Abstract
Biofilms formed by Bacillus subtilis confer protection against environmental stressors through extracellular polysaccharides (EPS) and sporulation. This study investigates the roles of these biofilm components in resistance to hydrogen peroxide, a common reactive oxygen species source and disinfectant. Using wild-type and mutant strains deficient in EPS or sporulation, biofilm colonies were cultivated at various maturation stages and exposed to hydrogen peroxide. EPS-deficient biofilms exhibited reduced resilience, particularly in early stages, highlighting the structural and protective importance of the matrix. Mature biofilms demonstrated additional protective mechanisms, potentially involving TasA protein fibers and/or the biofilm surface layer (BslA). In contrast, sporulation showed limited contribution to hydrogen peroxide resistance, as survival was primarily matrix-dependent. These findings underscore the necessity of targeting EPS and other matrix components in anti-biofilm strategies, suggesting that hydrogen peroxide-based disinfection could be enhanced by combining it with complementary sporicidal treatments. This study advances our understanding of biofilm resilience, contributing to the development of more effective sterilization protocols.
Keywords: B. subtilis, Biofilm, Hydrogen peroxide, Endospores, Exopolysaccharides, EPS, ROS, Oxidative stress
1. Introduction
The ability of microorganisms to grow either as swarming cells or sessile biofilms on surfaces offers numerous benefits compared with planktonic growth in a liquid medium. Transitioning from the single cell motility through coordinated swarming to the immobilized lifestyle provides a flexible approach for nutrient utilization, a uniform proliferation, and increased resilience to environmental stressors [[1], [2], [3]]. Thus, living in multicellular and multispecies communities is the most common form of microbial life, which is highlighted by their ubiquitous occurrence in medical, environmental and industrial settings [4,5]. The high impact of biofilms in these fields, notably within clinical environments, is underscored by statistics released by the National Institute of Health (NIH), revealing that biofilm-associated bacteria are responsible for 60 % of all bacterial infections in humans. Furthermore, biofilms account for 80 % of chronic and 65 % of all nosocomial infections [6,7]. Besides the health aspect, biofilms have a high economic relevance. According to a comparative study from 2022, it is estimated that biofilms have an economic impact about USD 5000 billion per year. The values refer to data from 2019, whereby the majority of the costs were caused by corrosive biofilms in industrial facilities [8]. These findings clearly illustrate the seriousness of biofilms as a health threat and the considerable challenge in inactivating them and developing anti-biofilm strategies. The increased resistance against eradication agents can be attributed to at least four categories [9]:
-
(I)
Slow growth: Similar to the stationary phase in unicellular lifestyles, biofilms undergo physiological adaptations due to slower nutrient diffusion, resulting in reduced metabolic activity [4,10]. This adjusted growth kinetics produce more persistent cells with decreased susceptibility to sterilization regimes and antibiotics that target rapid cell growth [11,12].
-
(II)
Communication: The capability to induce biofilm formation is highly dependent on an efficient cell-to-cell communication, termed quorum sensing (QS). QS systems differ between Gram-positive and negative bacteria and are based on the release of chemical signals [13]. Once these signals are recognized, they can be utilized for optimal environmental adaptation. Hence, QS facilitates efficient nutrient utilization and storage, genetic material transfer, and division of labor. This division of labor triggers cell differentiation, encompassing motility, secondary metabolite synthesis, and production of protective biofilm matrix components [14,15,16].
-
(III)
Extracellular matrix (EM): A key factor in enhancing resistance in biofilms is the protective extracellular matrix produced by the inhabiting cells, which they produce autonomously and occupy [17]. This complex matrix comprises biopolymers, such as extracellular polysaccharides (exopolysaccharides = EPS), proteins, lipids, and nucleic acids [18]. The precise composition of the EM varies among species and is dependent on cultivation conditions, substrates and medium [4,19]. It is assumed that the EM is primarily responsible for the resistance to disinfectants and antibiotics, as it prevents penetration either by adsorption or by reacting with the polymers in the EM [20,21].
-
(IV)
Unknown factors: In addition to I-III, there must be further protective factors. For example, EPS are crucial but not essential for biofilm formation and survival [22,23]. Identifying these protective factors could be challenging due to the dynamic nature of biofilms, which is influenced by various compounds and mechanisms.
To maintain hygienic standards which are necessary to enhance human health but also to reduce costs, effective interventions are necessary to minimize potential risks of infections [[24], [25], [26]]. Such interventions include the disinfection of contaminated surfaces with hydrogen peroxide-based chemicals, a registered disinfectant with bactericidal, viricidal, sporicidal and fungicidal properties [25,27]. Hydrogen peroxide (H2O2) is classified as one of the reactive oxygen species (ROS), which can arise from intracellular or extracellular oxidizing events, such as radiation exposure or mitochondrial phosphorylation [28]. H2O2 is a robust oxidizing agent that in the presence of Fe2+ generates highly reactive hydroxyl radicals (˙OH) which are able to damage macromolecules, such as DNA, lipids of the cell membrane, and proteins [[28], [29], [30]]. The imbalance between ROS and protective endogenous compartments results in oxidative stress which subsequently cause cell death [31].
Bacillus subtilis, a Gram-positive facultative anaerobic soil bacterium, forms complex biofilm consortia and exhibits remarkable resistance owing to its ability of sporulation [32]. Thus, this species is commonly utilized as a biological indicator in decontamination studies [33]. In addition to endospore (hereafter referred to as spores) formation, the multicellular lifestyle offers numerous potential protective properties. The EM primarily consists of exopolysaccharides (EPS) and the protein TasA, which forms amyloid fibers essential for the biofilm scaffold. Moreover, B. subtilis biofilms produce a hydrophobin protein coat, formed by BslA, crucial for overall protection against desiccation and selective permeability [[34], [35], [36]]. The organization of this biofilm assembly relies on nutrient availability and extracellular signals [[37], [38], [39]]. Upon signal recognition and adequate environmental conditions, part of the population start to express biofilm-related genes leading to phenotypic heterogeneity that allows coexistence of motile and matrix-producing cells, as well as development of highly resistant spores at the later stage that are assumed to contribute to dispersal [[40], [41], [42]]. This division of labor is tightly regulated and dynamic, with gene expression profiles adapting to environmental conditions [4,43]. These properties demonstrate the remarkable adaptation of biofilms to environmental stressors, making them challenging to inactivate once formed. B. subtilis biofilms have been utilized to dissect the influence of various treatment strategies, including disinfection agents, nanoparticles, and laser irradiation [[44], [45], [46], [47], [48]].
This study seeks to elucidate the impact of hydrogen peroxide on bacterial biofilms lacking EPS and spores, thus contributing to the development of targeted strategies for biofilm control and disinfection. Here, we used the architecturally complex colonies of B. subtilis to evaluate the role of these protective structures.
2. Material and methods
The biofilm cultivation was initiated with spores due to their consistency and stability and the inoculant. Their metabolic inactivity ensures a uniform starting point, preventing variations in metabolic states.
2.1. Spore production and purification
For spore production, 200 μL of an overnight culture were inoculated onto solidified Schaeffer sporulation medium (SSM) [49]. The strains used in this study are listed in Table 1. Plates were incubated for 5–7 days at 37 °C to achieve optimal spore quality and quantity. Spores were harvested from the plate using an inoculation loop and resuspended in 40 mL ddH2O containing sterile glass beads with a size of 3 mm in diameter. This facilitated resuspension using vortexing (2 min) and aided in dispersing cell debris released from the lysed mother cells. To achieve high spore quality and purity, the suspension was repeatedly washed until purity of >99 % spores was confirmed using phase contrast microscopy. The pure spore solution was then stored in glass tubes at 4 °C until utilized.
Table 1.
B. subtilis strains tested in survival ability to hydrogen peroxide. TetR-tetracycline resistance, CatR-chloramphenicol resistance.
| Strain | Genotype | Deficiency | Reference |
|---|---|---|---|
| NCIB3610 | Wild type | None | (38) |
| ZK3660 | ΔepsA-O::TetR | No production of exopolysaccharides within the EM. Remaining matrix components are accomplished by TasA and BslA. | (50) |
| F-030 | comIQ12LsigG::CatR | Deficiency in sporulation (inhibition of late forespore polymerase activities) | (51) |
2.2. Bacterial biofilm cultivation
To obtain biofilms which are standardized and reproducible, a cultivation method according to Fuchs et al. was conducted [52]. Briefly, an inoculum of spores with 108 Colony Forming Units (CFU) mL−1 was utilized and pipetted in the middle of a hydrophilized PTFE filter (polytetrafluoroethylene, Merck Millipore®, pore size 0.4 μM, Merck KGaA, Darmstadt, Germany) with a diameter of 30 mm. This PTFE filter separates the growing biofilm physically from the medium, while enabling water and nutrient diffusion. The inoculated filter material was air dried under sterile conditions for 10 min and placed on solidified minimal medium (MSgg), adapted after Branda et al. [38,53]. As B. subtilis biofilms are highly heterogenous populations with changing cell and EM profile over time, differently matured biofilms were tested here, ranging from 24 h to 72 h. For 0 h, pure inoculum of spores was pipetted onto the filter material. Here, the treatment was performed directly after the drying process. For the sporulation deficient strain of B. subtilis (ΔsigG) an overnight culture with planktonic cells from stationary phase with 108 CFU mL−1 was used as inoculum and 0 h control.
2.3. Sample treatment and CFU determination
Biofilms were grown to distinct development stages and exposed to H2O2. PTFE filters carrying the biofilms were placed into sterile six-well plates. ROS stress was induced by adding 910 μL of 3 % H2O2, diluted in PBS, to each biofilm. After treatment durations of 0, 10, 20, 40, and 60 min, the reaction was stopped by adding 10 mg mL−1 catalase solution. The treated biofilms were transferred into 2 mL reaction tubes containing glass beads (3 mm diameter). To ensure a reliable yield of viable cells, the reaction mixture suspension was also transferred to the 2 mL tube containing the biofilm. Each tube was vortexed for 2 min and survivability was assessed by calculating total CFU (compromises vegetative cells and spores) and the number of spores. To quantify the spores count, an aliquot of the sample was treated at 80 °C for 10 min to inactivate vegetative cells.
2.4. Statistical analysis
The CFU of the total cell mass and spores within the biofilm were calculated at each treatment point of the stress assays as well as the untreated control. Therefore, a dilution series was prepared and plated on LB agar. The average CFU was determined while all data are presented as the average of three biological replicates (n = 3) with according standard deviations. The statistical analysis has been performed by using Tukey's test with SigmaPlot (version 14.5) and OriginLab (version 2023).
3. Results
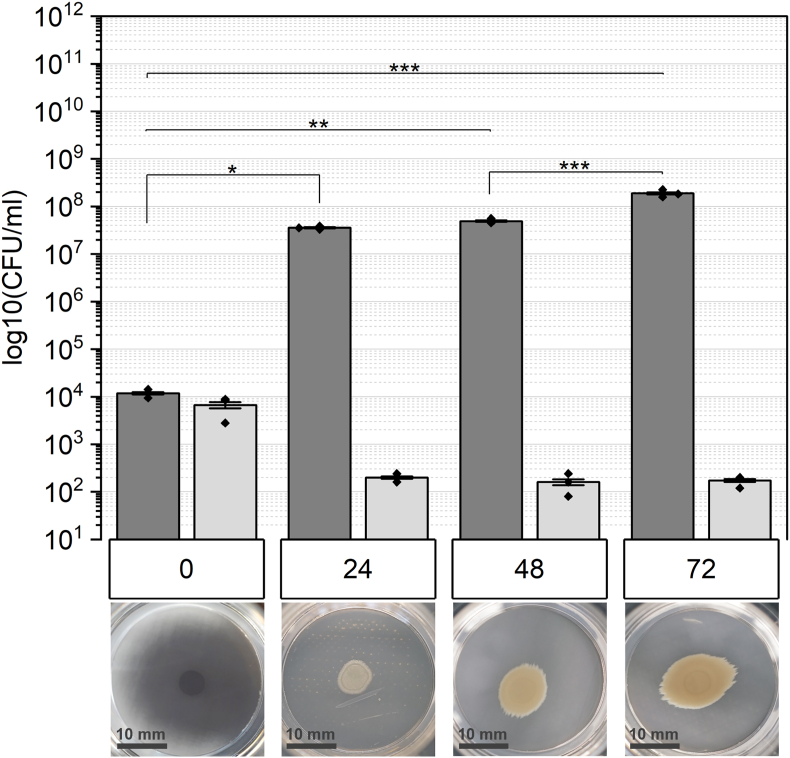
Hydrogen peroxide is known to be an efficient antimicrobial agent and is commercially used as a disinfectant. Numerous studies have demonstrated its efficacy in combating biofilms formed by Pseudomonas aeruginosa and Staphylococcus aureus, targeting both the matrix and the cells [25,54,55]. In this study, our objective was to assess the anti-biofilm activity of hydrogen peroxide against B. subtilis biofilms and the contribution of EPS and spores on survivability. Hence, macrocolony biofilms were cultivated at various growth stages, exposed to hydrogen peroxide and quantified via CFU determination. The cell count within wild-type biofilms consistently rises as maturity progresses, while total CFU comprises a mixture of vegetative cells and spores (Fig. 1). The inoculum (0 h) includes around 105 spores per ml, resulting in little difference in cell count between total CFU and spores at this time point. Spore count peaks in mature (72 h) biofilms, contributing to a notably reduced ratio of vegetative cells. The morphology of wt biofilms varies also with age. In the 24 h stage, the characteristic concentric rings develop. Matured biofilms exhibit increased wrinkling and size. Overall, particularly at 48 and 72 h, biofilms appear as highly heterogenous 3-dimensional structures.
Fig. 1.
The quantity of untreated wild-type (wt) biofilms is depicted according to each stage of biofilm maturation (in hours). The biofilms are composed of a mixture of vegetative cells and spores, termed as “CFU total” (dark grey bars). Additionally, the proportion of spores was quantified and is represented as light grey bars. The macroscopic morphology is illustrated below for each respective time point.
B. subtilis biofilms lacking extracellular polysaccharides within the matrix demonstrate variations in cell count when compared to the wt. Initially, at the 0 h timepoint, CFU total and spores exhibit similarity, approximately 104 CFU ml−1. However, after 24 h, this pattern reverses, with spore count lower than the inoculum and maintaining consistency over time. Meanwhile, the number of vegetative cells experiences a remarkable increase, approximately fivefold higher than the spore count. Additionally, the lack of EPS leads to a noticeably altered biofilm morphology (Fig. 2). The size of (matured) biofilms remains smaller and exhibits a more uniform structure without wrinkles, showing only one visible concentric ring. Overall, biofilms lacking eps appear less dense and thinner compared to wt.
Fig. 2.
The number of untreated biofilms lacking exopolysaccharides (ΔepsA-O) is illustrated for each stage of biofilm maturation (in hours). Dark grey bars represent the total colony-forming units (CFU), while light grey bars indicate the spore count. Statistical significance was determined using Tukey's test with a sample size of n = 3 and indicated by p-values: ∗ <0.05, ∗∗<0.01, ∗∗∗ <0.001.
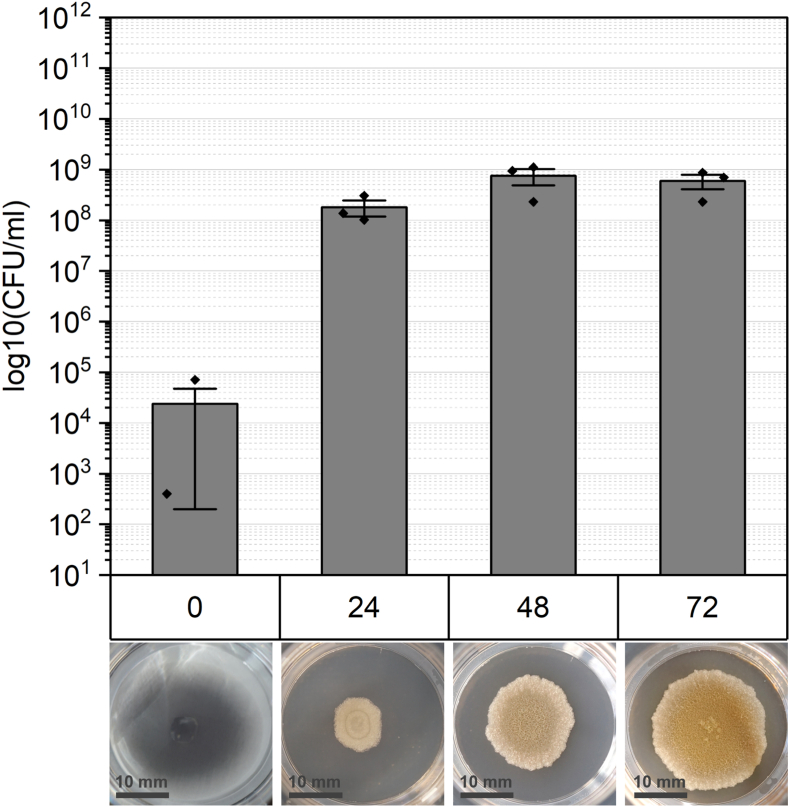
B. subtilis cell aggregates devoid of spores can be attributed to a deletion in the gene encoding the sigma factor G (Table 1, [51]. Starting from 0 h, with approximately ∼104 planktonic cells per ml, the biofilm expands to double this amount at mature levels. Once a certain threshold is reached, the cell count stabilizes with minimal further increase (Fig. 3).
Fig. 3.
The quantity of untreated biofilms lacking spores (ΔsigG) is depicted for each stage of biofilm maturation (in hours). The bars represent only vegetative cells and is termed as “CFU total”. Macroscopic variation among biofilm age is shown below.
Wt biofilms treated with hydrogen peroxide were recovered, and their survival was assessed via CFU determination (Fig. 4). The 0 h timepoint (Fig. 4 0 h) indicates the initial spore count and represents the inoculum reference. Regardless the incubation time, the CFU remains stable, with nearly identical quantities observed between spores and CFU total. Remarkably, even after a 60-min exposure, spore survival remained unaffected. 24 h old consortia showed slight impact in survival and a significant decrease among CFU total, while the spore count was unaffected (Fig. 4 24 h).
Fig. 4.
The quantity of wt biofilms treated with hydrogen peroxide is analyzed based on their maturation levels, represented as A: 0 h, B: 24 h, C: 48 h, and D: 72 h. Exposure durations to H2O2 include 0, 20, 40, and 60 min. Total colony-forming units (CFU) are represented by dark grey bars, while the count of spores is illustrated by light grey bars. Statistical significances were determined utilizing Tukey's test with a sample size of n = 3 and indicated by p-values: ∗ <0.05, ∗∗<0.01, ∗∗∗ <0.001.
However, the spore count remained nearly the same, while for 40 and 60 min treated biofilms, the spore quantity was similar to those of CFU total. Mature wt biofilms, cultivated for 48 and 72 h, exhibited efficient resistance to hydrogen peroxide treatment, as the cell quantity was unchanged throughout incubation time. The only distinction between these maturation stages lies in the higher spore count observed in the 72 h biofilms compared to those cultivated for 48 h.
The control inoculum of the eps deficient strain shows similar outcomes to those of the WT strain at 0 h and maintains a consistent cell count regardless of the exposure time (Fig. 5 0 h). Following biofilm formation, a mixture of vegetative cells and spores is present, with the spore count in this strain considerably lower than that in the WT strain, as observed in untreated samples. In early-stage biofilms (Fig. 524 h), the total CFU is roughly three times higher than the spore count at the 0-min mark.
Fig. 5.
The eps deficient biofilms' quantity treated with hydrogen peroxide was assessed at various maturation stages labeled as A: 0 h, B: 24 h, C: 48 h, and D: 72 h. These biofilms underwent exposure to H2O2 for durations of 0, 20, 40, and 60 min. Total CFU is depicted by dark grey bars, while the number of spores is shown by light grey bars. Statistical significance was evaluated through Tukey's test with a sample size of n = 3 and indicated by p-values: ∗ <0.05, ∗∗<0.01, ∗∗∗ <0.001.
However, for exposure durations between 20 and 60 min, the total CFU significantly declines, reaching levels comparable to those of spores.
The subsequent maturation stage, 48-h-old biofilms show a higher survival rate compared to those aged 24 h (Fig. 5 48 h). Nevertheless, a significant reduction in total CFU persists in comparison to the 0-min control. Additionally, there is a slight decrease in the spore count following a 60-min exposure to hydrogen peroxide. Biofilms grown for 72 h show increased susceptibility to hydrogen peroxide after a 20-min treatment compared to those exposed for 40 and 60 min (Fig. 5 72 h). Moreover, the total CFU count slightly increases with longer treatment durations, reaching its peak spore count at the 60-min mark.
The final strain tested for resistance to hydrogen peroxide lacked SigG, making it incapable of producing spores (Fig. 6). Planktonic cells from the stationary phase were used as inoculum (0 h, Fig. 6 0 h) to evaluate the survival ability by determining the total CFU. After 20 min of treatment, no CFU could be detected and this remained consistent for longer incubation periods. Accordingly, the inoculum of this strain can be regarded as a positive control. Interestingly, once consortia are formed, the cells showed an improved resilience against hydrogen peroxide. Biofilms aged from 24 to 72 h, showed similar results and were only slightly affected (Fig. 6 24 h–72 h). Exposure for 20 min resulted in minimal reduction in CFU, which was constant for longer incubation time.
Fig. 6.
Biofilms deficient in sigG were exposed to hydrogen peroxide and tested in survival across different maturation stages labeled as A: 0 h, B: 24 h, C: 48 h, and D: 72 h. Exposure durations to H2O2 ranged from 0 to 60 min. Statistical significance was analyzed using Tukey's test with a sample size of n = 3 and indicated by p-values: ∗ <0.05, ∗∗<0.01, ∗∗∗ <0.001.
4. Discussion
Hydrogen peroxide serves as a widely used commercial disinfectant, capable of targeting a broad spectrum of microbes, including spores. Some studies even report about its effectiveness against biofilms. However, there is a lack of available data regarding B. subtilis biofilms and the contributions of EPS and spores to resistance. Moreover, many studies overlook the impact of varying biofilm ages, which could be pivotal in understanding resistance mechanisms. Before the survival assay was conducted, the biofilms in maturation stages ranging from 0 to 72 h were cultivated and analyzed in their morphological phenotype as well as cell and spore quantity.
4.1. Comparison of the morphology and cell/spore quantity in wildtype biofilms versus EPS- and spore-lacking variants
The formation of architecturally complex structures as biofilms is attributed to a spatiotemporal cycle involving alternating phases of motile swarming and sessile matrix production [56,57]. This process results in the formation of concentric rings, as observed in wt biofilms, while being less abundant in eps-deficient and ΔsigG colony biofilms (Fig. 1, Fig. 2, Fig. 3 and [58]). Overall, biofilms lacking EPS show observable differences in texture and size. These consortia are impacted in their physical integrity and display a more homogenous appearance compared to wt. Although there are other matrix structures besides EPS, no wrinkles are visible. This effect strongly suggests that the formation of wrinkles is dependent on all matrix structures [59]. The vertical expansion, or the consolidation phase of biofilms is facilitated by matrix-producing cells, which are disrupted in the epsA-O-deficient strain, leading to the thin colony morphology [56]. Nevertheless, the preserved size in early-stage biofilms is maintained by swarming cells, which support two-dimensional expansion, or migration phase. Thus, the overexpression of the motile cell phenotype could compensate for the absence of EPS, thereby aiding in the growth of biofilms (57, Fig. 1 vs Fig. 3). The spatial arrangement of B. subtilis biofilms at specific time intervals prompts cell differentiation, including sporulation, at different stages and locations, culminating in characteristic colony morphology [60]. Sporulation seems to be associated with the development of complex architectural formations, as indicated by the diverse biofilm structure observed in our investigation. Furthermore, Aguilar et al. validated the correlation between sporulation and matrix production by the protein KinD [61]. Interestingly, Vlamakis et al. discovered that biofilms lacking spores due to sigF deletion do not undergo changes in biofilm structure but reduced spore quantity when matrix production is shut down [42,50].
In our study biofilms which lack EPS in the matrix show as well reduced levels of spores compared to wt (Fig. 2). The cell differentiation within biofilms is a highly regulated process, with matrix production and sporulation being connected through the activity of the bifunctional protein KinD. KinD mediates the phosphorylation (or dephosphorylation) of the master transcription factor Spo0A. As low quantities of phosphorylated Spo0A induces expression of matrix genes, EPS-deficient mutants exhibit delayed sporulation, leading to low spore count [61].
Comparing the overall cell numbers between the investigated strains, wt biofilms tend to an exponential increase in cell number, while the cell quantity of mutant strains reaches a plateau in mature stages. Alterations in nutrient storage and requirements resulting from a disrupted or altered matrix could influence cell and spore numbers, as well as morphology [59,62,63]In addition to the storage and transport, EPS contributes to quorum sensing (QS), which regulates the cell density and expansion of biofilms. The polysaccharides facilitate the stability of signal molecules necessary for QS, enhancing biofilm functionality and maintenance [64].
4.2. Contribution of extracellular polysaccharides to survival to hydrogen peroxide
The resistance of cells in a biofilm to a variety of disinfectants and antibiotics is attributed to the protective EM [65]. The matrix of B. subtilis biofilms is mainly composed of polysaccharides and proteins and organized in a mobile framework, interspersed with rigid aggregates of cells, Extracellular polysaccharides are more abundant in the mobile section, while protein fibers maintain rigidity [19,66]. Because of the multiple functions of EPS within the matrix, its protective capability against hydrogen peroxide was tested. The eps mutants are more susceptible to hydrogen peroxide than the wild type, especially in the early stages of maturation (24 h). Once a certain threshold of cell density is reached (at 72 h), survival is similar (Fig. 5B–D). Only spores (0 h) demonstrated a similar resistance and were not affected in terms of survival (Fig. 5). Interestingly, the resistance characteristics differ across the maturation stages tested. In young biofilms (24 h) only spores survived the hydrogen peroxide exposure, whereas mature biofilms did not exhibit the same resistance. Treated 48 h and 72 h biofilms show a higher number of total CFU than CFU of spores, indicating the survival of vegetative cells. Thus, apart from EPS, additional components within the matrix must contribute to the protection. The structural integrity of biofilms is crucial for surviving harsh environmental stressors, such as reactive oxygen species induced by hydrogen peroxide. Although the exact composition of the matrix varies depending on numerous factors and differs even among species, EPS and proteins are crucial for this integrity and are therefore highly abundant [19,67]. Besides the structural functionality, these compounds enhance the resistance to biocides. On one hand, the matrix acts as physical barrier against antimicrobial agents like hydrogen peroxide. On the other hand, they can react with them resulting in their depolymerization and thus disruption of aggressive hydroxyl radicals. In addition to polysaccharides, amyloid fibers formed by the TasA protein are known to contribute to the resistance [68]. Because of its characteristic beta-sheet structure, the interaction with antimicrobial agents that could lead to proteolysis is hindered, thereby TasA can be invoked in protection [[68], [69], [70], [71]]. Thus, a potential factor contributing to the survival of EPS-deficient (particularly mature) biofilms could be the presence of TasA fibers, but also the BslA. Branda et al. describes TasA and EPS as the most important and abundant structures in the biofilm matrix [37]. Further testing of a TasA mutant is necessary to shed more light on the role of amyloid fibers in hydrogen peroxide resistance. Overall, EPS are crucial for surviving oxidative stress, although they play a minor role in mature biofilms, as indicated by the slight reduction in cell quantity of biofilms. Hydrogen peroxide serves as a major source of reactive oxygen species (ROS) by generating hydroxyl radicals, which can initiate the depolymerization of EPS through the cleavage of glycosidic bonds [72]. However, our results indicate that survival rates in young biofilms are lower than in mature biofilms (Fig. 4). This finding suggests a correlation between biofilm survival and EPS quantity. A higher EPS content likely enhances resistance by acting as a diffusion barrier, limiting ROS penetration into the biofilm matrix. Additionally, certain molecules or compounds within the EPS may function as radical scavengers, further hindering ROS from reaching deeper layers. Following this hypothesis, ROS would be unable to penetrate the biofilm core, thereby protecting embedded cells. This concept is supported by a study conducted by Stewart et al., which demonstrated that hydrogen peroxide neither effectively penetrates P. aeruginosa biofilms nor inactivates them [73].
4.3. Role of sporulation in hydrogen peroxide resistance
Cell differentiation within B. subtilis biofilms is a crucial mechanism for adapting to dynamic environmental changes and stressors. This differentiation includes the formation of endospores, which allows the cells to persist under harsh conditions in a metabolically inactive (or reduced) state [74,75]. The transition into this dormant state is triggered by nutrient depletion and is accomplished by a range of different resistance mechanisms [76,77]. This is confirmed by the spore counts shown in Fig. 1 as biofilms mature and nutrient levels decrease. The survival of oxidative stress induced by hydrogen peroxide is ensured by enzymes such as catalases or superoxide dismutase localized in the spore coat [78]. For instance, spore-specific catalases like KatX are crucial for surviving hydrogen peroxide exposure during spore germination [79]. Additionally, we observed macroscopic differences in biofilm morphology appeared between sporulation-deficient populations and wt biofilms (Fig. 1 vs Fig. 3). This observation indicates that the differentiation into spores could contribute to the structural integrity and thus, to biofilm resilience against hydrogen peroxide [80,81]. Hu et al. investigated the resistance of spores and vegetative cells from biofilms of Clostridium perfringens to oxidative stress and confirmed that spores were more resistant than vegetative cells and the sessile lifestyle has an enhanced resilience [82]. However, numerous studies report the efficacy of hydrogen peroxide as sporicidal agent. Indeed, Sawale et al. determined D-values ranging from 0.08 to 0.95 min by using concentrations from 22 to 33 %. In our study, wt spores were not reduced after 60 min treatment, but the used concentration was more than ten times lower (Fig. 4 0 h). Using a similar concentration of H2O2, spores show decreased susceptibly and achieve “hardly any inactivation”,as confirmed by further studies [83,84]. Either increasing the incubation time or concentration of hydrogen peroxide could improve the sporicidal efficacy. Looking on multicellular lifestyle it was expected that based on the spore-specific protection mechanisms, spores would contribute to overall resistance to hydrogen peroxide. Interestingly, our results in turn, showed that the formation of spores did not contribute to the survival rate of biofilms (Fig. 6). While spore formation may not be the primary protective mechanism of the biofilms used in our study, spore formation contributes to protection of B. subtilis from hydrogen peroxide in planktonic cultures. The quantity of cells was regardless the maturation or incubation time not affected in survival which emphasizes the importance of an intact and functional biofilm matrix.
5. Conclusion
This study has shown that EPS in the matrix play a major role in the protection against hydrogen peroxide whereas sporulation does not. A functional structural integrity with intact EPS are even more protective than the ability of forming spores in surviving oxidative stress. Although our experiments clearly demonstrated the protective role of EPS against H2O2 treatment, these results can only be indirectly compared to those of the sporulation mutant. The comparison is influenced by the use of different inocula for biofilm initiation and the non-isogenic background of the strains. Furthermore, our results have shown that besides the EPS, especially in mature biofilms, additional protective structures remain which could be given by other matrix components, such as TasA fibers or the surface layer protein BslA. This research has revealed that EPS are crucial for surviving H2O2 exposure and need to be tackled. Newly developed sterilization approaches are often based on hydrogen peroxide and should be combined with additional sporicidal agents like UV or heat.
CRediT authorship contribution statement
Erika Muratov: Writing – original draft, Visualization, Methodology, Investigation, Formal analysis, Data curation, Conceptualization. Julian Keilholz: Methodology, Investigation, Formal analysis, Data curation. Ákos T. Kovács: Writing – review & editing. Ralf Moeller: Writing – review & editing, Supervision, Project administration, Funding acquisition, Conceptualization.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Given his role as Co-Editor in Chief, Ákos T. Kovács had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Birthe Kjellerup.
Acknowledgement
This paper is dedicated to the memory of Ralf Moeller. The authors express their appreciation to Prof. Dr. Jörg Stülke (University of Göttingen, Germany) and Prof. Dr. Madeleine Opitz (Ludwig-Maximilians-University München, Germany) for providing strains. Furthermore, we would like to thank Andrea Schröder for her help and support of this work.
This project was supported by the Deutsche Forschungsgemeinschaft (DFG) (MO 2023/3-1), and the German Aerospace Center (DLR), through Program Space - Research under Space Conditions, Partial Program 475, Project ISS LIFE 2.0 - Life in space: ISS and beyond to the Moon and Mars.
Data availability
Data will be made available on request.
References
- 1.Hernández-Jiménez E., del Campo R., Toledano V., Vallejo-Cremades M.T., Muñoz A., Largo C., Arnalich F., García-Rio F., Cubillos-Zapata C., López-Collazo E. Biofilm vs. planktonic bacterial mode of growth: which do human macrophages prefer? Biochem Biophys Res Commun. 2013;441:947–952. doi: 10.1016/j.bbrc.2013.11.012. [DOI] [PubMed] [Google Scholar]
- 2.Santos A., Milesi Galdino A.C., Mello T., Ramos L., Branquinha M., Bolognese A., Neto J., Roudbary M. What are the advantages of living in a community? A microbial biofilm perspective. Mem Inst Oswaldo Cruz. 2018;113 doi: 10.1590/0074-02760180212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Berlanga M., Guerrero R. Living together in biofilms: the microbial cell factory and its biotechnological implications. Microb Cell Fact. 2016;15:165. doi: 10.1186/s12934-016-0569-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.López D., Vlamakis H., Kolter R. Biofilms. Cold Spring Harbor Perspect Biol. 2010;2:a000398. doi: 10.1101/cshperspect.a000398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Watnick P., Kolter R. Biofilm, city of microbes. J Bacteriol. 2000;182:2675–2679. doi: 10.1128/jb.182.10.2675-2679.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Jamal M., Ahmad W., Andleeb S., Jalil F., Imran M., Nawaz M.A., Hussain T., Ali M., Rafiq M., Kamil M.A. Bacterial biofilm and associated infections. J Chin Med Assoc. 2018;Jan;81(1):7–11. doi: 10.1016/j.jcma.2017.07.012. [DOI] [PubMed] [Google Scholar]
- 7.Fleming D., Rumbaugh K. The consequences of biofilm dispersal on the host. Sci Rep. 2018;8 doi: 10.1038/s41598-018-29121-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cámara M., Green W., MacPhee C.E., Rakowska P.D., Raval R., Richardson M.C., Slater-Jefferies J., Steventon K., Webb J.S. Economic significance of biofilms: a multidisciplinary and cross-sectoral challenge. NPJ Biofilms Microbiomes. 2022;8:42. doi: 10.1038/s41522-022-00306-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Costerton J.W., Stewart P.S., Greenberg E.P. Bacterial biofilms: a common cause of persistent infections. Science. 1999;284:1318–1322. doi: 10.1126/science.284.5418.1318. [DOI] [PubMed] [Google Scholar]
- 10.Stewart P.S., Franklin M.J. Physiological heterogeneity in biofilms. Nat Rev Microbiol. 2008;6:199–210. doi: 10.1038/nrmicro1838. [DOI] [PubMed] [Google Scholar]
- 11.Spoering A.L., Lewis K. Biofilms and planktonic cells of Pseudomonas aeruginosa have similar resistance to killing by antimicrobials. J Bacteriol. 2001;183:6746–6751. doi: 10.1128/jb.183.23.6746-6751.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lewis K. Persister cells: molecular mechanisms related to antibiotic tolerance. Handb Exp Pharmacol. 2012:121–133. doi: 10.1007/978-3-642-28951-4_8. [DOI] [PubMed] [Google Scholar]
- 13.Brackman G., Coenye T. Quorum sensing inhibitors as anti-biofilm agents. Curr Pharm Des. 2015;21:5–11. doi: 10.2174/1381612820666140905114627. [DOI] [PubMed] [Google Scholar]
- 14.Kamaruzzaman N.F., Tan L.P., Mat Yazid K.A., Saeed S.I., Hamdan R.H., Choong S.S., Wong W.K., Chivu A., Gibson A.J. Targeting the bacterial protective armour; challenges and novel strategies in the treatment of microbial biofilm. Materials. 2018;11:1705. doi: 10.3390/ma11091705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.van Gestel J., Vlamakis H., Kolter R. Division of labor in biofilms: the ecology of cell differentiation. Microbiol Spectr. 2015;3(2) doi: 10.1128/microbiolspec.mb-0002-2014. Mb-0002-2014. [DOI] [PubMed] [Google Scholar]
- 16.Preda V.G., Săndulescu O. Communication is the key: biofilms, quorum sensing, formation and prevention. Discoveries. 2019;7:e100. doi: 10.15190/d.2019.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Claessen D., Rozen D.E., Kuipers O.P., Søgaard-Andersen L., Van Wezel G.P. Bacterial solutions to multicellularity: a tale of biofilms, filaments and fruiting bodies. Nat Rev Microbiol. 2014;12:115–124. doi: 10.1038/nrmicro3178. [DOI] [PubMed] [Google Scholar]
- 18.Hobley L., Harkins C., MacPhee C.E., Stanley-Wall N.R. Giving structure to the biofilm matrix: an overview of individual strategies and emerging common themes. FEMS Microbiol Rev. 2015;39:649–669. doi: 10.1093/femsre/fuv015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Branda S.S., Vik S., Friedman L., Kolter R. Biofilms: the matrix revisited. Trends Microbiol. 2005;13:20–26. doi: 10.1016/j.tim.2004.11.006. [DOI] [PubMed] [Google Scholar]
- 20.Mah T.-F.C., O'Toole G.A. Mechanisms of biofilm resistance to antimicrobial agents. Trends Microbiol. 2001;9:34–39. doi: 10.1016/s0966-842x(00)01913-2. [DOI] [PubMed] [Google Scholar]
- 21.Suci P.A., Mittelman M.W., Yu F.P., Geesey G.G. Investigation of ciprofloxacin penetration into Pseudomonas aeruginosa biofilms. Antimicrob Agents Chemother. 1994;38:2125–2133. doi: 10.1128/aac.38.9.2125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jiang Z., Nero T., Mukherjee S., Olson R., Yan J. Searching for the secret of stickiness: how biofilms adhere to surfaces. Front Microbiol. 2021;12 doi: 10.3389/fmicb.2021.686793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wang X., Liu M., Yu C., Li J., Zhou X. Biofilm formation: mechanistic insights and therapeutic targets. Mol Biomed. 2023;4:49. doi: 10.1186/s43556-023-00164-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bridier A., Briandet R., Thomas V., Dubois-Brissonnet F. Resistance of bacterial biofilms to disinfectants: a review. Biofouling. 2011;27:1017–1032. doi: 10.1080/08927014.2011.626899. [DOI] [PubMed] [Google Scholar]
- 25.Lineback C.B., Nkemngong C.A., Wu S.T., Li X., Teska P.J., Oliver H.F. Hydrogen peroxide and sodium hypochlorite disinfectants are more effective against Staphylococcus aureus and Pseudomonas aeruginosa biofilms than quaternary ammonium compounds. Antimicrob Resist Infect Control. 2018;7:154. doi: 10.1186/s13756-018-0447-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Quinn M.M., Henneberger P.K., Braun B., Delclos G.L., Fagan K., Huang V., Knaack J.L., Kusek L., Lee S.-J., Le Moual N. Cleaning and disinfecting environmental surfaces in health care: toward an integrated framework for infection and occupational illness prevention. Am J Infect Control. 2015;43:424–434. doi: 10.1016/j.ajic.2015.01.029. [DOI] [PubMed] [Google Scholar]
- 27.Omidbakhsh N., Sattar S.A. Broad-spectrum microbicidal activity, toxicologic assessment, and materials compatibility of a new generation of accelerated hydrogen peroxide-based environmental surface disinfectant. Am J Infect Control. 2006;34:251–257. doi: 10.1016/j.ajic.2005.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Masenga S., Kabwe L., Chakulya M., Kirabo A. Mechanisms of oxidative stress in metabolic syndrome. Int J Mol Sci. 2023;24:7898. doi: 10.3390/ijms24097898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Imlay J.A. The molecular mechanisms and physiological consequences of oxidative stress: lessons from a model bacterium. Nat Rev Microbiol. 2013;11:443–454. doi: 10.1038/nrmicro3032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rakotoarisoa M., Angelov B., Espinoza S., Khakurel K., Bizien T., Angelova A. Cubic liquid crystalline nanostructures involving catalase and curcumin: BioSAXS study and catalase peroxidatic function after cubosomal nanoparticle treatment of differentiated SH-SY5Y cells. Molecules. 2019;24:3058. doi: 10.3390/molecules24173058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Juan C.A., Pérez de la Lastra J.M., Plou F.J., Pérez-Lebeña E. The chemistry of reactive oxygen species (ROS) revisited: outlining their role in biological macromolecules (DNA, Lipids and Proteins) and induced pathologies. Int J Mol Sci. 2021;22:4642. doi: 10.3390/ijms22094642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Errington J., Aart L.T.V. Microbe profile: Bacillus subtilis: model organism for cellular development, and industrial workhorse. Microbiology (Read) 2020;166:425–427. doi: 10.1099/mic.0.000922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pribil W., Gehringer P., Eschweiler H., Cabaj A., Haider T., Sommer R. Assessment of Bacillus subtilis spores as a possible bioindicator for evaluation of the microbicidal efficacy of radiation processing of water. Water Environ Res. 2007;79:720–724. doi: 10.2175/106143007x175889. [DOI] [PubMed] [Google Scholar]
- 34.Hobley L., Ostrowski A., Rao F.V., Bromley K.M., Porter M., Prescott A.R., MacPhee C.E., van Aalten D.M.F., Stanley-Wall N.R. BslA is a self-assembling bacterial hydrophobin that coats the Bacillus subtilis biofilm. Proc Natl Acad Sci U S A. 2013;110:13600–13605. doi: 10.1073/pnas.1306390110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kobayashi K., Iwano M. BslA(YuaB) forms a hydrophobic layer on the surface of Bacillus subtilis biofilms. Mol Microbiol. 2012;85:51–66. doi: 10.1111/j.1365-2958.2012.08094.x. [DOI] [PubMed] [Google Scholar]
- 36.Vlamakis H., Chai Y., Beauregard P., Losick R., Kolter R. Sticking together: building a biofilm the Bacillus subtilis way. Nat Rev Microbiol. 2013;11:157–168. doi: 10.1038/nrmicro2960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Branda S.S., Chu F., Kearns D.B., Losick R., Kolter R. A major protein component of the Bacillus subtilis biofilm matrix. Mol Microbiol. 2006;59:1229–1238. doi: 10.1111/j.1365-2958.2005.05020.x. [DOI] [PubMed] [Google Scholar]
- 38.Branda S.S., González-Pastor J.E., Ben-Yehuda S., Losick R., Kolter R. Fruiting body formation by Bacillus subtilis. Proc Natl Acad Sci U S A. 2001;98:11621–11626. doi: 10.1073/pnas.191384198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kobayashi K. Bacillus subtilis pellicle formation proceeds through genetically defined morphological changes. J Bacteriol. 2007;189:4920–4931. doi: 10.1128/jb.00157-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kovács Á.T., Stanley-Wall N.R. Biofilm dispersal for spore release in Bacillus subtilis. J Bacteriol. 2021;203 doi: 10.1128/JB.00192-21. 10.1128/jb.00192-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Otto S.B., Martin M., Schäfer D., Hartmann R., Drescher K., Brix S., Dragoš A., Kovács Á.T. Privatization of biofilm matrix in structurally heterogeneous biofilms. mSystems. 2020;5 doi: 10.1128/msystems.00425-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Vlamakis H., Aguilar C., Losick R., Kolter R. Control of cell fate by the formation of an architecturally complex bacterial community. Genes Dev. 2008;22:945–953. doi: 10.1101/gad.1645008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.López D., Kolter R. Extracellular signals that define distinct and coexisting cell fates in Bacillus subtilis. FEMS Microbiol Rev. 2010;34:134–149. doi: 10.1111/j.1574-6976.2009.00199.x. [DOI] [PubMed] [Google Scholar]
- 44.Bridier A., Sanchez-Vizuete MdP., Le Coq D., Aymerich S., Meylheuc T., Maillard J.-Y., Thomas V., Dubois-Brissonnet F., Briandet R. Biofilms of a Bacillus subtilis hospital isolate protect Staphylococcus aureus from biocide action. PLoS One. 2012;7(9) doi: 10.1371/journal.pone.0044506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Epstein A.K., Pokroy B., Seminara A., Aizenberg J. Bacterial biofilm shows persistent resistance to liquid wetting and gas penetration. Proc Natl Acad Sci U S A. 2011;108:995–1000. doi: 10.1073/pnas.1011033108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Krawinkel J., Torres-Mapa M.L., Mhatre E., Kovács Á.T., Heisterkamp A. Structural damage of Bacillus subtilis biofilms using pulsed laser interaction with gold thin films. J Biophot. 2017;10:1043–1052. doi: 10.1002/jbio.201600146. [DOI] [PubMed] [Google Scholar]
- 47.Raie D.S., Mhatre E., El-Desouki D.S., Labena A., El-Ghannam G., Farahat L.A., Youssef T., Fritzsche W., Kovács Á.T. Effect of novel quercetin titanium dioxide-decorated multi-walled carbon nanotubes nanocomposite on Bacillus subtilis Biofilm Development. Materials. 2018;11(1):157. doi: 10.3390/ma11010157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Raie D.S., Mhatre E., Thiele M., Labena A., El-Ghannam G., Farahat L.A., Youssef T., Fritzsche W., Kovács Á.T. Application of quercetin and its bio-inspired nanoparticles as anti-adhesive agents against Bacillus subtilis attachment to surface. Mater Sci Eng C. 2017;70(Pt 1):753–762. doi: 10.1016/j.msec.2016.09.038. [DOI] [PubMed] [Google Scholar]
- 49.Schaeffer P., Millet J., Aubert J.P. Catabolic repression of bacterial sporulation. Proc Natl Acad Sci U S A. 1965;54:704–711. doi: 10.1073/pnas.54.3.704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Branda S.S., Chu F., Kearns D.B., Losick R., Kolter R. A major protein component of the Bacillus subtilis biofilm matrix. Mol Microbiol. 2006;59:1229–1238. doi: 10.1111/j.1365-2958.2005.05020.x. [DOI] [PubMed] [Google Scholar]
- 51.Fuchs F.M. Georg-August-University Göttingen; Germany: 2020. Bacillus subtilis biofilm formation under extreme terrestrial and simulated extraterrestrial conditions.https://ediss.uni-goettingen.de/handle/21.11130/00-1735-0000-0005-13A9-7 Doctoral dissertation. [Google Scholar]
- 52.Fuchs F.M., Driks A., Setlow P., Moeller R. An improved protocol for harvesting Bacillus subtilis colony biofilms. J Microbiol Methods. 2017;134:7–13. doi: 10.1016/j.mimet.2017.01.002. [DOI] [PubMed] [Google Scholar]
- 53.Freese E., Heinze J., Galliers E. Partial purine deprivation causes sporulation of Bacillus subtilis in the presence of excess ammonia, glucose and phosphate. J Gen Microbiol. 1979;115:193–205. doi: 10.1099/00221287-115-1-193. [DOI] [PubMed] [Google Scholar]
- 54.DeQueiroz G.A., Day D.F. Antimicrobial activity and effectiveness of a combination of sodium hypochlorite and hydrogen peroxide in killing and removing Pseudomonas aeruginosa biofilms from surfaces. J Appl Microbiol. 2007;103:794–802. doi: 10.1111/j.1365-2672.2007.03299.x. [DOI] [PubMed] [Google Scholar]
- 55.Tiwari S., Rajak S., Mondal D.P., Biswas D. Sodium hypochlorite is more effective than 70% ethanol against biofilms of clinical isolates of Staphylococcus aureus. Am J Infect Control. 2018;46:e37–e42. doi: 10.1016/j.ajic.2017.12.015. [DOI] [PubMed] [Google Scholar]
- 56.Tasaki S., Nakayama M., Shoji W. Morphologies of Bacillus subtilis communities responding to environmental variation. Dev Growth Differ. 2017;59:369–378. doi: 10.1111/dgd.12383. [DOI] [PubMed] [Google Scholar]
- 57.Srinivasan S., Vladescu I.D., Koehler S.A., Wang X., Mani M., Rubinstein S.M. Matrix production and sporulation in Bacillus subtilis biofilms localize to propagating wave fronts. Biophys J. 2018;114:1490–1498. doi: 10.1016/j.bpj.2018.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Wakita J-i, Shimada H., Itoh H., Matsuyama T., Matsushita M. Periodic colony formation by bacterial species Bacillus subtilis. J Phys Soc Jpn. 2001;70:911–919. doi: 10.1143/JPSJ.70.911. [DOI] [Google Scholar]
- 59.Azulay D.N., Spaeker O., Ghrayeb M., Wilsch-Bräuninger M., Scoppola E., Burghammer M., Zizak I., Bertinetti L., Politi Y., Chai L. Multiscale X-ray study of Bacillus subtilis biofilms reveals interlinked structural hierarchy and elemental heterogeneity. Proc Natl Acad Sci U S A. 2022;119 doi: 10.1073/pnas.2118107119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Huang Q., Zhang Z., Liu Q., Liu F., Liu Y., Zhang J., Wang G. SpoVG is an important regulator of sporulation and affects biofilm formation by regulating Spo0A transcription in Bacillus cereus 0–9. BMC Microbiol. 2021;21:172. doi: 10.1186/s12866-021-02239-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Aguilar C., Vlamakis H., Guzman A., Losick R., Kolter R. KinD is a checkpoint protein linking spore formation to extracellular-matrix production in Bacillus subtilis biofilms. mBio. 2010;1 doi: 10.1128/mbio.00035-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Zhang W., Seminara A., Suaris M., Brenner M.P., Weitz D.A., Angelini T. Nutrient depletion in Bacillus subtilis biofilms triggers matrix production. New J Phys. 2014;16 doi: 10.1088/1367-2630/ab76f7. [DOI] [Google Scholar]
- 63.Wilking J.N., Zaburdaev V., De Volder M., Losick R., Brenner M.P., Weitz D.A. Liquid transport facilitated by channels in Bacillus subtilis biofilms. Proc Natl Acad Sci U S A. 2013;110:848–852. doi: 10.1073/pnas.1216376110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Narla A.V., Borenstein D.B., Wingreen N.S. A biophysical limit for quorum sensing in biofilms. Proc Natl Acad Sci U S A. 2021;118 doi: 10.1073/pnas.2022818118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Anderson G., O'toole G. Innate and induced resistance mechanisms of bacterial biofilms. Curr Top Microbiol Immunol. 2008;322:85–105. doi: 10.1007/978-3-540-75418-3_5. [DOI] [PubMed] [Google Scholar]
- 66.Xue Y., Yu C., Ouyang H., Huang J., Kang X. Uncovering the molecular composition and architecture of the Bacillus subtilis biofilm via solid-state NMR spectroscopy. J Am Chem Soc. 2024;146:11906–11923. doi: 10.1021/jacs.4c00889. [DOI] [PubMed] [Google Scholar]
- 67.Ido N., Lybman A., Hayet S., Azulay D.N., Ghrayeb M., Liddawieh S., Chai L. Bacillus subtilis biofilms characterized as hydrogels. Insights on water uptake and water binding in biofilms. Soft Matter. 2020;16:6180–6190. doi: 10.1039/d0sm00581a. [DOI] [PubMed] [Google Scholar]
- 68.Romero D., Aguilar C., Losick R., Kolter R. Amyloid fibers provide structural integrity to Bacillus subtilis biofilms. Proc Natl Acad Sci U S A. 2010;107:2230–2234. doi: 10.1073/pnas.0910560107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ferrone F. Analysis of protein aggregation kinetics. Methods Enzymol. 1999;309:256–274. doi: 10.1016/s0076-6879(99)09019-9. [DOI] [PubMed] [Google Scholar]
- 70.Naiki H., Gejyo F. Kinetic analysis of amyloid fibril formation. Methods Enzymol. 1999;309:305–318. doi: 10.1016/s0076-6879(99)09022-9. [DOI] [PubMed] [Google Scholar]
- 71.Sunde M., Serpell L.C., Bartlam M., Fraser P.E., Pepys M.B., Blake C.C. Common core structure of amyloid fibrils by synchrotron X-ray diffraction. J Mol Biol. 1997;273:729–739. doi: 10.1006/jmbi.1997.1348. [DOI] [PubMed] [Google Scholar]
- 72.Qi H., Tang S., Bian B., Lai C., Chen Y., Ling Z., Yong Q. Effect of H2O2-VC degradation on structural characteristics and immunomodulatory activity of larch arabinogalactan. Front Bioeng Biotechnol. 2024;12 doi: 10.3389/fbioe.2024.1461343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Stewart P.S., Roe F., Rayner J., Elkins J.G., Lewandowski Z., Ochsner U.A., Hassett D.J. Effect of catalase on hydrogen peroxide penetration into Pseudomonas aeruginosa biofilms. Appl Environ Microbiol. 2000;66(2):836–838. doi: 10.1128/aem.66.2.836-838.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Qin Y., Angelini L.L., Chai Y. Bacillus subtilis cell differentiation, biofilm formation and environmental prevalence. Microorganisms. 2022;10:1108. doi: 10.3390/microorganisms10061108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Piggot P.J., Hilbert D.W. Sporulation of Bacillus subtilis. Curr Opin Microbiol. 2004;7:579–586. doi: 10.1016/j.mib.2004.10.001. [DOI] [PubMed] [Google Scholar]
- 76.Grossman A.D. Genetic networks controlling the initiation of sporulation and the development of genetic competence in Bacillus subtilis. Annu Rev Genet. 1995;29:477–508. doi: 10.1146/annurev.ge.29.120195.002401. [DOI] [PubMed] [Google Scholar]
- 77.Setlow P. Spores of Bacillus subtilis: their resistance to and killing by radiation, heat and chemicals. J Appl Microbiol. 2006;101:514–525. doi: 10.1111/j.1365-2672.2005.02736.x. [DOI] [PubMed] [Google Scholar]
- 78.Checinska A., Burbank M., Paszczynski A.J. Protection of Bacillus pumilus spores by catalases. Appl Environ Microbiol. 2012;78:6413–6422. doi: 10.1128/aem.01211-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Bagyan I., Casillas-Martinez L., Setlow P. The katX gene, which codes for the catalase in spores of Bacillus subtilis, is a forespore-specific gene controlled by sigmaF, and KatX is essential for hydrogen peroxide resistance of the germinating spore. J Bacteriol. 1998;180:2057–2062. doi: 10.1128/jb.180.8.2057-2062.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Lu X., Roe F., Jesaitis A., Lewandowski Z. Resistance of biofilms to the catalase inhibitor 3-amino-1,2,4-triazole. Biotechnol Bioeng. 1998;60 doi: 10.1002/(sici)1097-0290(19981005)60:1%3C135::aid-bit15%3E3.0.co;2-p. 135-135. [DOI] [PubMed] [Google Scholar]
- 81.Panmanee W., Gomez F., Witte D., Pancholi V., Britigan B.E., Hassett D.J. The peptidoglycan-associated lipoprotein OprL helps protect a Pseudomonas aeruginosa mutant devoid of the transactivator OxyR from hydrogen peroxide-mediated killing during planktonic and biofilm culture. J Bacteriol. 2008;190:3658–3669. doi: 10.1128/jb.00022-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Hu W.S., Woo D.U., Kang Y.J., Koo O.K. Biofilm and spore formation of Clostridium perfringens and its resistance to disinfectant and oxidative stress. Antibiotics (Basel) 2021;10(4):396. doi: 10.3390/antibiotics10040396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Zhang Y., Zhou L., Zhang Y., Tan C. Inactivation of Bacillus subtilis spores using various combinations of ultraviolet treatment with addition of hydrogen peroxide. Photochem Photobiol. 2014;90:609–614. doi: 10.1111/php.12210. [DOI] [PubMed] [Google Scholar]
- 84.Rezaee A., Kashi G., Jonidi jafari A., Khataee A., Nili-Ahmadabadi A. Effect of hydrogen peroxide on Bacillus subtilis spore removal in an electrophotocatalytic system. Fresenius Environ Bull. 2011;20:2750–2755. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data will be made available on request.