Abstract
Background
Agro-industrial byproducts offer promising solutions for reducing feed costs and mitigating environmental pollution. Efficient waste management through recycling and reuse not only minimizes environmental impact but also opens avenues for innovative animal feed products. The objectives of this research were to determine whether increasing the roughage-to-concentrate diet (R: C) ratio and adding citric waste fermented with yeast waste pellets (CWYWP) would improve gas production, digestibility, and fermentation outcomes. A 2 × 4 factorial experiment was conducted using a completely randomized design. The first factor comprised two levels of R: C ratios: 60:40 and 40:60. Rice straw was used as a roughage source. The second factor involves four levels of CWYWP supplementation (as a top-dressed substrate supplement) at 0%, 2%, 4%, and 6% dry matter (DM), respectively.
Results
A significant interaction between CWYWP level and R: C ratio was observed for cumulative gas production, with 2% and 4% CWYWP increasing gas volumes under the 40:60 ratio, peaking at 113.10 mL/0.5 g at 4% inclusion. Similarly, a significant interaction was found for in vitro digestibility at 24 h, where 4% CWYWP under 40:60 R: C significantly enhanced in vitro organic matter digestibility and in vitro dry matter digestibility compared to other levels. CWYWP supplementation elevated ammonia-nitrogen concentrations at both 2 and 4 h and slightly increased ruminal pH at 4% and 6% inclusion, with all pH values remaining within the physiological range. Volatile fatty acid profiles shifted toward higher propionate and lower acetate-to-propionate ratio, particularly at 6% CWYWP under 40:60 R: C, indicating improved glucogenic fermentation.
Conclusion
These findings suggest that CWYWP can be safely included at up to 4% in high-concentrate diets without impairing in vitro ruminal fermentation or digestibility.
Keywords: Industrial by-product, Yeast waste, Citric waste, Pellet diet, Rumen fermentation
Introduction
Ensuring the safety and adequacy of animal feed has become increasingly crucial, both in terms of quantity and quality. A lack of protein in livestock feed has been linked to poor animal performance [1]. High-quality protein sources, such as palm meal and soybean meal, are often expensive, leading to increased production costs in livestock systems [2]. Consequently, many researchers have investigated alternative protein sources to enhance the efficiency and productivity of livestock [3]. The use of agro-industrial byproducts in animal feed has gained global attention as a cost-effective and environmentally friendly approach to feed formulation [4]. Effective waste management through recycling and reusing agro-industrial residues can offer multiple benefits, including reduced environmental pollution, lower feed costs, and the development of novel feed ingredients [1].
Citric acid waste (CW) is a byproduct generated during citric acid production via the fermentation of substrates with Aspergillus niger [5]. With an annual output of over 1.7 million tons, citric acid is the second-largest fermentation product globally and a major organic acid in the food and feed industries. The production process yields up to 70% waste or byproduct, which is environmentally polluting and difficult to manage [6]. Citric acid waste contains cellulose, hemicellulose, and protein, making it a potential feed ingredient for ruminants [7]. However, Uriyapongson et al. [8] reported that CW inclusion in ruminant diets should be limited to 10% due to its high fiber content and low digestibility, which may impair feed intake and nutrient utilization. This might be because citric acid waste has a high fiber content and limited digestible nutrients, which can impair feed intake and nutrient utilization in ruminants. Therefore, nutritional enhancement through fermentation or supplementation with additives such as yeast is often necessary to increase its feeding value [9].
Global ethanol production exceeds 100 billion liters annually, with Saccharomyces cerevisiae being the most commonly used microbe in industrial fermentation [10]. During ethanol production, 10–12 L of liquid yeast waste (YW) may be generated per liter of ethanol. As a result, large volumes of yeast waste must be effectively recycled to reduce environmental impact. Yeast waste typically contains 60–70% live S. cerevisiae cells, which are rich in protein, minerals, vitamins, and prebiotics [11, 12].
The incorporation of citric acid waste (CW) and yeast waste (YW) into ruminant feed has shown promising results. Suriyapha et al. [1] developed a novel protein supplement by fermenting CW with YW. The resulting product, citric waste fermented with yeast waste (CWYW), contains a high crude protein content of 535 g/kg DM, making it a potential alternative to soybean meal. According to Suriyapha et al. [13], CWYW is also approximately 50% less expensive than soybean meal. Furthermore, previous studies by Suriyapha et al. [13] found that replacing 75% of soybean meal in a concentrate diet with CWYW had no adverse effects on tropical lactating cows or native beef cattle. Despite its nutritional benefits, CWYW in powder form poses challenges related to handling, storage, and feeding consistency, which can limit its practical application in ruminant feeding systems. One solution to improve its commercial viability is to process CWYW into pellet form, offering greater stability, ease of use, and nutrient uniformity.
Pelletized feeds are commonly used in ruminant diets because they promote uniform nutrient intake and increased feed efficiency [14]. Pellet production involves compressing ingredient mixtures into a dense and uniform form, which enhances storage, handling, and dietary consistency [15]. Pelletization also helps reduce feed waste, dust, and selective feeding, lowering the risk of diet-related disorders [16]. Moreover, feeding pellets supports precise rationing, ensuring animals receive adequate nutrients, especially those with higher nutritional demands [17]. Several studies have demonstrated that pellet forms of feed can increase nutrient digestibility and ruminal fermentation in ruminants [18].
Therefore, we hypothesized that CWYWP could serve as a strategic protein supplement to modulate rumen fermentation and digestion in ruminants. The objectives of this experiment were to demonstrate whether CWYWP supplementation utilized with different roughage to concentrate diet ratios influenced in vitro gas production, in vitro fermentation characteristics, and in vitro digestibility.
Results
Kinetics and cumulative gas production
Figure 1 shows the effect of CWYWP R: C ratio on cumulative gas production during 0 to 96 h of incubation. There were no observed interaction effects between the R: C ratio and levels of CWYWP in relation to several parameters, including final asymptotic gas volume (Vf), rate constant (k), discrete lag term (L), and cumulative gas production after 96 h of incubation (Table 1). However, the R: C ratio with the addition of CWYWP had interaction effects on cumulative gas production. There was a statistically significant interaction (p < 0.01) between CWYWP supplementation level and the R: C ratio on cumulative gas production at 96 h. Under the 40:60 R: C ratio, cumulative gas production increased progressively with 0%, 2%, and 4% CWYWP inclusion, reaching 109.95, 111.00, and 113.10 mL/0.5 g, respectively. However, further inclusion at 6% resulted in a slight reduction. In contrast, under the 60:40 R: C ratio, gas production remained lower overall and did not increase beyond 100.35 mL/0.5 g even at 6% CWYWP, suggesting a less favorable fermentation response under high-roughage conditions. However, the kinetics of the production of gas indicated no interaction between CWYWP levels and R: C ratio levels. No significant differences were observed in the primary factor of the R: C ratio for the final asymptotic gas volume (Vf), rate constant (k), and discrete lag term (L). Moreover, the 40:60 ratio has the impact of increasing a discrete lag time (h) by up to 14%. The levels of CWYWP did not affect the final asymptotic gas volume (Vf), rate constant (k), or discrete lag term (L).
Fig. 1.
Effect of citric waste fermented with yeast waste pellet (CWYWP) and roughage-to-concentrate ratio (R:C) ratio on cumulative gas during the 96 hours of incubation time (bars represent means ± SEM). T1 = 60:40 R:C ratio with 0% CWYWP, T2 = 60:40 with 2% CWYWP, T3 = 60:40 with 4% CWYWP, T4 = 60:40 with 6% CWYWP, T5 = 40:60 with 0% CWYWP, T6 = 40:60 with 2% CWYWP, T7 = 40:60 with 4% CWYWP, and T8 = 40:60 with 6% CWYWP
Table 1.
The combined impact of roughage to concentrate (R: C) ratio with the addition of citric waste fermented with yeast waste pellet (CWYWP) on gas kinetics and cumulative gas at 96 h after incubation
| R: C | CWYWP (%) | Gas Kinetics | Cumulative Gas at 96 h of Incubation (ml/0.5 g DM Substrate) | ||
|---|---|---|---|---|---|
| Vf | k | L | |||
| 60:40 | 0 | 107.95 | 0.05 | 1.10 | 108.50bc |
| 2 | 108.35 | 0.04 | 1.00 | 108.05bc | |
| 4 | 110.70 | 0.04 | 1.13 | 107.90bc | |
| 6 | 110.05 | 0.04 | 1.03 | 106.50c | |
| 40:60 | 0 | 109.00 | 0.04 | 1.25 | 109.95abc |
| 2 | 109.50 | 0.04 | 1.23 | 111.00ab | |
| 4 | 112.20 | 0.04 | 1.30 | 113.10a | |
| 6 | 104.20 | 0.04 | 1.37 | 93.45d | |
| SEM | 2.63 | 0.01 | 0.09 | 1.04 | |
| Comparison | |||||
| R: C ratio | 0.82 | 0.89 | 0.02 | 0.27 | |
| 60:40 | 109.26 | 0.04 | 1.12b | 107.74 | |
| 40:60 | 108.76 | 0.04 | 1.28a | 106.88 | |
| CWYWP (%) | 0.62 | 0.95 | 0.78 | < 0.01 | |
| 0 | 108.58 | 0.04 | 1.15 | 109.23a | |
| 2 | 108.92 | 0.04 | 1.20 | 109.53a | |
| 4 | 111.45 | 0.04 | 1.23 | 110.50a | |
| 6 | 107.12 | 0.04 | 1.24 | 99.98b | |
| Interaction | 0.62 | 0.51 | 0.78 | < 0.01 | |
a–d Value on the same row with different superscripts differ (p < 0.05); R: C = roughage to concentrate ratio. CWYWP = the supplementation of citric waste fermented with yeast waste pellet, Vf = final asymptotic gas volume corresponding to complete substrate digestion (mL/500 mg DM), k = a rate constant (units time − 1), L = a discrete lag term (h), SEM=standard error of the mean
In vitro digestibility
Table 2 presents the effects of R: C ratio and CWYWP supplementation on in vitro organic matter digestibility (IVOMD) and in vitro dry matter digestibility (IVDMD) at 12 h and 24 h of incubation. A statistically significant interaction was observed between the R: C ratio and CWYWP supplementation for both IVDMD and IVOMD at 24 h (p < 0.05). Under the 40:60 R: C ratio, 4% CWYWP supplementation significantly increased IVDMD and IVOMD compared to 0%, 2%, and 6% supplementation levels. However, no such effect was observed at the 60:40 R: C ratio, where digestibility did not significantly differ across CWYWP levels. This indicates that the increased digestibility with CWYWP occurred only under high-concentrate diets. At 12 h, there was no significant interaction between R: C ratio and CWYWP supplementation for either IVDMD or IVOMD. Nonetheless, main effects were observed. The 40:60 R: C ratio significantly increased IVDMD and IVOMD at 12 h compared to the 60:40 ratio (p < 0.01), with improvements ranging from 13.35 to 19.94%. CWYWP supplementation alone did not influence digestibility at 12 h.
Table 2.
The combined impact of roughage to concentrate (R: C) ratio with the addition of citric waste fermented with yeast waste pellet (CWYWP) on in vitro dry matter digestibility (IVDMD) and in vitro organic matter digestibility (IVOMD)
| R: C | CWYWP (%) | IVDMD (g/kg) | IVOMD (g/kg) | ||
|---|---|---|---|---|---|
| 12 h | 24 h | 12 h | 24 h | ||
| 0 | 387 | 429d | 405 | 497d | |
| 60:40 | 2 | 388 | 478c | 477 | 560c |
| 4 | 401 | 446cd | 488 | 528cd | |
| 6 | 387 | 458cd | 477 | 543cd | |
| 0 | 458 | 518b | 525 | 596b | |
| 40:60 | 2 | 478 | 530b | 549 | 607b |
| 4 | 473 | 582a | 533 | 615a | |
| 6 | 466 | 515b | 539 | 593b | |
| SEM | 7.60 | 11.22 | 19.64 | 10.29 | |
| Comparison | |||||
| R: C ratio | < 0.01 | < 0.01 | < 0.01 | < 0.01 | |
| 60:40 | 391b | 453b | 462b | 532b | |
| 40:60 | 469a | 536a | 537a | 603a | |
| CWYWP (%) | 0.31 | 0.03 | 0.14 | 0.04 | |
| 0 | 423 | 474c | 465 | 547b | |
| 2 | 433 | 504ab | 513 | 584a | |
| 4 | 437 | 514a | 511 | 572ab | |
| 6 | 427 | 487bc | 508 | 568ab | |
| Interaction | 0.63 | 0.02 | 0.18 | 0.04 | |
a–d Value on the same row with different superscripts differ (p < 0.05); R: C = roughage to concentrate ratio, CWYWP = the supplementation of citric waste fermented with yeast waste pellet, SEM=standard error of the mean
In vitro volatile fatty acid (VFA) concentration
Volatile fatty acid concentrations and proportions are summarized in Table 3. A significant interaction (p < 0.05) between the R: C ratio and CWYWP supplementation was observed for total VFA, butyric acid (C4) at both 2 and 4 h, and for acetic acid (C2), propionic acid (C3), and the C2:C3 ratio at 4 h of incubation. At 4 h, the highest total VFA concentrations were recorded under the 40:60 R: C ratio with 2% and 4% CWYWP supplementation (p < 0.01). Conversely, the lowest C4 concentrations at 4 h were also observed with the 40:60 R: C ratio at 2% and 4% CWYWP levels (p < 0.01). The C2:C3 ratio was significantly reduced when 6% CWYWP was combined with the 40:60 R: C ratio (p = 0.04), suggesting a shift toward propionate production under this condition. At 2 h, no significant interaction was found for C2, C3, or the C2:C3 ratio. However, main effects were noted. The concentration of C2 was unaffected by CWYWP level but was significantly higher in the 60:40 R: C group compared to 40:60 (p < 0.05). In contrast, C3 concentration at 2 h was significantly greater under the 40:60 R: C ratio than under 60:40, and diets supplemented with 4–6% CWYWP tended to have higher C3 concentrations than those with 0% or 2% CWYWP.
Table 3.
Effect of roughage to concentrate (R: C) ratio combined with the supplementation of citric waste fermented with yeast waste pellet (CWYWP) on in vitro volatile fatty acids (VFAs)
| R: C | CWYWP (%) | Total VFA (mmol/L) | C2 (mol/100 mol) | C3 (mol/100 mol) | C4 (mol/100 mol) | C2:C3 r atio | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 2 h | 4 h | 2 h | 4 h | 2 h | 4 h | 2 h | 4 h | 2 h | 4 h | ||
| 60:40 | 0 | 67.45c | 86.92c | 78.31 | 77.80a | 13.09 | 13.88d | 8.60bc | 8.32bc | 5.98 | 5.61a |
| 2 | 73.51ab | 88.76bc | 78.94 | 76.86ab | 13.07 | 14.50cd | 7.99cd | 8.64ab | 6.04 | 5.30ab | |
| 4 | 74.20ab | 92.49b | 78.49 | 77.77a | 13.65 | 14.20cd | 7.86d | 8.03c | 5.75 | 5.48ab | |
| 6 | 74.91ab | 91.88b | 78.13 | 77.51a | 13.49 | 14.37cd | 8.38bcd | 8.12c | 5.79 | 5.39ab | |
| 40:60 | 0 | 77.76a | 90.60bc | 78.00 | 76.00b | 13.39 | 15.63ab | 8.61ab | 8.37bc | 5.82 | 4.86cd |
| 2 | 78.60a | 99.55a | 78.02 | 77.70a | 13.31 | 14.57cd | 8.67ab | 7.74d | 5.86 | 5.33ab | |
| 4 | 74.64ab | 99.19a | 77.30 | 76.90ab | 13.75 | 15.13bc | 8.95a | 7.97cd | 5.62 | 5.08bc | |
| 6 | 72.02bc | 88.64bc | 76.21 | 74.77c | 14.86 | 16.39a | 8.93a | 8.84a | 5.13 | 4.56d | |
| SEM | 1.48 | 1.25 | 0.39 | 0.39 | 0.24 | 0.12 | 0.25 | 0.17 | 0.14 | 0.16 | |
| R: C | < 0.01 | < 0.01 | 0.04 | < 0.01 | < 0.01 | 0.93 | < 0.01 | 0.93 | < 0.01 | < 0.01 | |
| 60:40 | 72.43b | 90.01b | 78.45a | 77.48a | 13.37b | 14.24 | 8.18b | 8.28 | 5.87a | 5.44a | |
| 40:60 | 75.86a | 94.04a | 77.63b | 76.22b | 14.08a | 15.51 | 8.29a | 8.27 | 5.51b | 4.91b | |
| CWYWP (%) | 0.11 | < 0.01 | 0.02 | 0.04 | < 0.01 | < 0.01 | 0.62 | < 0.01 | 0.02 | 0.08 | |
| 0 | 72.61 | 88.76c | 78.15 | 76.90ab | 13.24b | 14.75b | 8.61 | 8.34a | 5.90a | 5.21 | |
| 2 | 76.57 | 93.08b | 78.43 | 77.19a | 13.24b | 14.53b | 8.33 | 8.28a | 5.92a | 5.31 | |
| 4 | 74.38 | 95.84a | 77.90 | 77.34a | 13.70ab | 14.66b | 8.40 | 8.00b | 5.68a | 5.28 | |
| 6 | 73.47 | 90.26c | 77.17 | 76.14b | 14.18a | 15.38a | 8.65 | 8.48a | 5.44b | 4.95 | |
| Interaction | < 0.01 | < 0.01 | 0.09 | < 0.01 | 0.22 | < 0.01 | 0.04 | < 0.01 | 0.12 | 0.04 | |
a–d Value on the same row with different superscripts differ (p < 0.05); R: C = roughage to concentrate ratio, CWYWP = the supplementation of citric waste fermented with yeast waste pellet, C2 = acetic acid, C3, propionic acid, C4 = butyric acid, SEM=standard error of the mean
Ruminal ammonia-nitrogen (NH3-N), pH, and protozoal population
The effects of R: C ratio and CWYWP supplementation on ruminal NH₃-N, pH, and protozoal population are summarized in Table 4. No significant interaction effects were detected for these parameters. However, significant main effects were observed. At both 2 h and 4 h, the 40:60 R: C ratio resulted in higher NH₃-N concentrations compared to the 60:40 group, with increases of approximately 37.67% and 14.45%, respectively (p < 0.01). CWYWP supplementation also significantly increased NH₃-N levels at both time points (p < 0.01), with the highest values recorded at 6% inclusion. Regarding ruminal pH, a modest but significant reduction was observed at 4 h under the 40:60 R: C ratio compared to 60:40 (p = 0.04). CWYWP supplementation at 4% and 6% slightly increased pH values at both 2 h and 4 h compared to lower levels (p < 0.01). Nevertheless, pH across all treatments remained within the physiological range (6.82–6.97), suggesting no risk of acidosis. No significant effects of R: C ratio or CWYWP supplementation were observed on protozoal populations, which remained statistically unchanged across all treatments and time points.
Table 4.
The effect of roughage to concentrate (R: C) ratio and citric fermented with yeast waste pellets (CWYWP) on ruminal ammonia-nitrogen (NH3-N), pH and protozoal population
| R: C | CWYWP (%) | NH3-N (mg/dL) | pH | Protozoal count (×105cell/mL) | |||
|---|---|---|---|---|---|---|---|
| 2 h | 4 h | 2 h | 4 h | 2 h | 4 h | ||
| 60:40 | 0 | 13.60 | 16.37 | 6.93 | 6.85 | 1.07 | 1.33 |
| 2 | 14.45 | 18.7 | 6.95 | 6.84 | 1.33 | 1.07 | |
| 4 | 14.68 | 19.11 | 6.94 | 6.93 | 1.07 | 2.93 | |
| 6 | 19.37 | 20.25 | 6.96 | 6.88 | 2.13 | 2.67 | |
| 40:60 | 0 | 19.51 | 20.38 | 6.91 | 6.82 | 1.6 | 1.07 |
| 2 | 20.82 | 20.86 | 6.90 | 6.84 | 2.13 | 3.73 | |
| 4 | 21.78 | 21.42 | 6.92 | 6.87 | 1.33 | 2.93 | |
| 6 | 23.41 | 22.54 | 6.97 | 6.86 | 1.87 | 2.93 | |
| SEM | 0.17 | 0.24 | 0.24 | 0.02 | 0.47 | 0.86 | |
| R: C | < 0.01 | < 0.01 | 0.06 | 0.04 | 0.33 | 0.29 | |
| 60:40 | 15.53b | 18.61b | 6.95 | 6.88a | 1.40 | 2.00 | |
| 40:60 | 21.38a | 21.30a | 6.93 | 6.85b | 1.73 | 2.67 | |
| CWYWP (%) | < 0.01 | < 0.01 | 0.02 | < 0.01 | 0.34 | 0.21 | |
| 0 | 16.56d | 18.38d | 6.92b | 6.84b | 1.34 | 1.20 | |
| 2 | 17.64c | 19.78c | 6.93b | 6.84b | 1.73 | 2.40 | |
| 4 | 18.23b | 20.27b | 6.93b | 6.90a | 1.20 | 2.93 | |
| 6 | 21.39a | 21.40a | 6.97a | 6.87ab | 2.00 | 2.80 | |
| Interaction | 1.00 | 1.00 | 0.29 | 0.42 | 0.71 | 0.33 | |
a–d Value on the same row with different superscripts differ (p < 0.05); R: C = roughage to concentrate ratio,. CWYWP = the supplementation of citric waste fermented with yeast waste pellet, SEM=standard error of the mean
Discussions
Chemical content in the diet
The citric waste used in this study contained 101 g/kg DM of CP, aligning closely with the value reported by Suriyapha et al. [1] at 110 g/kg DM. However, CP content in CW varies considerably across studies. For example, Tanpong et al. [19] reported a lower value of 61.1 g/kg DM, and de Farias Silva et al. [20] documented a broader range from 33.8 to 110.8 g/kg DM, likely reflecting differences in fermentation substrates, microbial strains, and industrial processing conditions [21]. These variations highlight the importance of standardizing raw materials to ensure consistent feed quality [1]. The YW used in this study had a CP content of 276 g/kg DM, which is within the previously reported range of 182.5–315 g/kg DM [1, 2, 22]. Differences in protein concentration are commonly influenced by the yeast strain, fermentation duration, and the nutrient profile of the ethanol production substrate [1]. When CW is co-fermented with YW, protein content typically increases. Suriyapha et al. [1] reported that CWYW in powder form reached 535 g/kg DM of CP, largely due to microbial biomass accumulation during fermentation. In addition, certain yeast strains are known to secrete fiber-degrading enzymes, such as cellulases, which can contribute to the breakdown of structural carbohydrates and improve overall feed digestibility [23].
In this study, the CWYWP contained 490 g/kg DM of CP (Table 5), slightly lower than the powder form reported by Suriyapha et al. [1]. This reduction may be attributed to either differences in the quality of raw inputs or losses during pelleting. Heat exposure during pelleting (typically 75–87 °C for 15–20 s) has been shown to induce Maillard reactions and protein denaturation, reducing nitrogen solubility and the measurable CP value [24]. Similar reductions in protein quality after thermal processing have been reported by Homan et al. [25] and Boney et al. [26]. As CWYWP was added at increasing levels (0–6%), CP concentration rose proportionally in all experimental diets. In the 60:40 R: C group, CP increased from 78 to 101 g/kg DM, while in the 40:60 group, it increased from 97 to 119 g/kg DM (Table 6). At the same time, NDF and ADF concentrations slightly declined. For instance, in the 60:40 group, NDF decreased from 473 to 466 g/kg DM and ADF from 362 to 356 g/kg DM as CWYWP levels increased. This reflects the relatively lower fiber content of CWYWP compared to the basal roughage components. However, despite the decrease in fiber concentration (g/kg DM), the total fiber mass per incubation bottle actually increased with higher CWYWP inclusion. This is because CWYWP was supplemented on top of the fixed 0.5 g substrate, increasing the total dry matter in the bottle. As a result, the absolute quantities of NDF and ADF (mg/bottle) increased across treatments. This dual effect—dilution in fiber percentage but increase in fiber load—may influence microbial colonization, fermentation efficiency, and fiber degradability. In addition to its protein contribution, CWYWP likely provided residual fermentable carbohydrates, peptides, and yeast-derived cofactors from molasses and microbial biomass [1, 15]. These components may have stimulated rumen microbial activity and fermentation, helping to explain the enhanced cumulative gas production and digestibility observed at 2% and 4% CWYWP inclusion.
Table 5.
Feed ingredients and chemical composition (g/kg DM) of concentrate diet, rice straw, yeast waste (YW), citric waste (CW) and citric waste fermented with yeast waste pellet (CWYWP) used in the in vitro study
| Items | Concentrate | Rice straw | YW | CW | CWYWP |
|---|---|---|---|---|---|
| Ingredients (g/kg dry matter; DM) | |||||
| Cassava chips | 500 | ||||
| Rice bran | 190 | ||||
| Soybean meal | 140 | ||||
| Palm kernel meal | 140 | ||||
| Premix | 10 | ||||
| Salt | 10 | ||||
| Urea | 10 | ||||
| Chemical composition | |||||
| Dry matter (g/kg) | 934 | 934 | 360 | 919 | 949 |
| Organic matter (g/kg DM) | 926 | 888 | 899 | 889 | 901 |
| Crude protein (g/kg DM) | 137 | 38 | 276 | 101 | 490 |
| Neutral detergent fiber (g/kg DM) | 189 | 662 | 217 | 719 | 357 |
| Acid detergent fiber (g/kg DM) | 118 | 465 | 44 | 416 | 253 |
| Gross energy (Mcal/kg DM) | 4.33 | 3.76 | 3.23 | 3.88 | 3.90 |
Premix = Vitamins and minerals; A: 10,000,000 IU; vitamin E 70,000 IU; vitamin D: 1,600,000 IU; Fe: 50 g; Zn: 40 g; Mn: 40 g; Co: 0.1 g; Se: 0.1 g; I: 0.5 g. YW = yeast waste; CW = citric waste; CWYWP = citric waste fermented with yeast waste pellet;
Table 6.
Chemical composition of substrate diets containing two roughage-to-concentrate (R: C) ratios and different level of citric waste fermented with yeast waste pellet (CWYWP) used in the in vitro study
| Items | Roughage-to-concentrate (R: C) ratios | |||||||
|---|---|---|---|---|---|---|---|---|
| 60:40 | 40:60 | |||||||
| Citric waste fermented with yeast waste pellet (CWYWP) (%) | ||||||||
| 0% | 2% | 4% | 6% | 0% | 2% | 4% | 6% | |
| Chemical composition | ||||||||
| Dry matter (DM; g/kg) | 934 | 934 | 935 | 935 | 934 | 934 | 935 | 935 |
| Organic matter (g/kg DM) | 903 | 903 | 903 | 903 | 911 | 911 | 911 | 910 |
| Crude protein (g/kg DM) | 78 | 86 | 94 | 101 | 97 | 105 | 112 | 119 |
| Neutral detergent fiber (g/kg DM) | 473 | 471 | 469 | 466 | 378 | 378 | 377 | 377 |
| Acid detergent fiber (g/kg DM) | 362 | 360 | 358 | 356 | 257 | 257 | 257 | 257 |
| Gross energy (Mcal/kg DM) | 3.99 | 3.99 | 3.99 | 3.98 | 4.10 | 4.10 | 4.09 | 4.09 |
Overall, the changes in dietary composition—both in nutrient proportions and total nutrient mass—are closely linked to the fermentation outcomes observed in this study. Accurate reporting of both the chemical composition and formulation dynamics is essential when evaluating CWYWP as a functional feed additive. Continued refinement of fermentation and pelleting processes will be important to maximize the nutritional value and consistency of this by-product in ruminant feeding systems [14, 17].
Kinetics and cumulative gas production
No significant interaction was observed between CWYWP levels and the R: C ratio for gas kinetics parameters, including final gas volume (Vf), rate constant (k), and discrete lag time (L). Therefore, the effects of each factor are interpreted independently. The R: C ratio had a significant effect on the lag time (L), with a longer lag phase observed at the 40:60 ratio compared to the 60:40 ratio. This finding suggests that diets with a higher proportion of concentrate may delay microbial fermentation onset due to the need for microbial adaptation. High-concentrate diets typically contain more fermentable carbohydrates, which can shift microbial colonization dynamics and slow the initial fermentation process [27–29].
In contrast, a significant interaction between CWYWP inclusion and the R: C ratio was detected for cumulative gas production at 96 h. Under the 40:60 R: C ratio—where fermentable substrates are more abundant—CWYWP supplementation at 2% and 4% led to increased cumulative gas production. This may be attributed to a synergistic effect between the nutrient-rich components in CWYWP (e.g., peptides, sugars, and nucleotides) and the higher concentrate content, which together support increased microbial activity in later fermentation stages [1, 11, 13]. However, at 6% CWYWP inclusion, cumulative gas production declined slightly. Although the NDF concentration decreased marginally (e.g., from 473 to 466 g/kg DM in the 60:40 diet), the total fiber mass introduced per bottle increased due to the additional dry matter from CWYWP. This increase in absolute fiber load may have imposed physical limitations on microbial access to fermentable substrates, thereby reducing fermentation efficiency [13]. This interpretation is supported by the measured composition data (Table 6), and highlights the importance of considering both concentration and total nutrient mass when evaluating fermentation responses. This pattern aligns with earlier reports indicating that excessive dietary fiber or structurally complex byproducts can reduce microbial fermentation efficiency, even when fermentable components are present [1, 29]. Under the 60:40 R: C ratio, cumulative gas production remained lower and showed limited response to CWYWP supplementation. This may reflect a constraint imposed by the higher fiber content of the basal diet, which could suppress microbial activity and diminish the potential benefits of CWYWP [13]. These results highlight the importance of both dietary composition and additive inclusion level when evaluating fermentation outcomes. The increased gas production under a 40:60 R: C ratio with 2–4% CWYWP inclusion suggests that moderate supplementation is optimal. This treatment showed increased CP (from 97 to 112 g/kg DM) while maintaining consistent NDF levels (~ 377 g/kg DM), indicating a favorable nutrient profile. These results suggest that this treatment has sufficient fermentable carbohydrates to support microbial growth, thereby enhancing fermentation efficiency (Table 6).
The differences between our findings and previous studies may be partially explained by feed processing methods. In the current study, CWYWP was provided in pellet form, which could have increased fermentation by improving substrate consistency, reducing particle size variability, and increasing surface area for microbial colonization. Moreover, heat and pressure during pelleting may have promoted starch gelatinization, thereby increasing substrate digestibility and gas production potential [29]. Although pelleting was not a variable under investigation, its influence on fermentation dynamics should not be overlooked.
Overall, the R: C ratio emerged as the primary factor influencing fermentation kinetics in this in vitro study. These findings reinforce the critical role of fiber-to-concentrate balance in modulating microbial fermentation behavior in the rumen. As noted by Elghandour et al. [30], high dietary fiber content tends to suppress gas production—consistent with the lower cumulative gas values observed at the 60:40 ratio. Optimizing this balance remains essential for improving rumen fermentation efficiency in practical ruminant feeding systems [31].
In vitro digestibility
This study demonstrated a significant interaction between the R: C ratio and CWYWP supplementation on IVDMD and IVOMD at 24 h. At the 40:60 R: C ratio, 4% CWYWP supplementation led to the highest IVDMD and IVOMD values, significantly outperforming 0%, 2%, and 6% inclusion levels. In contrast, no digestibility improvements were observed across CWYWP levels under the 60:40 R: C ratio. These results suggest that CWYWP is most effective at moderate inclusion levels in concentrate-rich diets, where microbial fermentation is likely more responsive to added fermentable substrates [1, 13].
The increased digestibility observed at 4% CWYWP inclusion is consistent with the reduction in NDF and ADF concentrations at this level (Table 6), which likely enhanced ruminal degradability by improving microbial access to structural carbohydrates. Lower fiber concentrations are known to facilitate more rapid fermentation and increase substrate availability in the rumen [28]. Additionally, CP content increased from 97 to 112 g/kg DM, which may have further supported microbial growth and enzymatic activity during fermentation. Although the total fiber mass also increased with CWYWP addition, the improved protein-to-fiber ratio at 4% inclusion likely contributed to more efficient microbial utilization of the substrate. These results suggest that the improved digestibility at this level is directly driven by shifts in the nutrient profile of the diet—specifically, increased crude protein and slightly reduced fiber concentration—supporting the role of CWYWP as a balanced nutritional enhancer. In addition to compositional effects, bioactive compounds from the yeast waste component may have contributed to the observed response at 4%. Previous studies have reported that YW can be rich in bioavailable nutrients such as nucleotides, peptides, amino acids, vitamins, and minerals, which support microbial metabolism and enzyme production [11]. Yeast-derived components such as β-glucans and mannan oligosaccharides (MOS) may also act as prebiotics that stimulate beneficial microbial populations. Dagaew et al. [11] showed that hydrolyzed YW enhanced rumen bacterial proliferation, while Sommai et al. [31] observed that yeast-fermented cassava pulp increased cellulolytic bacterial counts, potentially improving fiber digestion. Although the bioactive profile of YW was not directly measured in this study, the observed digestibility enhancement at 4% CWYWP inclusion is consistent with both the measured chemical composition and previously reported functional properties of YW.
However, no further improvement in digestibility was observed at 6% CWYWP inclusion, despite slightly lower NDF and ADF concentrations. This may be due to the increased total fiber load introduced into the system, which could physically hinder microbial colonization or limit enzyme access to fermentable substrates [26]. Moreover, fiber derived from citric waste may be less degradable, potentially offsetting the benefits of increased protein. It is also possible that nutrient synchrony was disrupted at this level—where the balance between fermentable carbohydrates and nitrogen became less optimal—thereby reducing microbial efficiency. Similar findings were reported by Suriyapha et al. [1], who noted that replacing soybean meal with CWYW increased digestibility only up to 75% inclusion, while complete replacement (100%) led to significant declines.
At 12 h, no interaction between CWYWP and R: C ratio was detected. However, the main effect of R: C ratio was significant, with the 40:60 diet producing higher digestibility than the 60:40 diet across all inclusion levels. This result likely reflects the greater availability of fermentable carbohydrates in high-concentrate diets, which accelerate early microbial colonization and activity [11, 13].
These findings underscore the potential of CWYWP, particularly at 4% inclusion in diets with higher concentrate levels, to increase in vitro digestibility. However, optimal inclusion thresholds should be observed to avoid excess indigestible fiber and preserve fermentation efficiency.
In vitro VFA concentration
The measured chemical composition of the diets (Table 6) helps explain several observed shifts in VFA concentration. The 40:60 R: C ratio combined with 2% or 4% CWYWP inclusion resulted in the highest total VFA (TVFA) at 4 h, which corresponds with the higher concentrate proportion and increased CP levels (from 97 to 112 g/kg DM) observed in those treatments. The relatively lower NDF concentration and improved protein-to-fiber ratio likely enhanced substrate degradability, supporting greater microbial activity and fermentation. As degradability increased, C3 concentrations rose while the C2:C3 ratio declined—an expected shift associated with increased availability of fermentable carbohydrates [29, 32].
CWYWP supplementation also altered VFA profiles under the 40:60 R: C ratio. At 4 h, the lowest C4 concentrations were observed at 2% and 4% CWYWP, possibly reflecting a metabolic shift favoring C3 over butyrate pathways. The most pronounced reduction in C2:C3 ratio occurred with 6% CWYWP, reinforcing this shift. These results are consistent with the increased CP and soluble components in the diet, which may have created favorable conditions for propionate-producing microbes. Although the bioactive compounds in CWYWP were not analyzed directly, previous studies provide additional support. Yeast-based additives have been reported to promote glucogenic fermentation pathways. Cherdthong et al. [2] and Gunun et al. [29] observed that yeast-derived substrates can increase propionate and reduce acetate concentrations in the rumen. Similarly, Phesatcha et al. [32] reported that high-concentrate diets elevate both total and individual VFA concentrations. Dagaew et al. [11] noted a 33.67% increase in C3 when the R: C ratio shifted from 30:70 to 70:30, while Suriyapha et al. [13] found a 10.49% increase in C3 with a 40:60 R: C ratio compared to 60:40. One possible explanation for the increased C3 is the promotion of Megasphaera elsdenii, a lactate-utilizing bacterium involved in the acrylate pathway. Although not measured here, prior reports suggest that yeast-derived peptides, β-glucans, and mannan oligosaccharides (MOS) may stimulate this population [30], contributing to enhanced propionate production observed in the 40:60 diets supplemented with 4–6% CWYWP.
At 2 h of incubation, C3 concentrations were significantly higher under the 40:60 R: C ratio and tended to increase with 4–6% CWYWP, though no interaction effect was detected. These early increases may reflect the immediate availability of fermentable carbohydrate in concentrate-rich diets, supported by the rapid stimulation of microbial activity. CWYWP may also supply small amounts of residual sugars or peptides, further supporting microbial fermentation during early stages. Despite reductions in C4 concentration under some treatments, this effect may be condition-specific. Soluble carbohydrates are known to promote butyrate under certain fermentation profiles, and yeast may stimulate Butyrivibrio spp., which produce butyrate and degrade hemicellulose [2, 3]. However, competition for substrates or nutrient imbalance may shift fermentation toward propionate, as observed here. A numerical reduction in TVFA at 2 h was found in the 60:40 R: C diet without CWYWP, likely reflecting lower fermentable substrate availability and delayed microbial activation. Although not statistically significant, this trend supports the idea that early-stage fermentation is sensitive to carbohydrate supply, especially under high-roughage, unsupplemented conditions.
Lastly, differences between the current findings and those of Suriyapha et al. [1], who found no significant effect of CWYW on VFA profiles, may be attributed to feed form. Their study used CWYW in powder form, whereas the present study used pelleted CWYWP, which underwent thermal conditioning (75–87 °C for 15–20 s). Pelleting may have improved physical structure, enhanced starch gelatinization, and disrupted fiber matrices—thereby improving microbial accessibility and fermentation efficiency [25, 26]. This may help explain the higher TVFA observed here compared to the unprocessed form used by Suriyapha et al. [1].
Ruminal NH3-N, pH, and protozoal population
The R: C ratio significantly influenced NH₃-N concentrations at both 2 and 4 h, with higher levels observed under the 40:60 ratio. This corresponds with the higher crude protein content in these diets (Table 6), which likely supplied more degradable nitrogen for microbial metabolism. The elevated NH₃-N also reflects greater microbial deamination activity in concentrate-rich diets, a response that aligns with previous reports by Suriyapha et al. [1] and Gunun et al. [29]. CWYWP supplementation further increased NH₃-N concentrations, which can be explained by its high protein content (490 g/kg DM) and the likely presence of non-protein nitrogen such as urea. As shown in our composition data, increasing CWYWP inclusion raised dietary CP levels—reaching up to 119 g/kg DM under the 40:60 R: C ratio. This higher nitrogen availability may have enhanced microbial protein turnover and ammonia release. These findings are consistent with the mechanisms proposed by Polyorach et al. [3], who reported that microbial degradation of yeast biomass and enzymatic hydrolysis of non-protein nitrogen rapidly increase ammonia in the rumen. Despite the rise in NH₃-N, ruminal pH values remained within the optimal physiological range (6.82–6.97) throughout incubation, indicating stable fermentation. A slight decrease in pH at 4 h under the 40:60 R: C ratio likely reflects increased acid production from rapid carbohydrate fermentation. Interestingly, CWYWP supplementation at 4% and 6% maintained or slightly elevated pH values at both time points. This may be due to the buffering effect of increased NH₃-N and possible prebiotic activity of mannan oligosaccharides (MOS) in the yeast cell wall. These compounds may support lactic acid-utilizing bacteria such as Megasphaera elsdenii and Selenomonas ruminantium [13], helping reduce acid accumulation and stabilize rumen pH.
Protozoal populations were not significantly affected by either the R: C ratio or CWYWP supplementation. This stability is likely due to the maintained pH environment and consistent availability of fermentable substrates and soluble sugars across treatments. These conditions support protozoal survival and activity and are consistent with the findings of Polyorach et al. [3], who reported unchanged protozoal counts with yeast-fermented cassava supplementation. Similar outcomes were also associated with diets that maintained stable fermentation parameters [18].
Conclusions and recommendations
This in vitro study demonstrated that CWYWP has potential as feed additive in ruminant diets. The results showed that a 40:60 roughage-to-concentrate (R: C) ratio increased in vitro feed digestibility, and that CWYWP inclusion—particularly at 2% and 4%—was associated with positive effects on ruminal fermentation parameters. However, no significant difference in gas production was observed between 4%, 2%, and 0% CWYWP under the 40:60 ratio, suggesting that the effect may be more related to the high concentrate level than the additive itself. Importantly, CWYWP did not negatively affect in vitro ruminal fermentation or digestibility at any inclusion level. Further research is needed to evaluate whether the positive effects of CWYWP on in vitro fermentation and digestibility are reproducible under in vivo conditions as feed additive, and whether they translate into improved animal performance and health.
Materials and methods
The method of producing citric waste fermented with yeast waste pellets (CWYWP)
One hundred liters of yeast waste (YW) derived from Saccharomyces cerevisiae, a byproduct of the ethanol industry, were collected from Mitr Phol Bio-Power Co., Ltd., in Chaiyaphum, Thailand. Additionally, 200 kg of citric waste (CW), a byproduct of the citric acid industry, were obtained from Sam Mor Farm in Udon Thani, Thailand. Molasses (M Molasses, Do Home Co., Ltd., Khon Kean, Thailand) and commercial-grade urea had been purchased from a local shop. The formulation of citric waste fermented with yeast waste pellets (CWYWP) was adapted from Suriyapha et al. [1]. First, 100 mL of YW were transferred to a flask (A). Then, in a flask (B), 20 g of molasses and 50 g of urea were mixed in 100 mL of distilled water and blended. Solutions of A and B were mixed in a 1:1 ratio to produce the YW media solution. This media was subsequently flushed with air for 16 h at room temperature using an electromagnetic air compressor (Model: SOBO MINI AC/DC, HEIYKUNGPLATU Co., Ltd., Khon Kaen, Thailand) to promote aerobic microbial activation. After flushing, formic acid (RED HORSE, KD RWMKASET Co., Ltd., Nong Khai, Thailand) was added to the YW media to adjust the pH to a range of 3.9–4.5. After 16 h of incubation, the prepared YW media solution and mixed with CW at a ratio of 1 mL YW per 1 g CW. The resulting mixture was packed into plastic bags (size 22 × 44 inches, PP Plastic Co., Ltd., Nakhon Ratchasima, Thailand) and vacuum sealed using a vacuum sealer (IMAFLEX 1400 W VS-921, Imarflex Industrial Co., Ltd., Bangkok, Thailand) to maintain anaerobic conditions. The sealed bags were stored at room temperature without oxygen for 14 days to allow for fermentation. Following anaerobic fermentation, the material was sun-dried for 48 h until the final product reached a minimum of 10% moisture, then prepared for subsequent use as CWYWP.
Design of experiments and dietary procedures
The study was conducted using a 2 × 4 factorial arrangement in a completely randomized design (CRD) with three replications. The first factor comprised two levels of R: C ratios: 60:40 and 40:60, using rice straw as the roughage source. The second factor involved four levels of CWYWP supplementation, applied as a top-dressed supplement at 0%, 2%, 4%, and 6% of the DM of the in vitro dietary substrate (0.5 g DM consisting of both concentrate and roughage). All substrate samples were oven-dried at 60 °C and ground through a 1-mm screen (Cyclotech Mill, Tecator, Sweden) for chemical composition analysis. The chemical analyses followed the AOAC [33] guidelines, focusing on the determination of dry matter (DM; ID 967.03) and ash content (ID 942.05). Crude protein (CP) was determined using a nitrogen analyzer (LECO Corporation, Saint Joseph, MI, USA). The neutral detergent fiber (NDF) and acid detergent fiber (ADF) contents were analyzed following the procedure of Van Soest et al. [34]. The gross energy (GE) contents of feeds were determined in a AC500 Isoperibol Calorimeter for Gross Calorific Content (LECO Corporation, USA). Table 5 demonstrated feed ingredients and chemical composition (g/kg DM) of concentrate diet, rice straw, YW, CW and CWYWP used in the in vitro study. The experimental concentrate diets contained 137 g/kg DM of CP. The CWYWP is composed of 590 g/kg DM of CP and 357 and 253 g/kg DM of NDF and ADF fibers, respectively. Furthermore, the ratios of R: C at 40:60 and 60:40 resulted in a CP content of 97 and 78 g/kg DM, respectively. Chemical composition of substrate diets containing two R: C ratios and different level of CWYWP used in the in vitro study are presented in Table 6. CP increased with CWYWP inclusion, from 78 to 101 g/kg DM in the 60:40 R: C group and from 97 to 119 g/kg DM in the 40:60 group. In contrast, NDF and ADF slightly decreased in the 60:40 group (473 to 466 and 362 to 356 g/kg DM, respectively), but remained unchanged in the 40:60 group. Gross energy (GE) was stable across treatments, ranging from 3.98 to 4.10 Mcal/kg DM.
Animal hosts and ruminal inoculum Preparation
Rumen fluid was collected from three healthy male Thai native beef cattle, each weighing 280 ± 15.0 kg and aged 1.5 years. The animals were sourced from the Beef and Buffalo Section, Department of Animal Science, Faculty of Agriculture, Khon Kaen University, Thailand. They were housed individually in pens with clean flooring and had free access to clean water and mineral licks. The animals were fed ad libitum rice straw and offered a concentrate diet containing 140 g/kg crude protein (CP) and 16.04 Mcal/kg of metabolizable energy at a rate of 1% of body weight (BW), administered twice daily at 07:00 and 16:00. After 15 days of dietary adaptation, approximately 2,000 mL of rumen fluid was collected from each animal via oral suction using a stomach tube. The collected rumen fluid was pooled, filtered through four layers of cheesecloth into an Erlenmeyer flask, and used as a composite inoculum for in vitro incubation. The samples were transported to the laboratory in pre-warmed thermos bottles maintained at 39 °C. Experimental feed samples were each weighed (0.5 g DM) and placed into 50-mL serum bottles. Each bottle was flushed with CO₂ for 15 s prior to inoculation. The bottles were separated into three sets: the first set of 27 bottles [(3 bottles (replication) × 8 treatments) + 3 bottles of blank was used to evaluate gas kinetics and total gas production (incubated for 96 h). The second set of 51 bottles [(3 bottles (replication) × 8 treatments × 2 observation times at 2 and 4 h) + 3 bottles of blank] was used for ruminal parameters including pH, ruminal NH3-N, volatile fatty acids, and protozoa. The third set of 51 bottles [(3 bottles (replication) × 8 treatments × 2 observation times at 12 and 24 h) + 3 blank bottles] was utilized to determine nutrient digestibility.
All bottles were sealed with aluminum and rubber caps and incubated at 39 °C for 96 h. Rumen-fluid medium was prepared following the method of Menke and Steingass [35] by mixing 1,095 mL of distilled water, 0.23 mL of micromineral solution, 365 mL of macromineral solution, 1 mL of resazurin solution, 60 mL of reducing solution, and 730 mL of buffer solution with 660 mL of pooled rumen liquor, under continuous CO₂ flushing. Each bottle was filled with 40 mL of buffered rumen fluid and was incubated in a hot air oven (Model: BOHA-318, Biolab Scientific Ltd., Ontario, Canada) at 39 °C without agitation.
Characteristics of in vitro production of gases and ruminal fermentation
Using the modified procedures of Cherdthong et al. [2], gas production was measured after 0, 0.5, 1, 2, 4, 6, 8, 12, 18, 24, 48, 72, and 96 h of incubation. For curve fitting and gas kinetics analysis, the Schofield [36] model was used:
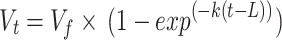 |
1 |
where Vt is the volume of gas at time t, Vf is the final asymptotic gas volume corresponding to complete substrate digestion (mL/g DM), t is the incubation time (h), k is a rate constant (units time − 1), and L is a discrete lag term (h).
Ruminal pH was measured using a digital pH meter (HANNA Instruments HI 8424 microcomputer, Singapore) at 0 and 4 h of incubation. The fermented ruminal fluid was divided into two portions. According to Fawcett and Scott [37], the first half (20 mL) was stored with 5 mL of 1 M H₂SO₄ and kept at − 20 °C for ammonia nitrogen (NH₃-N) analysis. The in vitro volatile fatty acid (VFA) concentration was analyzed by gas chromatography (Hewlett-Packard Co., Ltd., USA), equipped with a capillary column and flame ionization detector (Supelco, Inc., USA).
The remaining portion (1 mL) was preserved in 9 mL of 10% formalin solution for direct protozoal population counting [37]. Techniques by Tilley and Terry [38] were used to assess IVOMD and IVDMD after 12 and 24 h of incubation. The percentage of IVDMD was calculated using the following equation:
 |
2 |
The difference between dried and ignited weights was taken as the indigestible organic matter, and the results were calculated according to the equation:
 |
3 |
Statistical analysis
The data were statistically analyzed using a 2 × 4 factorial arrangement in a completely randomized design (CRD), utilizing PROC GLM in SAS software [39]. The following statistical model was applied:
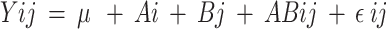 |
4 |
where: Y = observations; µ = overall mean;  = effect of factor A (the level of R: C ratio at 60:40 and 40:60; i = 1 to 2);
= effect of factor A (the level of R: C ratio at 60:40 and 40:60; i = 1 to 2); 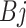 = effect of factor B (The supplementation of CWYWP at 0, 2, 4 and 6%; j = 1 to 4),
= effect of factor B (The supplementation of CWYWP at 0, 2, 4 and 6%; j = 1 to 4), 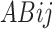 = the interaction effect of R: C ratio and CWYWP, and
= the interaction effect of R: C ratio and CWYWP, and 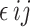 = the residual effect. Duncan’s New Multiple Range Test (DMRT) was used to compare treatment means [40]. A 95% confidence level was applied to determine significant differences.
= the residual effect. Duncan’s New Multiple Range Test (DMRT) was used to compare treatment means [40]. A 95% confidence level was applied to determine significant differences.
Acknowledgements
The authors express their sincerest gratitude to the Tropical Feed Resource Research and Development Center (TROFREC), Department of Animal Science, Faculty of Agriculture, Khon Kaen University for the use of facilities and providing raw materials.
Abbreviations
- ADF
Acid detergent fiber
- BW
Body weight
- C2
Acetic acid
- C3
Propionic acid
- C4
Butyric acid
- CF
Crude fiber
- CP
Crude protein
- DM
Dry matter
- EE
Ether extract
- KKU
Khon Kaen University
- N
Nitrogen
- NDF
Neutral detergent fiber
- NH3-N
Ammonia-nitrogen
- NRC
National Research Council
- OM
Organic matter
- TVFAs
Total volatile fatty acids
Author contributions
Planning and design of the study, N.K., R.P., C.S., and A.C.; conducting and sampling, N.K and A.C.; sample analysis, N.K. and A.C.; statistical analysis, N.K., A.C., S.W. and C.S.; manuscript drafting, N.K., A.C., S.W., R.P., C.S. and C.S.; manuscript editing and finalizing, N.K., A.C., S.W., R.P., C.S., A.L., R.P., P.C. and C.S.; All authors have read and agreed to the published version of the manuscript.
Funding
The authors express their sincerest gratitude to the Fundamental Fund of Khon Kaen University, which has received funding support from the National Science, Research, and Innovation Fund (NSRF). Research Program on the Research and Development of Winged Bean Root Utilization as Ruminant Feed, Increase Production Efficiency, and Meat Quality of Native Beef and Buffalo Research Group and Research and Graduate Studies, Khon Kaen University (KKU), were also acknowledged. Natdanai Kanakai received a grant from the Research Fund for Supporting Lecturers to Admit High Potential Students to Study and Research on His Expert Program from the Graduate School, KKU.
Data availability
The dataset produced and/or examined during the present study is not accessible to the public as it forms an initial component of another study. Nevertheless, interested parties can obtain the data from the corresponding author upon making a reasonable request.
Declarations
Ethics approval and consent to participate
The Institutional Animal Care and Use Committee of Khon Kaen University (KKU) approved the experimental procedures for using animals in the metabolism experiments (Record No. IACUC-KKU-80/66). The animals were obtained from Beef and buffalo Section, Department of Animal Science, Faculty of Agriculture, Khon Kaen University, Thailand.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Suriyapha C, Cherdthong A, Suntara C, Polyorach S. Utilization of yeast waste fermented citric waste as a protein source to replace soybean meal and various roughage to concentrate ratios on in vitro rumen fermentation, gas kinetic, and feed digestion. Fermentation. 2021;7:120. 10.3390/fermentation7030120. [Google Scholar]
- 2.Cherdthong A, Prachumchai R, Supapong C, Khonkhaeng B, Wanapat M, Foiklang S, Milintawisamai N, Gunun N, Gunun P, Chanjula P. Inclusion of yeast waste as a protein source to replace soybean meal in concentrate mixture on ruminal fermentation and gas kinetics using in vitro gas production technique. Anim Prod Sci. 2018;59:1682–8. 10.1071/AN18491. [Google Scholar]
- 3.Polyorach S, Wanapat M, Cherdthong A. Influence of yeast fermented cassava chip protein (YEFECAP) and roughage to concentrate ratio on ruminal fermentation and microorganisms using in vitro gas production technique. Asian-Australas J Anim Sci. 2014;27:36–45. 10.5713/ajas.2013.13298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Salam SA, Luciano G, O’Grady MN, Biondi L, Newbold CJ, Kerry JP, Priolo A. Sustainability of feeding plant by-products: A review of the implications for ruminant meat production. Anim Feed Sci Technol. 2019;251:37–55. 10.1016/j.anifeedsci.2019.02.006. [Google Scholar]
- 5.Roukas T, Kotzekidou P. Pomegranate. Peel waste: a new substrate for citric acid production by Aspergillus Niger in solid-state fermentation under non-aseptic conditions. Environ Sci Pollut Res. 2020;27:13105–13. 10.1007/s11356-020-07928-9 [DOI] [PubMed] [Google Scholar]
- 6.Börekçi BS. Evaluating wastes as low-cost substrates for use in microbial citric acid production. in green chemistry for the development of eco-friendly products; IGI Global: 2022; 43–76. https://doi.org/0.4018/978-1-7998-9851-1.ch003.
- 7.Tanpong S, Wongtangtintharn S, Cherdthong A, Prachumchai R, Tengjaroenkul B, Chanjula P, Suntara C, Wachirapakorn C. Recycling of citric acid waste for potential use as animal feed through fermentation with lactic acid bacteria and a mixture of fibrolytic enzymes. Animals. 2022;12:3049. 10.3390/ani12213049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Uriyapongson S, Wacharapakorn C, Navanukraw C, Phoemchalard C, Panatuk J, Toburan W. Digestibility and performance of Buffalo fed total mixed ration with different levels of citric waste. Buffalo Bull. 2013;32:829–33. [Google Scholar]
- 9.Reynolds CA, Morrison SY. Effects of Kefir fermented with or without 1% autolyzed yeast powder on dry matter intake, intestinal permeability, and rumen fermentation profile of Holstein calves. J Dairy Sci. 2025. 10.3168/jds.2024-26190. [DOI] [PubMed] [Google Scholar]
- 10.da Silva Fernandes F, de Souza ÉS, Carneiro LM, Alves Silva JP, de Souza JVB, da Silva Batista J. Current ethanol production requirements for the yeast Saccharomyces cerevisiae. Int J Microbiol. 2022;2022(7878830). 10.1155/2022/7878830 [DOI] [PMC free article] [PubMed]
- 11.Dagaew G, Cherdthong A, Wongtangtintharn S, Wanapat M, Suntara C. Manipulation of in vitro ruminal fermentation and feed digestibility as influenced by yeast waste-treated cassava pulp substitute soybean meal and different roughage to concentrate ratio. Fermentation. 2021;7:196. 10.3390/fermentation7030196. [Google Scholar]
- 12.van Dyk J, Görgens JF, van Rensburg E. Valorisation of industrial waste for second-generation ethanol production: towards the scale-up of high-solids paper sludge fermentation. Waste Biomass Valor. 2025. 10.1007/s12649-025-02962-5. [Google Scholar]
- 13.Suriyapha C, Supapong C, So S, Wanapat M, Cherdthong A. Bioconversion of agro-industrial residues as a protein source supplementation for multiparous Holstein Thai crossbreed cows. PLoS ONE. 2022;17:e0273916. 10.1371/journal.pone.0273916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Papandrea SF, Cataldo MF, Palma A, Gallucci F, Zimbalatti G, Proto AR. Pelletization of compost from different mixtures with the addition of exhausted extinguishing powders. Agronomy. 2021;11:1357. 10.3390/agronomy11071357. [Google Scholar]
- 15.Aguado-Giménez F. Effect of feed delivery rate and pellet size on rearing performance, feed wastage and economic profitability in Gilthead seabream (Sparus aurata) on growing. Water. 2020;12:954. 10.3390/w12040954. [Google Scholar]
- 16.Paulk CB, Stark CR, Dunmire KM. Feed Processing Technology and Quality of Feed. Sustainable swine nutrition. In: Chiba LI, editor. Sustainable swine nutrition. 2nd ed. Hoboken: Willey Blackwell; 2022. pp. 429–44. 10.1002/9781119583998.ch16. [Google Scholar]
- 17.Prachumchai R, Cherdthong A, Wanapat M, Supapong C, Khonkhaeng B, Suntara C, Foiklang S. Effect of feeding a pellet diet containing high sulphur with fresh cassava root supplementation on feed use efficiency, ruminal characteristics and blood metabolites in Thai native beef cattle. J Anim Physiol Anim Nutr. 2021;105:653–63. 10.1111/jpn.13514. [DOI] [PubMed] [Google Scholar]
- 18.Phesatcha B, Phesatcha K, Wanapat M. Mitragyna speciosa Korth leaf pellet supplementation on feed intake, nutrient digestibility, rumen fermentation, microbial protein synthesis and protozoal population in Thai native beef cattle. Animals. 2022;12:3238. 10.3390/ani12233238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tanpong S, Cherdthong A, Tengjaroenkul B, Tengjaroenkul U, Wongtangtintharn S. Evaluation of physical and chemical properties of citric acid industrial waste. Trop Anim Health Prod. 2019;51:2167–74. 10.1007/s11250-019-01917-y. [DOI] [PubMed] [Google Scholar]
- 20.de Farias Silva CE, Gois GNSB, da Silva LMO, Almeida RMRG, de Souza Abud AK. Citric waste saccharification under different chemical treatments. Acta Sci Technol. 2015;37:387–95. 10.4025/actascitechnol.v37i4.28133. [Google Scholar]
- 21.Armghan Khalid M, Niaz B, Saeed F, Afzaal M, Islam F, Hussai M, Mahwish Muhammad SKH, Siddeeg A, Al-Farga A. Edible coatings for enhancing safety and quality attributes of fresh produce: A comprehensive review. Int J Food Prop. 2022;25:1817–47. 10.1080/10942912.2022.2107005. [Google Scholar]
- 22.Raza M, Yousaf M, Rashid M, Maqsood A, Azam B, Mustafa R, Ahmad S, Khalid A, Naseem M, Rehman H. Yeast cell wall supplementation modulates pre-weaning stress in male Nili Ravi Buffalo calves. S Afr J Anim Sci. 2022;52:460–71. 10.4314/sajas.v52i4.06. [Google Scholar]
- 23.Suntara C, Cherdthong A, Uriyapongson S, Wanapat M, Chanjula P. Novel Crabtree negative yeast from rumen fluids can improve rumen fermentation and milk quality. Sci Rep. 2021;11:6236. 10.1038/s41598-021-85643-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Croes C-A, Briceno Noriega D, Wichers H, Savelkoul HFJ, Ruinemans-Koerts J, Teodorowicz M. Characterization of different stages of Maillard reaction in soy: impact on physicochemical properties and immunogenicity of soy proteins. Food Funct. 2025;16:2577–88. 10.1039/D4FO04400B. [DOI] [PubMed] [Google Scholar]
- 25.Homan KA, Gupta N, Kroll KT, Kolesky DB, Skylar-Scott M, Miyoshi T, Mau D, Valerius MT, Ferrante T, Bonventre JV. Flow-enhanced vascularization and maturation of kidney organoids in vitro. Nat Methods. 2019;16:255–62. 10.1038/s41592-019-0325-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Boney J, Moritz J. The effects of Spirulina algae inclusion and conditioning temperature on feed manufacture, pellet quality, and true amino acid digestibility. Anim Feed Sci Technol. 2017;224:20–9. 10.1016/j.anifeedsci.2016.11.008. [Google Scholar]
- 27.Ma H, Dong A, Xu Y, Wu Q, Lambo MT, Zhang Y, Dou X, Li Y. Regulatory effects of high concentrate diet synergistically fermented with cellulase and lactic acid bacteria: in vitro ruminal fermentation, methane production, and rumen Microbiome. Anim Feed Sci Technol. 2025;319:116194. 10.1016/j.anifeedsci.2024.116194. [Google Scholar]
- 28.Zhang X, Liu X, Xie K, Pan Y, Liu F, Hou F. Effects of different fiber levels of energy feeds on rumen fermentation and the microbial community structure of grazing sheep. BMC Microbiol. 2025;25:180. 10.1186/s12866-024-03644-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gunun N, Ouppamong T, Khejornsart P, Cherdthong A, Wanapat M, Polyorach S, Kaewpila C, Kang S, Gunun P. Effects of rubber seed kernel fermented with yeast on feed utilization, rumen fermentation and microbial protein synthesis in dairy heifers. Fermentation. 2022;8:288. 10.3390/fermentation8060288. [Google Scholar]
- 30.Elghandour M, Vázquez J, Salem A, Kholif A, Cipriano M, Camacho L, Márquez O. In vitro gas and methane production of two mixed rations influenced by three different cultures of Saccharomyces cerevisiae. J Appl Anim Res. 2017;45:389–95. 10.1080/09712119.2016.1204304 [Google Scholar]
- 31.Sommai S, Ampapon T, Mapato C, Totakul P, Viennasay B, Matra M, Wanapat M. Replacing soybean meal with yeast-fermented cassava pulp (YFCP) on feed intake, nutrient digestibilities, rumen microorganism, fermentation, and N-balance in Thai native beef cattle. Trop Anim Health Prod. 2020;52:2035–41. 10.1007/s11250-020-02228-3. [DOI] [PubMed] [Google Scholar]
- 32.Phesatcha K, Phesatcha B, Wanapat M, Cherdthong A. Roughage to concentrate ratio and Saccharomyces cerevisiae inclusion could modulate feed digestion and in vitro ruminal fermentation. Vet Sci. 2020;7:151. 10.3390/vetsci7040151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Association of Official Analytical Chemists (AOAC). Official methods of analysis. Volume 2, 16th ed. Arlington, VA, USA. 1998. [Google Scholar]
- 34.Van Soest PV, Robertson JB, Lewis BA. Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J Dairy Sci. 1991;74:3583–97. 10.3168/jds.S0022-0302(91)78551-2. [DOI] [PubMed] [Google Scholar]
- 35.Menke KH, Steingass H. Estimation of the energetic feed value obtained from chemical analysis and gas production using rumen fluid. Anim Res Develop. 1988;28:7–55. [Google Scholar]
- 36.Schofield P. Gas production methods. Farm Animal Metabolism and Nutrition. Editor, D’Mello JPF. CABI Publishing. 2000;209–32.
- 37.Galyean M. Laboratory procedure in animal nutrition research. Department of Animal and Life Science. USA: New Mexico State University; 1989. p. 188. [Google Scholar]
- 38.Tilley J, Terry DR. A two-stage technique for the in vitro digestion of forage crops. Grass Forage Sci. 1963;18:104–11. 10.1111/j.1365-2494.1963.tb00335.x. [Google Scholar]
- 39.Statistical Analysis System (SAS). User’s Guide: Statistic, Version 9.3; SAS Institute, Inc.: Cary, NC, USA. 2013.
- 40.Steel GD, Torrie JH. Principles and Procedures of Statistics, a Biometrical Approach; McGraw-Hill Kogakusha, Ltd. 1980. 10.1002/bimj.19620040313
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The dataset produced and/or examined during the present study is not accessible to the public as it forms an initial component of another study. Nevertheless, interested parties can obtain the data from the corresponding author upon making a reasonable request.



