassed the UNAIDS 95-95-95 targets to help end the HIV epidemic [12, 13]. With good viral control, PLWH now have a longer lifespan but are faced with an increased risk of developing cardiometabolic diseases [14].
Successful viral suppression has been achieved through the decentralization of HIV care in many lower- and middle-income countries as physician-led secondary-care has progressed to nurse-led primary-care [15]. Decentralization has improved access to ART treatment, however comphrehensive service provision in the primary-care setting is vital to ensure healthcare is equitable, and optimize management of co-morbidities. The screening and management of indicators for non-communicable diseases such as albuminuria is limited across different levels of HIV care both in Botswana and elsewhere in the region. In sub-Saharan Africa, very little is known about the longitudinal changes in albuminuria in this patient population and the contextual factors that may contribute to differences in prospective changes in albuminuria.
Methods
Aim
The current study aimed to characterize prospective changes in albuminuria among ART treated PLWH across different levels of HIV care service delivery in Gaborone, Botswana.
Design, setting, participants
PLWH on antiretroviral therapy (ART), aged 21 years or older, who were participating in an albuminuria cross-sectional study were invited to participate in a sub-study to assess longitudinal changes in albuminuria. The study target was that about half (50%) of main study participants would agree to being followed up for 12 months, and that most (> 80%) would have complete follow-up data. The study was conducted at a referral hospital HIV clinic where care is provided by medical specialists. Recruitment also took place at HIV satellite clinics across the city of Gaborone which are primarily staffed by nurses/junior medical officers. These satellite clinics differ in their level of care, with prescribing practices and clinical management potentially varying compared to the specialist care provided at the referral hospital HIV clinic. At all these clinical sites, patients receive free medical services and are scheduled to attend HIV clinic every 6 months if they are virally suppressed. All clinics provide multi-month dispensing of antiretroviral therapy of up to 6 months. The national HIV treatment guidelines is silent of screening for albuminuria, and there is little integration of other medical services into the HIV service platform. All required sample (blood, urine, etc.) testing is done in the laboratory and there is no or very limited point of care testing for non communicable diseases (glucose, albuminuria, etc.) in the clinic consulting rooms.
Inclusion and exclusion criteria
Inclusion criteria includes (1) participation in the main albuminuria prevalence study, (2) living with HIV, (3) on antiretroviral therapy at time of enrollment, (4) willingness to provide a morning urine sample during HIV clinic appointment, and (5) willingness to attend month 6 and 12 study visits.
Persons were excluded from participation if they reported any condition or activity that is associated with albuminuria including muscle injury, use of medications.
associated with muscle breakdown, recent heavy physical activity and fever [16, 17].
Data sources
Baseline (month 0) demographic data and medical information were available from the parent study but these were updated at each future study visit (month 6 and 12) among those who consented to participate in the prospective study. Future study visits were linked to the HIV care clinic visit- which meant that if the HIV clinic visit date changed, there was no added burden for a separate additional study visit in this observational study. All study data was directly entered into the study electronic data collection system, RedCap©.
Exposure variable
The main exposure variable was time measured and assessed at 0, 6 and 12 months. Participants were allowed to attend as early or late as 3 months from their scheduled 6 or 12 month study visit, especially if the delayed study visit was to coincide with the HIV clinical care visit.
Outcome variable
The main outcome variable was albuminuria (as a continuos variable) measured as albumin creatinine ratio (ACR) in mg/g. For month 0 visit, ACR was measured using participant morning urine samples submitted immediately after enrolment. Month 6 and 12 ACR was ascertained from urine samples (stored at 2–8 degrees celsius) that we batched for analysis. At all visits, urine albumin was measured using immunoturbidimetric assay while urine creatinine was measured using a colorimetric assay (Jaffe Method). The laboratory internally computed the ACR and shared the ready ratio for entry into RedCap©. All testing took place at the Botswana Harvard HIV Reference Laboratory, Gaborone, Botswana.
Potential confounders
Location of enrollment into the study was captured because clinical care and prescribing patterns may differ between clinics. Enrollment took place at a referral hospital HIV clinic where care is provided by medical specialists. On the other hand, recruitment also took place at HIV satellite clinics across the city of Gaborone. These satellite clinics are staffed by nurses/junior medical officers whose level of care and prescribing patterns may differ when compared to the referral hospital HIV clinic. Key demographic data including age and sex were obtained from clinical records and confirmed during participant interview. Participant medical records were reviewed at each visit and potential confounders such as diabetes mellitus and kidney diseasewere recorded. Medication history was abstracted from clinical records and confirmed with participants at each study visit. Use of Angiotensin Converting Enzyme inhibitors (ACEi) and Angiotensin-receptor 1 blockers (ARB) were recorded and the rest of the hypertension drugs not usually indicated for clinical albuminuria recorded as “other blood pressure medications”. Waist and hip circumference were measured in triplicate using a non-stretchable tape measure following standard protocols, and a mean of the three measurements used to calculate waist-hip ratio (WHR). Blood pressure (BP) was also measured following standard procedure in triplicate from the right arm at each visit with approximately 1–2 min between measurements. A mean right arm systolic and diastolic BP was then used from each visit for longitudinal data analysis.
Statistical analysis
Albuminuria trajectory groups
Growth mixture modelling (GMM) was used to identify albuminuria trajectory groups, first assuming linear, and then quadratic growth. While we used model fitness parameters such as AIC and BIC in the final model selection, additional model selection criteria included, (1) clinically meaningful interpretation, (2) sizeable number of participants per albuminuria trajectory group (> 10% of the data set) to provide meaningful interpretation and/or comparision with other identified ACR trajectory groups, etc. Baseline categorical variables were compared using chi-squire or Fisher’s test, while continuous variables were compared using Mann-Whitney U test. Finally, we evaluated for factors associated with ACR trajectory using modified Poisson regression and reported risk ratios. Covariates with p-value < 0.2 in univariate models were included in the final multi-variate models where p-value < 0.05 was considered statistically significant. If generalized variance inflation factors (GVIF) for categorical variables or variance inflation factors (VIF) for continuous variables exceeded 2 during assessment for collinearity, such factors were not included in the same multivariate models, and results of the sensitivity analysis provided in supplementary tables. We also include a supplementary table depicting final multivariable models using a criteria of p < 0.2 versus p < 0.1 for selection of variables from univariate analysis. All statistical analysis was conducted using R statistical software©, 2023, version 4.3.2.
Results
Between January 2020 and March 2022, about 57% of participants (7% more than predicted) in an ongoing study of albumuniria prevalence were randomly selected and consented for a repeat albuminuria assessment at 6 and 12 months post the baseline visit (Fig. 1). Repeat albuminuria assessment were naturally completed for those whose scheduled study visit coincided with their HIV clinic visit, thereby resulting in a final analysis population of 623 participants (Fig. 1), which approached 100% of the anticipated study analysis population [the initial study plan was that > 80% of those who consented for 12 month of follow up would have complete data- and the current study analysis population is 81% of the anticipated population].
Fig. 1.
Source population, prospective longitudinal assessment of albuminuria cohort and analysis population
ACR trajectory groups
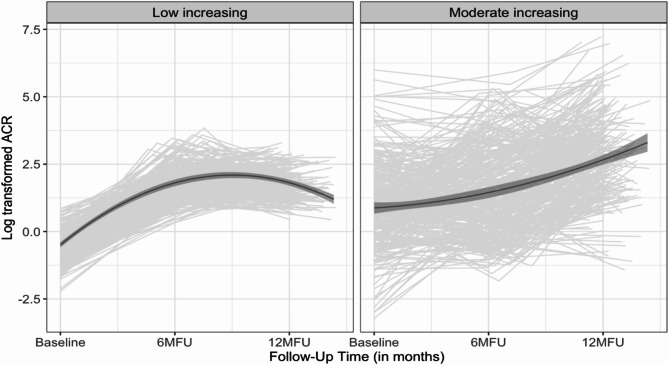
Growth mixture modelling (quadratic term) revealed two separate ACR trajectory groups with low increasing ACR trajectory, N = 290 (46.5%) and moderate increasing ACR trajectory, N = 333 (53.5%), groups respectively as shown below in Fig. 2. See supplementary Table 1 for model fitness parameters and supplementary Fig. 1 compares the distribution of ACR values by ACR trajectory groups at the three study visits.
Fig. 2.
Log transformed ACR trajectory groups during 12 months of follow-up
Baseline characteristics
The study enrolled 623 PLWH with a median age of 50 (42–57) years and 266 (42.7%) of them were female with a median HIV disease duration of 13.1 (8.7–16.7) years. The moderate increasing ACR trajectory group had worse off cardio-metabolic profile at baseline. Detailed differences in base line characteristics by ACR trajectory group are shown in Table 1.
Table 1.
Baseline characteristics of the study population
| Clinical Characteristic | Total (N = 623) |
Low increasing N = 290 (46.5%) |
Moderate increasing N = 333 (53.5%) |
p-value |
|---|---|---|---|---|
| Age (years)# | 50 (42–57) | 48 (42–56) | 51 (44–58) | 0.004 |
| Females | 266 (42.7%) | 104 (35.9%) | 162 (48.6%) | 0.002 |
| HIV duration (years)# | 13.1 (8.7–16.7) | 12.8 (7.8–16.5) | 13.7 (9.4–16.8) | 0.043 |
| ART duration (years)# | 12.2 (7.7–15.7) | 11.9 (6.7–15.3) | 12.6 (8.5–16.1) | 0.020 |
| Viral load < 400 copies/ml | 621 (99.7%) | 289 (99.7%) | 332 (99.7%) | > 0.999 |
| CD4 count cells/ul# |
579.0 (439.5–770.5) |
561 (422–763) |
597 (453–778) |
0.114 |
| Tenofovir containing ART | 582 (93.4%) | 276 (95.2%) | 306 (91.9%) | 0.174 |
| Diabetes Mellitus | 13 (2.1%) | 3 (1.0%) | 10 (3.0%) | 0.099 |
| Duration of Diabetes Mellitus (years)# | 6.0 (4.1–10.8) | 5.9 (4.9–6.0) | 8.7 (4.3–11.0) | 0.469 |
| Hypertension | 178 (28.6%) | 49 (16.9%) | 129 (38.7%) | < 0.001 |
| Duration of Hypertension (years)# | 7.3 (3.5–11.5) | 8.8 (3.8–12.2) | 6.9 (3.3–11.2) | 0.476 |
| Care at Main Referral HIV clinic | 248 (39.8%) | 95 (32.8%) | 153 (45.9%) | 0.001 |
| ACEi/ARB | 111 (17.8%) | 23 (7.9%) | 88 (26.4%) | < 0.001 |
| Other BP medications | 168 (27.0%) | 46 (15.9%) | 122 (36.6%) | < 0.001 |
| Systolic blood pressure (mmHg)# |
127.0 (115.0–138.4) |
124.5 (112.5–133.9) |
129.8 (116.5–143.5) |
< 0.001 |
| Diastolic blood pressure (mmHg)# |
83.5 (75.0–91.0) |
82.0 (72.5–90.0) |
85.0 (76.0–91.5) |
0.008 |
| High risk waist circumference (cm) [> 102 for men and > 88 for females] | 211 (33.9%) | 85 (29.3%) | 126 (37.8%) | 0.033 |
| High risk Waist-Hip Ratio [> 0.9 for males and > 0.85 for females] | 330 (53.0%) | 131 (45.2%) | 199 (59.8%) | < 0.001 |
| Baseline ACR (g/mg) | 1.0 (0.5–2.9) | 0.6 (0.4–0.9) | 2.7 (1.1–6.5) | < 0.001 |
| 6MFU ACR (g/mg) | 5.7(2.5–12.6) | 6.3 (4.1–10.5) | 3.5 (1.1–17.2) | < 0.001 |
| 12MFU ACR (g/mg) | 7.5 (4.1–19.5) | 5.4 (3.7–8.3) | 15.6 (5.2–37.0) | < 0.001 |
| 12 month ACR change# | 6.0 (2.9–17.4) | 4.7 (3.1–7.3) | 11.5 (2.57–31.9) | < 0.001 |
#Median (Interquatile range)
None of the participants had known chronic liver disease or chronic viral hepatitis B or C
Only one (1) participant reported having knonwn chronic kidney disease. Only one (1) participant reported having known cardiovascular disease. Only two (2) participants had macroalbuminuria. The participants with chronic kidney disease, cardiovascular disease and macroalbuminuria belonged to the “stable increasing” ACR category
ART; Antiretroviral therapy: HIV; Human Immunodeficieny Virus: ACEi/ARB; Angiotensin Converting Enzyme Inhibitor/Angiotension Receptor Blocker: ACR; Albumin-Creatininre Ratio: MFU; Month follow-up
Unadjusted & adjusted association between ACR and clinical factors
Table 2 below shows that other than duration of diagnosis of hypertension, all studied clinical factors were associated with an elevated risk ratio in the moderate increasing ACR trajectory as compared to being in the low increasing ACR trajectory group. However, in the final multivariate analysis current use of Tenofovir and ACEi/ARB plus having a higher baseline (month 0 study visit) ACR were all significantly associated with elevated risk ratio for belonging in the moderate increasing ACR trajectory group than the low increasing ACR trajectory group. Supplementary Table 2 shows the same conlusions of adjusted analysis after exchanging the four variables that were considered highly collinear to be included together in the original multivariate model building.
Table 2.
Univariate & multivariate modelling ACR trajectory groups
| Variable | Unadjusted | Adjusted | ||||
|---|---|---|---|---|---|---|
| RR | 95%CI | p-value | aRR | 95% CI | p-value | |
| Sex (Female)***** | 1.27 | 1.10–1.47 | 0.001 | 1.13 | 0.98–1.31 | 0.092 |
| Age (per 5 yrs) | 1.06 | 1.02–1.09 | 0.001 | 1.01 | 0.97–1.05 | 0.598 |
| Main Clinic (PMH) | 1.29 | 1.11–1.48 | < 0.001 | 1.04 | 0.88–1.22 | 0.655 |
| HIV duration (per 2yrs)* | 1.03 | 1.00–1.06 | 0.063 | n/a | n.a | n/a |
| ART duration (per 2yrs)* | 1.04 | 1.01–1.07 | 0.016 | 1.02 | 0.98–1.05 | 0.338 |
| CD4 count (per 100 cells) | 1.01 | 1.00–1.02 | 0.105 | 1.00 | 0.99–1.01 | 0.519 |
| Tenofovir exposure | 0.81 | 0.64–1.03 | 0.083 | 1.27 | 1.02–1.60 | 0.036 |
| Hypertension** | 1.58 | 1.38–1.81 | < 0.001 | 1.07 | 0.87–1.30 | 0.526 |
| Duration of hypertension (per 2yrs) | 1.00 | 0.98–1.02 | 0.821 | n/a | n/a | n/a |
| ACEi/ARB | 1.66 | 1.45–1.89 | < 0.001 | 1.31 | 1.07–1.61 | 0.008 |
| Other BP medications** | 1.57 | 1.37–1.79 | < 0.001 | n/a | n/a | n/a |
| Diastolic BP (per 5mmHg)*** | 1.04 | 1.01–1.07 | 0.003 | n/a | n/a | n/a |
| Systolic BP (per 5mmHg)*** | 1.04 | 1.02–1.05 | < 0.001 | 1.00 | 0.98–1.02 | 0.984 |
| WC (per 5 cm)**** | 1.04 | 1.02–1.06 | < 0.001 | n/a | n/a | n/a |
| High risk WHR | 1.32 | 1.13–1.54 | < 0.001 | 1.09 | 0.95–1.26 | 0.232 |
| Baseline ACR (log-transformed) | 1.35 | 1.29–1.41 | < 0.001 | 1.34 | 1.28–1.41 | < 0.001 |
*, **, *** and ****were collinear
ART; Antiretroviral therapy: HIV; Human Immunodeficieny Virus: ACEi/ARB; Angiotensin Converting Enzyme Inhibitor/Angiotension Receptor Blocker: ACR; Albumin-Creatininre Ratio: DBP; Diastolic blood pressure
Discussion
Some PLWH in Botswana are at risk of Major Adverse Renal Events (MARE) [18] as evidenced by increase in albuminuria in the both ACR trajectory groups in our study. Consistent with prior observations, traditional risk factors for albuminuria were more prevalent or elevated in the “moderate increasing ACR trajectory” group. These include older age, diagnosis of hypertension or higher systolic/diastolic pressure and central obesity [19]. Among HIV specific factors, those with longer HIV/ART exposure time, and also receiving care at the main referral HIV clinic were more likely to belong to the “moderate increasing ACR trajectory” group.
In the Women’s Interagency Health Study (WIHS) from the Unites States, persistent and worsening albuminuria among women living with HIV was associated with systolic blood pressure [20]. Although our study population was not limited to women, we observed an association between increasing blood pressure and albuminuria in the ‘moderate increasing ACR trajectory group’ only in unadjusted analysis as compated to the adjusted analysis. Our smaller sample size may have predcluded the ability to adequately assess the link between blood pressure and ACR trajectory group. Of note, systolic blood pressure has been linked persistence of albuminuria in several other patients cohorts: pediatric cohort from Brazil [21], adult cohort from Pakistan [22], adult cohort comprising relatives of patients with known chronic kidney disease in Egypt [23], and a large observation study of adults (n > 6,000) in the Netherlands [24]. Therefore, relationship between systolic blood pressure and persistent albuminuria among PLWH requires further investigation.
Using growth mixture modelling allowed us to effectively delineate the effect of site of HIV care on outcome of persistent albuminuria. While participants in the “moderate increasing ACR trajectory group” were more likely to receive care at the main refererral hospital HIV clinic, in the final fully adjusted results, the effect of site of care seen in unadjusted analysis no longer reached statistical significance. This favourable neutral effect of albuminuria outcomes irrespective of level of care where HIV services are offered is an encouraging observation for some indicator ofquality of primary care for both HIV and other medical cormorbidities in this setting- or it may just reflect that patients with more cormobidities are likely to remain at the hospital based HIV clinic while those with no or fewer comorbidities are the ones being transferred out to satellite clinics. Future studies that will evaluate the degree and quality of integrated HIV/NCD care in this setting, by site of care, may provide other alternative explations. Further to this, the referral hospital HIV clinic may have more access to tools for testing for albuminuria and albuminuria lowering medications such as ACEi/ARBs compared to the satellite clinics. This is supported by significantly higher use of ACEi/ARB at the main hospital HIV clinic when compared to saltellite clinics in our study. More studies in quality improvement and patient centered outcomes research in sub-Saharan Africa will help identify and address contextual factors that may produce different patient outcomes within the same healthcare system.
The use of ACEi/ARBs was associated with increased likelihood of being in the “moderate increasing ACR trajectory group” in this cohort, a finding that differs from prospective studies of albuminuria in other patient populations such as those with Diabetes Mellitus [25] when initiated on ACEi/ARB. Given our current study size, we were limited to assess the full effect of ACEi/ARB because of the small sample size (n ~ 600), limited follow-up (~ 12 months), few repeat measures (~ 3 per participant), and finally, lack of randomization to ACEi/ARB versus other treatments. There is likelihood of confounding by indication given that majority of those who were prescribed ACEi/ARB received care from referral hospital larger HIV clinic- where there were speciaists who may identify clients who would benefit from early exposure to ACEi/ARB. Our small sample size precluded us from accounting for confounding by indication (propensity score matching, etc.) to evaluate fully the effect of ACEi/ARB in this cohort. Majority of the literature on use of ACEi/ARB among PLWH have focused on longterm benefits among those with HIV associated nephropathy [26], and not the more likely scenario of PLWH without HIV associated nephropathy. However, in a small study of 13 ART treated PLWH in Italy, the use of Telmisartan (ARB) was associated with a statistically significant decline in albuminuria [9]. Given reports showing cardiovascular benefits of ACEi/ARB in a large Veterans Hospital based cohort of PLWH in the United States of America [27], further research on use of ACEi/ARB in this population in sub-Saharan Africa would provide additional insights on the impact of ACEi/ARB on persistent albuminuria among PLWH.
In one of the largest studies of the association between obesity and albuminuria in the general adult population, the Fifth Korea National Health and Nutrition Examination Survey done in 2011 with 3841 participants, women in the fourth quartile by waist circumference and waist-hip ratio were more likely to have albuminuria. Of note, this was a larger data set than ours (n = 623), and there were more females (n = 2111) than males (n = 1730) [28]. While those with elevated waist-hip ratio were more likely to be categorized into the “moderate increasing ACR trajectory group”, our study however did not reveal an association between central obesity (measured using waist-hip ratio) and albuminuria in the final fully adjusted analysis. For now, we know that at least body mass index (BMI), which is a less sensitive marker of central obesity, was associated with a lower risk of proteinuria [19] in sub-Saharan Africa in a larger study of PLWH. Future studies using larger samples sizes and different measures of obesity (such as body mass index, waist-circumference, magnetic resonance imaging, etc) among PLWH will shed more light in the relationship between obesity and persistent albuminuria in SSA.
Data on the relationship between antiretroviral therapy (ART) and albuminuria is mixed- in part because some studies are cross-sectional, duration and type of ART use is mixed, different cut off points for albuminuria (any albuminuria, clinical cut-off points of e.g., > 30mcg/mg or other) and sometimes the cohorts being studied are too region or race specific even though more recent studies suggest that all these factors matter may influence observed associations [19]. For instance, a recent global evaluation of proteinuria and albuminuria in a representative sample of 2,791 PLWH revealed that abnormal proteinuria and albuminuria were more common among PLWH in sub-Saharan Africa, and that use of tenofovir disoproxil fumerate (TDF) [19] was strongly linked to proteinuria, but not albuminuria (> 30mcg/mg). This study did not explore the relationship between “any ACR” value versus TDF as we did in our current smaller study. However, a previous cross-sectional study of albuminuria among PLWH in Botswana also reported that duration of exposure to tenofovir disoproxil fumerate was associated with an estimated 13% increase risk of having albuminuria [29]. Studies involving chronic use of TDF among PLWH have revealed a similar link in other settings [30]. Participants in the current cohort were on the Tenofovir disoproxil fumerate (TDF) and not tenofovir alafenamide (TAF) which is known to reduce albuminuria in studies where TDF was switched to TAF in PLWH [31]. Similarly, emerging data suggests that participants preferentially initiated on non-TDF ART such as Abacavir/Lamivudine are less likely to experience persistent albuminuria [32]. However, in the multi-center AIDS cohort study (MACS) from the Unites States of America, there was no association between cumulative TDF exposure and albuminuria in a subset of 884 men living with HIV [33]. Of note, this observation is constrained by the fact that MACS was not enriched for a population more likely to have proteinuria or albuminuria such as black women living wth HIV in sub-Saharan Africa [19].
The current study is limited due to its design as a longitudinal observational study and would be strengthened if conducted as a randomized controlled trial to assess clinical and contextual factors associated with persistent albuminuria among PLWH. Ascertainment of albuminuria was limited by a one time assessment due to resource constraints. Ideally, 2–3 measurements per study visit would have provided more stable estimate in longitudinal assessment of ACR. Limited screening for non-communicable diseases (NCDs) (e.g., glucose level, albuminuria, etc.) in this setting limited our ability to have sufficient paticipants with diagnosis of important NCDs that are associated albuminuria such as diabetes mellitus (only 2.1% of our study population had a pre-existing diagnosis of diabetes mellitus). Bias in study site selection and adoption of ACEi/ARB by clinicians at different sites also limit the strength of this study. Further indicators of the quality of care could have been examined to explore this relationship in more detail. Inadequate documentation of comorbidities and the reason for prescription of ACEi/ARB is another limiting factor that may affect interpretation of study findings. Also, the study population lacked enough participants on ACEi/ARB to allow us to draw meaningful information about the impact of ACEi/ARB therapy on ACR. Additionally, because most PLWH in Botswana tend to be on the same national guideline approved ART, we may not have had sufficient variability in ART regimen/products to be able to fully assess the impact of ART of ACR but the potential effect of Tenofovir detected in this study is worth noting especially in the era of nucleoside reverse transcriptase inhibitors (NRTI) sparing dual antiretroviral therapy [34, 35]. Also, because the study did not have the resources to provide eGFR at every study visit (and it is not standard of care), dynamic change of renal function on ACR could not be assessed. A comparison of longitudinal changes in ACR with a similar cohort of adults without HIV in the same setting would have provided additional insights on the impact of HIV in ACR. Notable strengths of our study include the ability to corroborate old findings (such as the association between tenofovir disoproxil fumerate and persistent albuminuria) and newly identify that those who start of with a higher baseline ACR are likely to experience faster increase in ACR.
In conclusion, PLWH experienced persistence and an increase in albuminuria during 12 months of follow-up. Further studies are needed to assess end-organ dysfunction and clinical outcomes from persistent albuminuria among PLWH.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
The study team is grateful to the study participants and healthcare providers and leaders at the sites that took part in this study. We are particularly indebted to participants for their willingness to undergo the study precedures during a very difficult time as follow-up took place during the SARS-CoV-2 pandemic.
Abbreviations
- PLWH
People living with HIV
- ART
Antiretroviral therapy
- HIV
Human immunodeficieny virus
- ACEi/ARB
Angiotensin converting enzyme inhibitor/angiotension receptor blocker
- ACR
Albumin-creatininre ratio
- DBP
Diastolic blood pressure
- GMM
Growth mixture modelling
Author contributions
MM, SL, RG, JJ, SJ and DW designed the study. MM, KK, PP, LM, KM and TM conceptualized the analysis and drafted the manuscript with input from all co-authors. MM, KK, KM and TM revised and finalised the manuscript. MM, PP, and KM contributed to data collection and interpretation. All co-authors read and approved the manuscript.
Funding
We also thank the sponsor our study sponsor, The European & Developing Countries Clinical Trials Partnership (EDCTP)- Fellowship Grant awarded to Mosepele Mosepele [TMA2017CDF-1928]. The study funders did not play any role in the design, data collection, analysis, or interpretation of the data.
Data availability
All data supporting the study’s findings are accessible and will be shared in accordance with the guidelines set by the ethics committees that approved this research. For data requests, interested parties should contact both the corresponding author, Dr. Mosepele Mosepele, and the Ministry of Health of Botswana’s Human Research Subjects Ethics Committee at hhealthresearch@govbots.onmicrosoft.com.
Declarations
Ethics approval and consent to participate
The study protocol was approved by the University of Botswana Institutional Review Board and Ministry of Health Human Research Development Committee (HRDC)-HPDME 13/18/1. Participation was voluntary, and all participants had to sign an informed consent form to participate.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Gerstein HC, et al. Albuminuria and risk of cardiovascular events, death, and heart failure in diabetic and nondiabetic individuals. JAMA. 2001;286(4):421–6. [DOI] [PubMed] [Google Scholar]
- 2.Gupta SK, et al. The effects of highly active antiretroviral therapy on albuminuria in HIV-infected persons: results from a randomized trial. Nephrol Dial Transpl. 2005;20(10):2237–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Szczech LA, et al. Microalbuminuria in HIV infection. AIDS. 2007;21(8):1003–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Baekken M, et al. Microalbuminuria associated with indicators of inflammatory activity in an HIV-positive population. Nephrol Dial Transpl. 2008;23(10):3130–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wyatt CM, et al. Microalbuminuria is associated with all-cause and AIDS mortality in women with HIV infection. J Acquir Immune Defic Syndr. 2010;55(1):73–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ryom L, et al. Association between antiretroviral exposure and renal impairment among HIV-positive persons with normal baseline renal function: the D:A:D study. J Infect Dis. 2013;207(9):1359–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Scherzer R, et al. Association of Tenofovir exposure with kidney disease risk in HIV infection. AIDS. 2012;26(7):867–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pinto-Sietsma SJ, et al. Smoking is related to albuminuria and abnormal renal function in nondiabetic persons. Ann Intern Med. 2000;133(8):585–91. [DOI] [PubMed] [Google Scholar]
- 9.Ucciferri C, et al. Microalbuminuria and hypertension in HIV-infected patients: a preliminary study of Telmisartan. Eur Rev Med Pharmacol Sci. 2012;16(4):491–8. [PubMed] [Google Scholar]
- 10.Vecchiet J, et al. Antihypertensive and metabolic effects of Telmisartan in hypertensive HIV-positive patients. Antivir Ther. 2011;16(5):639–45. [DOI] [PubMed] [Google Scholar]
- 11.Song J, Okano JT, Ponce J, Busang L, Seipone K, Valdano E, et al. The role of migration networks in the development of Botswana’s generalized HIV epidemic. Elife. 2023;12:e85435. 10.7554/eLife.85435. PMID: 37665629; PMCID: PMC10476964. [DOI] [PMC free article] [PubMed]
- 12.Gaolathe T, et al. Botswana’s progress toward achieving the 2020 UNAIDS 90-90-90 antiretroviral therapy and virological suppression goals: a population-based survey. Lancet HIV. 2016;3(5):e221–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Marukutira T et al. A Tale of two countries: progress towards UNAIDS 90-90-90 targets in Botswana and Australia. J Int AIDS Soc, 2018;21(3). [DOI] [PMC free article] [PubMed]
- 14.Tuan J, Igiraneza G, Ogbuagu O. Analysis of drug-drug interactions in patients with HIV and metabolic syndrome. Expert Opin Drug Metab Toxicol. 2024;20(10):953–65. 10.1080/17425255.2024.2401044. Epub 2024 Sep 10. PMID: 39230187. [DOI] [PubMed]
- 15.Bassett IV, et al. Does type of antiretroviral therapy pick-up point influence 12-month virologic suppression in South Africa? AIDS Care. 2024;36(10):1518–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Johnson DW, et al. Chronic kidney disease and measurement of albuminuria or proteinuria: a position statement. Med J Aust. 2012;197(4):224–5. [DOI] [PubMed] [Google Scholar]
- 17.Miller WG, et al. Current issues in measurement and reporting of urinary albumin excretion. Clin Chem. 2009;55(1):24–38. [DOI] [PubMed] [Google Scholar]
- 18.Prischl FC, et al. Major adverse renal events (MARE): a proposal to unify renal endpoints. Nephrol Dial Transpl. 2021;36(3):491–7. [DOI] [PubMed] [Google Scholar]
- 19.Overton ET, Kantor A, Fitch KV, Mosepele M, Aberg JA, Fichtenbaum CJ, et al. Proteinuria and albuminuria among a global primary cardiovascular disease prevention cohort of people with HIV. AIDS. 2025;39(1):31–9. 10.1097/QAD.0000000000004016. Epub 2024 Sep 13. PMID: 39283736; PMCID: PMC11624062. [DOI] [PMC free article] [PubMed]
- 20.Muiru AN, et al. Associations of CKD risk factors and longitudinal changes in urine biomarkers of kidney tubules among women living with HIV. BMC Nephrol. 2021;22(1):296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Alcalde-Ortiz ML, et al. Pediatric kidney dimensions and risk of persistent albuminuria in Mexican adolescents. Kidney Int; 2024. [DOI] [PubMed]
- 22.Ejaz M, et al. Persistent albuminuria as a surrogate marker of chronic kidney damage among newly diagnosed hypertensives: prevalence and risk factors in an urban population in Karachi, Pakistan. Clin Exp Hypertens. 2017;39(1):65–73. [DOI] [PubMed] [Google Scholar]
- 23.Gouda Z, et al. Egypt information, prevention, and treatment of chronic kidney disease (EGIPT-CKD) programme: prevalence and risk factors for microalbuminuria among the relatives of patients with CKD in Egypt. Saudi J Kidney Dis Transpl. 2011;22(5):1055–63. [PubMed] [Google Scholar]
- 24.Ozyilmaz A, et al. Screening for elevated albuminuria and subsequently hypertension identifies subjects in which treatment May be warranted to prevent renal function decline. Nephrol Dial Transpl. 2017;32(suppl2):ii200–8. [DOI] [PubMed] [Google Scholar]
- 25.Vejakama P, et al. Reno-protective effects of renin-angiotensin system Blockade in type 2 diabetic patients: a systematic review and network meta-analysis. Diabetologia. 2012;55(3):566–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Guaraldi G et al. [Inhibition of the renin-angiotensin system in HIV nephropathy]. G Ital Nefrol, 2014;31(1). [PubMed]
- 27.Rethy LB, et al. Antihypertensive class and cardiovascular outcomes in patients with HIV and hypertension. Hypertension. 2021;77(6):2023–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Noh HM, et al. Sex differences in the association between obesity and albuminuria among Korean adults: a cross-sectional study using the Korea National health and nutrition examination survey data. Clin Exp Nephrol. 2017;21(1):27–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Akerele T, Rivera YP, Rwegerera GM. Duration of Tenofovir use and diabetes mellitus predict microalbuminuria among Well-controlled human immunodeficiency Virus-infected patients attending a tertiary clinic in Gaborone, Botswana. Saudi J Kidney Dis Transpl. 2022;33(3):393–403. [DOI] [PubMed] [Google Scholar]
- 30.Falasca K, et al. Predictive factors and prevalence of microalbuminuria in HIV-infected patients: a cross-sectional analysis. BMC Nephrol. 2017;18(1):255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Schwarze-Zander C, et al. Switching Tenofovir disoproxil fumarate to Tenofovir Alafenamide in a real life setting: what are the implications? HIV Med. 2020;21(6):378–85. [DOI] [PubMed] [Google Scholar]
- 32.Wyatt CM, et al. Changes in proteinuria and albuminuria with initiation of antiretroviral therapy: data from a randomized trial comparing Tenofovir disoproxil fumarate/emtricitabine versus abacavir/lamivudine. J Acquir Immune Defic Syndr. 2014;67(1):36–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Jotwani V, et al. Brief report: cumulative Tenofovir disoproxil fumarate exposure is associated with biomarkers of tubular injury and fibrosis in HIV-Infected men. J Acquir Immune Defic Syndr. 2016;73(2):177–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Papot E, et al. Adaption of an ongoing clinical trial to quickly respond to gaps in changing international recommendations: the experience of D(2)EFT. HIV Res Clin Pract. 2022;23(1):37–46. [PMC free article] [PubMed] [Google Scholar]
- 35.Gibas KM, et al. Two-drug regimens for HIV treatment. Lancet HIV. 2022;9(12):e868–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data supporting the study’s findings are accessible and will be shared in accordance with the guidelines set by the ethics committees that approved this research. For data requests, interested parties should contact both the corresponding author, Dr. Mosepele Mosepele, and the Ministry of Health of Botswana’s Human Research Subjects Ethics Committee at hhealthresearch@govbots.onmicrosoft.com.