Abstract
Rationale
In chronic obstructive pulmonary disease (COPD), impaired left ventricular (LV) filling might be associated with coexisting heart failure with preserved ejection fraction (HFpEF) or due to reduced pulmonary venous return indicated by small LV size.
Objectives
We investigated the all-cause mortality associated with small LV or HFpEF and clinical features discriminating between both patterns of impaired LV filling in patients with COPD.
Methods
We performed transthoracic echocardiography (TTE) in patients with stable COPD from the COSYCONET (COPD and Systemic Consequences and Comorbidities Network) cohort to define small LV as LV end-diastolic diameter below the normal range and HFpEF features according to recommendations of the European Society of Cardiology. We assessed the ratio of early to late ventricular filling velocity (E/A), ratio of early mitral inflow velocity to annular early diastolic velocity (E/e′), serum N-terminal pro-brain natriuretic peptide, high-sensitivity troponin I, airflow limitation (FEV1), lung hyperinflation (residual volume), and gas transfer capacity (DlCO) and discriminated patients with small LV from those with HFpEF features or no relevant cardiac dysfunction as per TTE (normalTTE). The primary outcome was all-cause mortality after 4.5 years.
Measurements and Main Results
In 1,752 patients with COPD, the frequency of small LV, HFpEF features, and normalTTE was 8%, 16%, and 45%, respectively. Patients with small LV or HFpEF features had higher all-cause mortality rates than patients with normalTTE: hazard ratio, 2.75 (95% confidence interval, 1.54–4.89) and 2.16 (95% confidence interval, 1.30–3.61), respectively. Small LV remained an independent predictor of all-cause mortality after adjusting for confounders including exacerbation frequency and measures of residual lung volume, DlCO, or FEV1. Compared with normalTTE, patients with small LV had reduced LV filling, as indicated by lowered E/A. Yet, in contrast to patients with HFpEF features, patients with small LV had normal LV filling pressure (E/e′) and lower concentrations of N-terminal pro-brain natriuretic peptide and high-sensitivity troponin I.
Conclusions
In COPD, both small LV and HFpEF features are associated with increased all-cause mortality and represent two distinct patterns of impaired LV filling.
Clinical trial registered with www.clinicaltrials.gov (NCT01245933).
Keywords: ventricular underfilling, heart failure with preserved ejection fraction, COPD, lung hyperinflation, emphysema
At a Glance Commentary
Scientific Knowledge on the Subject
In chronic obstructive pulmonary disease, impaired left ventricular (LV) filling might be associated with either coexisting heart failure with preserved ejection fraction or reduced pulmonary venous return, indicated by a smaller LV size. Differentiating between these two patterns is crucial, as their precise impact on mortality remains unclear and warrants further investigation.
What This Study Adds to the Field
Cardiac biomarkers and echocardiographic parameters help differentiate between disturbed LV filling from reduced venous return and coexisting heart failure with preserved ejection fraction in chronic obstructive pulmonary disease. Both patterns signal higher all-cause mortality but represent distinct, treatable traits.
Chronic obstructive pulmonary disease (COPD) and cardiovascular disease are among the leading causes of global mortality and disability (1). Both diseases coexist frequently, which confers poor disease outcomes and worsens survival (2, 3). A well-established instance is the interaction between COPD and cardiac dysfunction, namely heart failure, as both conditions share risk factors and clinical features, such as aging, acute hospitalization, and disease progression (4). Although heart failure with preserved ejection fraction (HFpEF) is believed to be a frequent cooccurring cardiac condition in COPD (5), left ventricular dysfunction might also arise as a disease-related cardiac manifestation in patients with COPD and hyperinflation. Imaging studies have indicated a close association of lung hyperinflation or emphysema with the reduction of end-diastolic left ventricular volume and the ensuing decline in cardiac output (6, 7). Likewise, airflow limitation and lung hyperinflation were linked to decreased left ventricular filling and cardiac chamber size, measured by echocardiography, in patients with COPD (8, 9). A further disease-related aspect is the reduction in pulmonary blood flow of emphysematous lung regions (10, 11) and the subsequent left ventricular underfilling (12). Reduced left ventricular filling in patients with COPD and hyperinflation is a treatable trait, as lung deflation with either pharmacological or bronchoscopic lung volume reduction improves cardiac function indicated by an increase in the left ventricle (LV) size (13–15).
We have previously reported that left ventricular diastolic dysfunction in patients with COPD is associated with exercise intolerance and physical inactivity, independent from lung function impairment (9, 16). However, data investigating the mortality risk in patients with COPD and impaired ventricular filling, either due to HFpEF or to COPD-related reduction of left ventricular filling, are still lacking. Furthermore, the identification of clinical features that distinguish HFpEF from COPD-related reduction of left ventricular filling is important and highly relevant for patient management (17).
Therefore, we aimed to investigate the mortality rate in patients with COPD-related reduction of left ventricular filling, indicated by small LV size, or in those with coexisting HFpEF compared with patients with COPD and normal cardiac function in a well-characterized COPD cohort. In addition, we sought to elucidate clinical features, echocardiographic measures, and cardiac biomarkers that distinguish COPD-related reduction of left ventricular filling from diastolic dysfunction associated with coexistent HFpEF. Some of the results of these studies have been previously reported in the form of an abstract (18).
Methods
Study Design
This analysis involves patients from the multicenter COSYCONET (COPD and Systemic Consequences and Comorbidities Network), a prospective longitudinal observational cohort study of patients with COPD. Patients were recruited at 31 German study centers, primarily after referrals from respiratory specialists (19). The main aim of the COSYCONET cohort is to investigate the impact of extrapulmonary disease manifestations, particularly cardiovascular comorbidities, on disease outcomes and all-cause mortality. The inclusion and exclusion criteria, as well as detailed study aims and methods, have been previously described (19). The study was approved by the ethics committee at the University of Marburg and is registered at ClinicalTrials.gov (NCT01245933). A written informed consent was obtained before enrollment.
Assessment of Lung Physiology, Cardiac Biomarkers, Anthropometric Measures, and Computed Tomography Imaging
Lung function testing, including spirometry, body plethysmography, and single-breath carbon monoxide lung uptake, was done as previously described (19). Furthermore, we measured serum levels of N-terminal pro-brain natriuretic peptide (NT-proBNP) (MILLIPLEX; Merck Millipore) and high-sensitivity troponin I (hs-troponin I) (ARCHITECT STAT; Abbott Diagnostics), as previously described (20).
We used a whole-body bioelectrical impedance analysis (Nutribox analyzer; Pöcking, Germany) to calculate the skeletal muscle mass according to the equation provided by Janssen and colleagues (21) and the muscle mass index by dividing it by the body surface area. Pulmonary computed tomography (CT) was available for a subgroup of patients, and low-attenuation areas (LAA) were calculated as previously described (22).
Echocardiography and Stratification of the Study Population
We performed two-dimensional and Doppler echocardiography as recommended by the American Society of Echocardiography and the European Association of Cardiovascular Imaging (23, 24). Briefly, the main measures of echocardiography were the left ventricular ejection fraction, left atrial diameter, left ventricular end-diastolic diameter (LVEDD), left ventricular mass index, left ventricular posterior wall diameter, interventricular septal diameter, tricuspid annular plane systolic excursion (TAPSE), the ratio between early mitral inflow velocity and mitral annular early diastolic velocity (E/e′), and the ratio between early and late ventricular filling velocity (E/A). The evaluation of right ventricular function relied on the assessment of experienced examiners and was reported as dichotomous variable. Additional details on measures of echocardiography are previously described (8, 20, 25) and provided in the online supplement.
The stratification of the study groups and possibly confounding groups was performed in the following order: First, we identified heart failure with mid-range or reduced ejection fraction in patients with left ventricular ejection fraction <50% (26). Then, we defined right ventricular dysfunction (RVD) based on three criteria: TAPSE <17 mm, moderate to severe tricuspid value regurgitation, or impaired right ventricular function based on visual inspection (27, 28). Next, we defined HFpEF based on current guidelines (29). In detail, we identified patients who had objective structural features consistent with left ventricular diastolic dysfunction underlying HFpEF (23, 29) as follows: 1) left ventricular hypertrophy defined by moderate to severely increased left ventricular mass index, posterior wall, or interventricular septal thickness; or 2) moderate to severely increased left atrial diameter (27). In this subgroup of patients with structural features consistent with left ventricular dysfunction, we stratified those who also had NT-proBNP concentrations >220 pg/ml as having HFpEF features (29); otherwise, those who had NT-proBNP levels ⩽ 220 pg/ml were stratified as having HFpEF risk. In addition to the aforementioned structural features, atrial fibrillation was also a criterion to stratify patients as HFpEF features (29). To avoid misclassifying patients with reduced venous return as HFpEF in the primary analysis, we did not consider parameters of diastolic dysfunction in stratifying the patients to HFpEF as recommended (23). Therefore, for the purpose of sensitivity analyses, we identified a subgroup of patients with more frank HFpEF, which included patients with HFpEF features who also had an E/e′ > 9. Furthermore, in patients who did not have any of the previously indicated cardiac dysfunction patterns, we stratified patients who had LVEDD below the sex-specific normal range as small LV (i.e., LVEDD <42 mm in males and <39 mm in females) (23). Last, we stratified patients who did not demonstrate any of the above-indicated abnormalities as normal echocardiography (normalTTE). Patients with moderate to severe left valvular heart disease were excluded from normalTTE and small LV, as this might affect the left ventricular filling. For the same reason, patients with small left atrium, without small LV, were excluded from normalTTE, as this might indicate early reduced LV filling. Details on measures and cutoff values of echocardiography used in patient stratification are provided in the online supplement. According to the aims of this analysis, the study population comprised patients with HFpEF features, small LV, and normalTTE.
Statistical Analyses
To determine the statistical significance of the observed differences among the clinical variables between the study groups, we performed a one-way ANOVA, Kruskal-Wallis, or Fisher’s exact test. For pairwise comparisons, post hoc analyses were performed using either Tukey’s test or Dunn’s test. To test for statistical dependence between two continuous variables, we used Pearson’s test or, for skewed variables, Spearman’s rank test.
Survival status during follow-up was ascertained as previously described (20). The primary outcome of this report was all-cause mortality after 4.5 years of follow-up. Accordingly, we used mixed-effects Cox models to demonstrate differences in survival time among the study groups. Independent predictors were cardiac dysfunction patterns of HFpEF features or small LV versus normalTTE with further stepwise adjustment for age, sex, body mass index, smoking status, frequency of exacerbations, and one of the following lung function parameters: FEV1, residual lung volume (RV), or DlCO. The random intercept accounts for clustering by study centers. Hazard ratios demonstrate the relative rate of death in patients with either HFpEF features or small LV compared with patients with normalTTE. Kaplan-Meier curves with the corresponding log-rank test were used to visualize survival probability between the study groups. Statistical analyses were performed using R (version 2022.12.0, RStudio; R Foundation). An α error of <5% was considered statistically significant.
Results
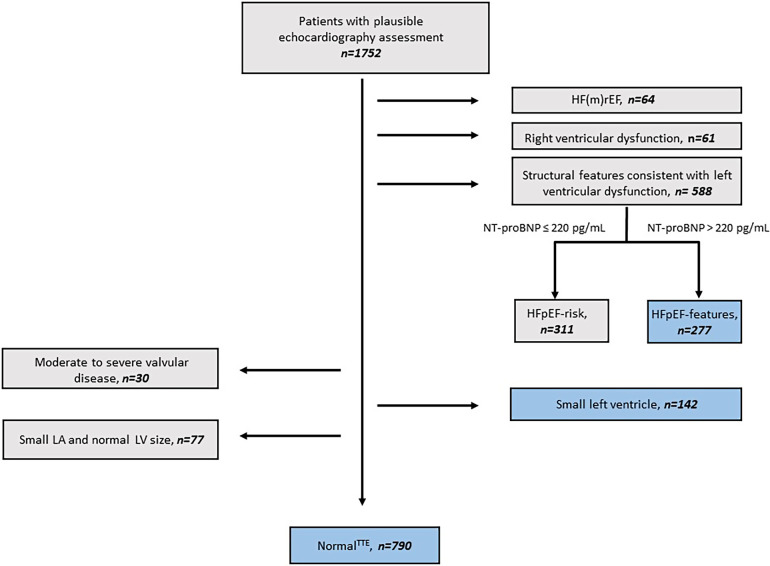
In 1,752 eligible patients, we first identified heart failure with mid-range or reduced ejection fraction and RVD in 3.6% and 3.5% of the patients, respectively (Figure 1). Then, we identified patients who demonstrated morphological features consistent with HFpEF and stratified those who also had elevated NT-proBNP as having HFpEF features (16%) and those with normal NT-proBNP as HFpEF risk (17%) (Figure 1). Furthermore, in patients who did not fulfill the criteria of the indicated cardiac dysfunction patterns, we identified patients with small LV, who represented 8% of the cohort population (Figure 1). Subsequently, patients with HFpEF features, small LV, and those who had normal echocardiography, as per the study definition, comprised the study population of this analysis (n = 1,209).
Figure 1.
Study selection flowchart. The whole cohort: N = 1,752; patients with HF(m)rEF: n = 64; heart failure with preserved ejection fraction (HFpEF) features: n = 277; HFpEF risk: n = 311; right ventricular dysfunction: n = 61; small LV: n = 142. HF(m)rEF = heart failure with mid-range reduced ejection fraction; LA = left atrium; LV = left ventricle; normalTTE = no cardiac dysfunction as per transthoracic echocardiography; NT-proBNP = N-terminal pro-brain natriuretic peptide.
Patient demographics and pulmonary and cardiac characteristics according to study groups are shown in Table 1. Compared with patients with small LV or normalTTE, patients with HFpEF features were older and had more frequent cardiovascular comorbidities. Furthermore, patients with small LV or HFpEF features had reduced exercise tolerance. In addition, patients with small LV had significantly lower body mass index, particularly lower skeletal muscle mass, than patients with HFpEF features or normalTTE.
Table 1.
Baseline Clinical Characteristics of the Patients
| NormalTTE (n = 790) | HFpEF Features (n = 277) | Small LV (n = 142) | P Value | |
|---|---|---|---|---|
| Demographics | ||||
| Sex, % males | 55 | 51 | 54 | 0.67 |
| Age, yr | 63 ± 8 | 68 ± 8 | 64 ± 8 | <0.001*† |
| Body mass index, kg/m2 | 26.7 ± 4.8 | 27.0 ± 5 | 24.4 ± 5 | <0.001*‡ |
| Muscle mass, kg | 26.7 ± 7.4 | 26.4 ± 7.0 | 24.8 ± 7.6 | 0.019*‡ |
| Muscle mass index, kg/m2 | 17.6 (10.6–30.8) | 16.0 (9.4–30.4) | 12.4 (7.5–25.6) | 0.005*‡ |
| Current smokers, % | 25 | 28 | 22 | 0.48 |
| Pulmonary characteristics | ||||
| Severe COPD,§ % | 35 | 34 | 57 | <0.001 |
| FEV1, % | 60 ± 21 | 59 ± 20 | 51 ± 22 | <0.001*‡ |
| RV, % | 163 ± 51 | 157 ± 51 | 183 ± 59 | <0.001*‡ |
| DlCO, % | 60 ± 22 | 60 ± 21 | 52 ± 21 | 0.002*‡ |
| CT-LAA, % | 16 (5.8–26.4) | 12.1 (3.1–14.5) | 30 (14–35.5) | 0.017*‡ |
| Exacerbation rate,‖ % | 32 | 34 | 42 | 0.094 |
| 6-min-walk distance, m | 438 ± 101 | 406 ± 105 | 411 ± 100 | <0.001†‡ |
| mMRC dyspnea scale :2, % | 40 | 45 | 48 | 0.15 |
| Cardiac characteristics | ||||
| Cardiovascular comorbidities, % | ||||
| CAD | 12 | 27 | 9 | <0.001 |
| Hypertension | 52 | 63 | 48 | 0.003 |
| Diabetes mellitus | 11 | 18 | 8 | 0.004 |
| Hyperlipidemia | 40 | 45 | 36 | 0.24 |
| NT-proBNP, pg/ml | 158 (27–335) | 439 (314–694) | 223 (51–371) | <0.001*† |
| hs-troponin I, ng/L | 3.6 (2.2–6.4) | 4.0 (2.6–7.4) | 3.3 (2.2–4.8) | 0.002*†‡ |
| E/A ratio | 0.89 (0.73–1.1) | 0.83 (0.71–1.05) | 0.81 (0.70–1.0) | 0.021‡ |
| E/e′ ratio | 7.8 (6.4–9.4) | 9.1 (6.9–11.0) | 7.7 (5.7–9.7) | <0.01*† |
| e′septal, cm/s | 7.6 (6.2–9.1) | 7.3 (5.9–9.0) | 8.1 (6.3–10.0) | 0.037* |
| e′lateral, cm/s | 9.8 (8.0–11.7) | 9.0 (7.5–11.2) | 9.1 (7.9–12.0) | 0.15 |
| a′septal, cm/s | 10.7 (9.2–12.0) | 9.5 (8.0–11.6) | 11.0 (9.2–13) | <0.001*† |
| a′lateral, cm/s | 11.6 (9.3–14.0) | 11.0 (8.6–13.0) | 13.0 (10.0–15.0) | <0.01*† |
| LV mass index | 92 (78–105) | 135 (113–153) | 66 (55–79) | <0.001*†‡ |
| TAPSE, cm | 2.41 ± 0.39 | 2.34 ± 0.5 | 2.28 ± 0.37 | <0.001†‡ |
| Left atrial diameter, cm | 3.5 ± 0.44 | 3.9 ± 0.84 | 3.1 ± 0.54 | <0.001*†‡ |
| ACE inhibitors, % | 22 | 26 | 22 | 0.51 |
| β blockers, % | 17 | 40 | 11 | <0.01*† |
| Statins, % | 18 | 26 | 14 | <0.01*† |
Definition of abbreviations: a′ = late diastolic velocity obtained by tissue doppler imaging; ACE = angiotensin-converting enzyme; CAD = coronary artery disease; CT-LAA = lung low attenuation area per computed tomography; e′ = mitral annular early diastolic velocity; E/A = the ratio between early and late ventricular filling velocity; E/e′ = ratio between early mitral inflow velocity and mitral annular early diastolic velocity; HFpEF = heart failure with preserved ejection fraction; LV = left ventricle; mMRC = modified Medical Research Council; NT-proBNP = N-terminal pro-brain natriuretic peptide; RV = residual lung volume; TAPSE = tricuspid annular plane systolic excursion; TTE = transthoracic echocardiography.
Values are presented as mean ± SD or median (interquartile range). CT-LAA was available for n = 84, n = 25, and n = 14 patients with normalTTE, HFpEF, and small LV, respectively. E/e′ was available for n = 699, n = 169, and n = 85 patients with normalTTE, HFpEF, and small LV, respectively. E/A was available for n = 906, n = 221, and n = 135 patients with normalTTE, HFpEF, and small LV, respectively.
Post hoc analysis P < 0.05 between small LV versus HFpEF.
Post hoc analysis P < 0.05 between HFpEF versus normalTTE.
Post hoc analysis P < 0.05 between small LV versus normalTTE.
Severe COPD indicates patients with Global Initiative for Chronic Obstructive Lung Disease stages III–IV.
Moderate-severe exacerbation within the previous 12 months to baseline study visit.
Lung Function in Patients with Small LV
A larger proportion of patients with small LV had severe COPD (Table 1). Accordingly, patients with small LV had more severe lung hyperinflation, as indicated by significantly higher residual lung volume, lower DlCO, and larger attenuation areas on pulmonary CT than patients with HFpEF-features or normalTTE (Table 1).
Diastolic Function and Cardiac Biomarkers in Patients with Small LV or HFpEF Features
We observed differences regarding echocardiographic functional parameters between the groups. Although patients with HFpEF features had higher left ventricular end-diastolic pressure, indicated by a higher E/e′ ratio (median, 9.1; interquartile range [IQR], 6.9–11.0) than patients with small LV (median, 7.7; IQR, 5.7–9.7) or normalTTE (median, 7.8; IQR, 6.4–9.4; P values < 0.001), the E/e′ ratio was not increased in patients with small LV compared with those with normalTTE (P = 0.45) (Table 1). Likewise, serum levels of both NT-proBNP and hs-troponin I were not higher in patients with small LV compared with normalTTE but rather significantly higher in patients with HFpEF features (Table 1). However, patients with small LV demonstrated reduced left ventricular filling as indicated by lower E/A ratio (median, 0.81; IQR, 0.70–1.0) than patients with normalTTE (median, 0.89; IQR, 0.73–1.1; P = 0.02), similar to patients with HFpEF features (Table 1). In addition, TAPSE was lower in patients with small LV compared with patients with normalTTE. Noteworthy was that patients with small LV had a significantly lower LV mass index than those with HFpEF features or normalTTE. The LV mass adjusted for age, sex, and height correlated well with the skeletal muscle mass across all the groups (R = 0.53; P < 0.001).
Impaired LV Filling and All-Cause Mortality in the COSYCONET Cohort
Out of 1,187 patients with available survival data who had normalTTE, HFpEF features, or small LV, 82 patients died during a median follow-up time of 46 months (IQR, 23–55 months). Both patients with small LV or HFpEF features demonstrated a higher mortality rate than those with normalTTE (Table 2 and Figure 2). Table 2 demonstrates the results of different mixed-effects Cox models. Small LV or HFpEF features remained independent predictors of all-cause mortality after adjustment for basic confounders, exacerbation frequency and either RV, DlCO, or FEV1 (Table 2). Although small LV characterizes a subgroup of patients with more severe COPD, small LV was a predictor of all-cause mortality independent from features of disease severity such as the Body-mass index, Obstruction, Dyspnea, and Exercise (BODE) score index or its single components or the skeletal muscle mass index (see Table E1 in the online supplement). In a further analysis, a subgroup of patients with HFpEF features who had E/e′ ratio > 9 also showed a higher mortality rate than normalTTE (Table E2).
Table 2.
Mixed Effects Cox Models Demonstrate Survival Probabilities in Patients with Small Left Ventricle and Heart Failure with Preserved Ejection Fraction Features Compared with Normal Transthoracic Echocardiography
| Exposure Variable | HR | 95% CI | P Value |
|---|---|---|---|
| Model 1: unadjusted model | |||
| HFpEF features | 2.16 | 1.30–3.61 | 0.003 |
| Small LV | 2.75 | 1.54–4.89 | 0.001 |
| Model 2: adjusted for basic confounders | |||
| HFpEF features | 1.63 | 0.95–2.79 | 0.077 |
| Small LV | 2.29 | 1.26–4.18 | 0.007 |
| Model 3a: adjusted for basic confounders, exacerbations, and RV% | |||
| HFpEF features | 1.77 | 1.02–3.06 | 0.042 |
| Small LV | 2.26 | 1.23–4.16 | 0.009 |
| Model 3b: adjusted for basic confounders, exacerbations, and DlCO% | |||
| HFpEF features | 2.03 | 1.15–3.57 | 0.015 |
| Small LV | 2.12 | 1.12–4.03 | 0.020 |
| Model 3c: adjusted for basic confounders, exacerbations, and FEV1% | |||
| HFpEF features | 1.73 | 1.00–2.96 | 0.048 |
| Small LV | 1.88 | 1.03–3.45 | 0.040 |
Definition of abbreviations: CI = confidence interval; HFpEF = heart failure with preserved ejection fraction; HR = hazard ratio; LV = left ventricle; RV = residual lung volume.
Confounders: sex, age, body mass index, and smoking status versus former smoker. Exacerbations were defined as moderate to severe exacerbation within the last 12 months. Log-likelihood P values of all models was <0.001.
Figure 2.
Kaplan-Meier curves demonstrate reduced survival probability of patients with small LV and HFpEF features compared with patients with normal TTE. P values are of the log-rank test. HFpEF = heart failure with preserved ejection fraction; LV = left ventricle; TTE = transthoracic echocardiography.
Using Kaplan-Meier analysis, we visualize that the mortality rate associated with small LV is independent from lung hyperinflation. Therefore, we stratified patients with normalTTE and RV values :180% as normalTTE with hyperinflation (Figure 3). Patients with small LV had a higher mortality rate than patients who had normalTTE and hyperinflation (n = 471; hazard ratio, 2.09; 95% confidence interval, 1.11–3.97; P = 0.02) (Figure 3). Of note, mean predicted RV values were rather higher in patients with normalTTE and hyperinflation than those with small LV (192 ± 41 vs. 183 ± 59). In addition, we also noticed that patients with small LV had lower LV mass index as well as lower skeletal muscle mass index than patients with normalTTE and hyperinflation (mean [IQR] LV mass index, 66 [55–79] vs. 90 [77–104]; P < 0.0001; and mean [IQR]muscle mass index, 12.4 [7.5–25.6] vs. 13.2 [8.5–21]; P = 0.04).
Figure 3.
Kaplan-Meier curves demonstrate reduced survival probability of patients with small LV compared with patients without hyperinflation and normal transthoracic echocardiography (TTE) or patients with hyperinflation but normal TTE. P values are of the log-rank test. LV = left ventricle.
Discussion
In this cohort of well-characterized patients with COPD, we report that patients with small LV size, an indicator of reduced LV filling, or patients with HFpEF features have an increased rate of all-cause mortality compared with patients with COPD and no relevant cardiac dysfunction. Moreover, patients with small LV demonstrate a disturbed left ventricular filling pattern that is distinct from HFpEF. Accordingly, patients with small LV have indicators of reduced LV filling, yet, in contrast to patients with HFpEF features, they have normal ventricular filling pressure and do not exhibit elevated markers of ventricular stress or cardiac injury, namely serum NT-proBNP and hs-troponin I.
In COPD, cardiac dysfunction is a multifaceted condition that is often believed to present as HFpEF. Nevertheless, impaired ventricular filling might also present as a disease-related cardiac manifestation in patients with hyperinflation or emphysema. Previous studies have already demonstrated impaired ventricular filling in patients with COPD compared with healthy control subjects, using either echocardiography (25, 30, 31) or magnetic resonance imaging (32). In addition, we have previously shown an association between reduced ventricular filling and COPD severity, using echocardiographic parameters of diastolic function (16). However, so far, studies investigating left ventricular dysfunction in patients with COPD have not distinguished COPD-related reduced ventricular filling from diastolic dysfunction associated with HFpEF. In previous work, Barr has highlighted the need for discriminating COPD-related reduction in ventricular filling from coexisting HFpEF and its consecutive role in treating impaired ventricular filling in patients with COPD (17). Moreover, knowledge about the effects of reduced ventricular filling on COPD outcomes is largely limited to its negative impacts on exercise capacity (9) and physical activity (16), whereas its impact on patient survival is still unclear. To our knowledge, this is the first study to report increased all-cause mortality in patients with COPD and reduced ventricular filling, indicated by small LV size, in a cohort of strictly stratified patients according to clearly defined patterns of cardiac dysfunction. Small LV was an independent predictor of all-cause mortality after adjustment for the main predictors of mortality in COPD, including the BODE index, smoking status, exacerbation frequency, and measures of lung function such as residual lung volume, gas transfer capacity, and FEV1. A possible explanation of this later finding might be that many patients with small LV suffer from severe COPD and have worse lung function and emphysema. However, our finding that small LV is a predictor of mortality independent from lung function supports the notion that reduced ventricular filling is a clinical phenotype that confers worse prognosis through cardiopulmonary intermediates beyond lung function limitation.
The high frequency of patients with features of HFpEF in this COPD cohort can be mainly attributed to shared risk factors and etiologies, such as aging, smoking, and further cardiovascular risk factors like hypertension and dyslipidemia (4). However, COPD has also been shown to be an independent risk factor for some cardiovascular diseases (33, 34), including heart failure (34), and the diagnosis of COPD in patients with heart failure was associated with increased cardiovascular mortality and hospitalization (5). Nonetheless, cohort studies investigating the prevalence of HFpEF and other cardiac dysfunction patterns in patients with COPD based on objective assessments of cardiac structure, function, and biomarkers are scarce. In our cohort, almost one-third of the patients showed structural heart changes suggestive of HFpEF, such as LV hypertrophy or left atrial enlargement, of whom nearly half had more classical HFpEF features, with left ventricular stress indicated by elevated serum levels of NT-proBNP. A further key finding of this study is that patients with COPD and HFpEF features have a higher rate of all-cause mortality than patients with COPD and no cardiac dysfunction. As we report that having HFpEF features is a frequent comorbidity in patients with COPD, we suggest that HFpEF in COPD might be a treatable trait, and its management has the potential to decrease mortality in this subgroup of patients with COPD.
A hallmark of HFpEF is the increase in LV filling pressure caused by diastolic dysfunction, which is attributed to left ventricular stiffness often caused by ventricular hypertrophy (35). Likewise, reduced LV filling in patients with COPD and small LV could be accompanied by impaired diastolic function. However, it is pivotal not to misclassify patients with COPD and disturbed diastolic function due to reduced venous return, indicated by small LV, as HFpEF. In this context, the role of established measures of diastolic dysfunction and cardiac biomarkers appears to be key. Here, we report that patients with small LV did not differ from those with HFpEF features regarding impaired LV filling indicated by a lowered E/A ratio, yet patients with small LV had a normal E/e′ ratio, a parameter of LV filling pressure and a core indicator for HFpEF. Furthermore, serum levels of NT-proBNP, a marker of ventricular stress that strongly correlates with echocardiographic indices of ventricular filling pressure (36), were low in patients with small LV, similar to patients with COPD and no cardiac dysfunction. Therefore, we suggest that NT-proBNP might be an appropriate marker to distinguish reduced LV filling in COPD from coexisting HFpEF. Likewise, hs-troponin I, an indicator of myocardial injury, was significantly lower in patients with small LV than in those with HFpEF features. Furthermore, patients with small LV had lower TAPSE than patients with normalTTE, despite the fact that patients of both groups had values within the normal range. An explanation of this finding might be that low TAPSE is associated not only with reduced right ventricular function but also with reduced volume (37), which has been previously reported in patients with emphysema (14, 15). These findings highlight that small LV is a distinct pattern of cardiac dysfunction in COPD, which, despite the lack of elevated cardiac markers, is associated with increased all-cause mortality.
The underlying mechanisms of reduced LV filling in patients with COPD have been studied before. Previous studies have linked reduced left ventricular size and filling to lung hyperinflation and emphysema (7, 9, 12). It also has been shown that the decrease in left heart size is a consequence of reduced venous return because of the regional loss of the low-pressured pulmonary capillary beds in emphysematous lung areas (6, 12, 38, 39). The findings of our study are consistent with these previous findings, as they demonstrate that patients with small LV size have more severe lung hyperinflation, reduced gas transfer, and a higher percentage of emphysema, as per CT imaging in a subgroup of our cohort. A further unexplored aspect of small LV size might be the decrease in LV mass. Interestingly, our data indicate that patients with small LV have decreased skeletal muscle mass, which correlated well with the decrease in LV mass, as has been previously reported in elderly subjects with sarcopenia and reduced LV mass (40, 41). Future studies are warranted to elucidate the role of cardiac sarcopenia in the pathogenesis of reduced LV filling in COPD.
We acknowledge the limitations of this study. First is the lack of cardiac magnetic resonance imaging, a gold standard for the assessment of LV size. However, we used well-established echocardiographic structural features in addition to cardiac biomarkers to stratify a large cohort of patients with COPD based on their cardiac dysfunction patterns. Second, pulmonary CT imaging to evaluate lung emphysema is not part of the study protocol of the COSYCONET cohort. Here, we used lung volumes to quantify lung hyperinflation and gas transfer capacity as a surrogate for the extent of emphysema. Furthermore, pulmonary CT scans were available in a subgroup of patients, and their results support our findings. Third, the criteria used to define HFpEF in our study, including the use of LA diameter instead of LA volume, were adapted to the available echocardiographic measures and did not include the functional measures of diastolic dysfunction to avoid misclassifying patients with reduced LV filling and small LV as HFpEF. However, our definition of HFpEF features was based on central criteria such as LV hypertrophy and LA enlargement in addition to NT-proBNP. In addition, a sensitivity analysis that considered echocardiographic functional parameters in defining elevated LV filling pressure has confirmed that patients with COPD with HFpEF have higher mortality rates than those with no cardiac dysfunction. Finally, because systolic pulmonary artery pressure was not reported as a continuous variable, we cannot exclude the possibility of incipient pulmonary hypertension in patients with small LV. To mitigate a possible confounding effect of pulmonary hypertension, individuals with obvious RVD were excluded from the analyses. Moreover, patients with small LV exhibited an average TAPSE within the normal range, and their NT-proBNP concentrations did not significantly differ from those in patients without cardiac dysfunction. Therefore, it appears improbable that there is significant pulmonary hypertension affecting the LV filling.
In conclusion, small LV and HFpEF features indicate two different patterns of impaired LV filling that are associated with increased all-cause mortality in patients with COPD. As both patterns of impaired LV filling represent two distinct treatable traits, a guideline-directed treatment of HFpEF and an intensified management of lung hyperinflation and emphysema in patients with small LV have the potential to reduce mortality in COPD.
Footnotes
Supported by the German Federal Ministry of Education and Research (BMBF) Competence Network Asthma and COPD (ASCONET) and performed in collaboration with the German Centre for Lung Research (DZL). The project is funded by the BMBF grant number 01 GI 0881 and is supported by unrestricted grants from AstraZeneca GmbH, Bayer Schering Pharma AG, Boehringer Ingelheim Pharma GmbH & Co. KG, Chiesi GmbH, GlaxoSmithKline, Grifols Deutschland GmbH, MSD Sharp & Dohme GmbH, Mundipharma GmbH, Novartis Deutschland GmbH, Pfizer Pharma GmbH, and Takeda Pharma Vertrieb GmbH & Co. KG for patient investigations and laboratory measurements. The funding body was not involved in the design of the study or the collection, analysis, or interpretation of the data. Funding information for this article has been deposited with the Crossref Funder Registry. EEG was funded by the German Research Foundation (project number 413490537).
Author Contributions: Conception and design: M.A., B.W., and H.W. Analysis and interpretation: M.A., B.W., H.W., and K.F.R. Bioinformatic analysis: M.A. and B.W. Writing and reviewing the manuscript: M.A., B.W., P.A., K.K., F.T., E.E.G., M.C., A.-M.K., T.W., R.A.J., C.F.V., R.B., and K.F.R.
A data supplement for this article is available via the Supplements tab at the top of the online article.
Originally Published in Press as DOI: 10.1164/rccm.202310-1848OC on July 10, 2024
Author disclosures are available with the text of this article at www.atsjournals.org.
References
- 1. GBD 2013 Mortality and Causes of Death Collaborators. Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet . 2015;385:117–171. doi: 10.1016/S0140-6736(14)61682-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Rabe KF, Hurst JR, Suissa S. Cardiovascular disease and COPD: dangerous liaisons? Eur Respir Rev . 2018;27:180057. doi: 10.1183/16000617.0057-2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Divo M, Cote C, de Torres JP, Casanova C, Marin JM, Pinto-Plata V, et al. BODE Collaborative Group Comorbidities and risk of mortality in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med . 2012;186:155–161. doi: 10.1164/rccm.201201-0034OC. [DOI] [PubMed] [Google Scholar]
- 4. de Miguel Díez J, Chancafe Morgan J, Jiménez García R. The association between COPD and heart failure risk: a review. Int J Chron Obstruct Pulmon Dis . 2013;8:305–312. doi: 10.2147/COPD.S31236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Hawkins NM, Petrie MC, Jhund PS, Chalmers GW, Dunn FG, McMurray JJV. Heart failure and chronic obstructive pulmonary disease: diagnostic pitfalls and epidemiology. Eur J Heart Fail . 2009;11:130–139. doi: 10.1093/eurjhf/hfn013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Jörgensen K, Müller MF, Nel J, Upton RN, Houltz E, Ricksten S-E. Reduced intrathoracic blood volume and left and right ventricular dimensions in patients with severe emphysema: an MRI study. Chest . 2007;131:1050–1057. doi: 10.1378/chest.06-2245. [DOI] [PubMed] [Google Scholar]
- 7. Barr RG, Bluemke DA, Ahmed FS, Carr JJ, Enright PL, Hoffman EA, et al. Percent emphysema, airflow obstruction, and impaired left ventricular filling. N Engl J Med . 2010;362:217–227. doi: 10.1056/NEJMoa0808836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Alter P, Watz H, Kahnert K, Pfeifer M, Randerath WJ, Andreas S, et al. Airway obstruction and lung hyperinflation in COPD are linked to an impaired left ventricular diastolic filling. Respir Med . 2018;137:14–22. doi: 10.1016/j.rmed.2018.02.011. [DOI] [PubMed] [Google Scholar]
- 9. Watz H, Waschki B, Meyer T, Kretschmar G, Kirsten A, Claussen M, et al. Decreasing cardiac chamber sizes and associated heart dysfunction in COPD: role of hyperinflation. Chest . 2010;138:32–38. doi: 10.1378/chest.09-2810. [DOI] [PubMed] [Google Scholar]
- 10. Estépar RSJ, Kinney GL, Black-Shinn JL, Bowler RP, Kindlmann GL, Ross JC, et al. COPDGene Study Computed tomographic measures of pulmonary vascular morphology in smokers and their clinical implications. Am J Respir Crit Care Med . 2013;188:231–239. doi: 10.1164/rccm.201301-0162OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Hueper K, Vogel-Claussen J, Parikh MA, Austin JHM, Bluemke DA, Carr J, et al. Pulmonary microvascular blood flow in mild chronic obstructive pulmonary disease and emphysema: the MESA COPD study. Am J Respir Crit Care Med . 2015;192:570–580. doi: 10.1164/rccm.201411-2120OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Smith BM, Prince MR, Hoffman EA, Bluemke DA, Liu C-Y, Rabinowitz D, et al. Impaired left ventricular filling in COPD and emphysema: is it the heart or the lungs? The Multi-Ethnic Study of Atherosclerosis COPD study. Chest . 2013;144:1143–1151. doi: 10.1378/chest.13-0183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Hohlfeld JM, Vogel-Claussen J, Biller H, Berliner D, Berschneider K, Tillmann H-C, et al. Effect of lung deflation with indacaterol plus glycopyrronium on ventricular filling in patients with hyperinflation and COPD (CLAIM): a double-blind, randomised, crossover, placebo-controlled, single-centre trial. Lancet Respir Med . 2018;6:368–378. doi: 10.1016/S2213-2600(18)30054-7. [DOI] [PubMed] [Google Scholar]
- 14. van der Molen MC, Hartman JE, Vanfleteren LEGW, Kerstjens HAM, van Melle JP, Willems TP, et al. Reduction of lung hyperinflation improves cardiac preload, contractility, and output in emphysema: a clinical trial in patients who received endobronchial valves. Am J Respir Crit Care Med . 2022;206:704–711. doi: 10.1164/rccm.202201-0214OC. [DOI] [PubMed] [Google Scholar]
- 15. Stone IS, Barnes NC, James W-Y, Midwinter D, Boubertakh R, Follows R, et al. Lung deflation and cardiovascular structure and function in chronic obstructive pulmonary disease: a randomized controlled trial. Am J Respir Crit Care Med . 2016;193:717–726. doi: 10.1164/rccm.201508-1647OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Watz H, Waschki B, Boehme C, Claussen M, Meyer T, Magnussen H. Extrapulmonary effects of chronic obstructive pulmonary disease on physical activity: a cross-sectional study. Am J Respir Crit Care Med . 2008;177:743–751. doi: 10.1164/rccm.200707-1011OC. [DOI] [PubMed] [Google Scholar]
- 17. Barr RG. Rethinking chronic obstructive pulmonary disease. chronic pulmonary insufficiency and combined cardiopulmonary insufficiency. Ann Am Thorac Soc . 2018;15:S30–S34. doi: 10.1513/AnnalsATS.201708-667KV. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Waschki B, Watz H, Alter P, Kahnert K, Trudzinski FC, Groth E, et al. Impaired left ventricular filling and all-cause mortality in COPD [abstract] Am J Respir Crit Care Med . 2024;209:A7229. doi: 10.1164/rccm.202310-1848OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Karch A, Vogelmeier C, Welte T, Bals R, Kauczor H-U, Biederer J, et al. COSYCONET Study Group The German COPD cohort COSYCONET: aims, methods and descriptive analysis of the study population at baseline. Respir Med . 2016;114:27–37. doi: 10.1016/j.rmed.2016.03.008. [DOI] [PubMed] [Google Scholar]
- 20. Waschki B, Alter P, Zeller T, Magnussen C, Neumann JT, Twerenbold R, et al. German COSYCONET Cohort High-sensitivity troponin I and all-cause mortality in patients with stable COPD: an analysis of the COSYCONET study. Eur Respir J . 2020;55:1901314. doi: 10.1183/13993003.01314-2019. [DOI] [PubMed] [Google Scholar]
- 21. Janssen I, Heymsfield SB, Baumgartner RN, Ross R. Estimation of skeletal muscle mass by bioelectrical impedance analysis. J Appl Physiol (1985) . 2000;89:465–471. doi: 10.1152/jappl.2000.89.2.465. [DOI] [PubMed] [Google Scholar]
- 22. Kahnert K, Jörres RA, Kauczor H-U, Biederer J, Jobst B, Alter P, et al. COSYCONET Study Group Relationship between clinical and radiological signs of bronchiectasis in COPD patients: results from COSYCONET. Respir Med . 2020;172:106117. doi: 10.1016/j.rmed.2020.106117. [DOI] [PubMed] [Google Scholar]
- 23. Lang RM, Bierig M, Devereux RB, Flachskampf FA, Foster E, Pellikka PA, et al. European Association of Echocardiography Recommendations for chamber quantification: a report from the American Society of Echocardiography’s Guidelines and Standards Committee and the Chamber Quantification Writing Group, developed in conjunction with the European Association of Echocardiography, a branch of the European Society of Cardiology. J Am Soc Echocardiogr . 2005;18:1440–1463. doi: 10.1016/j.echo.2005.10.005. [DOI] [PubMed] [Google Scholar]
- 24. Nagueh SF, Smiseth OA, Appleton CP, Byrd BF, Dokainish H, Edvardsen T, et al. Recommendations for the evaluation of left ventricular diastolic function by echocardiography: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr . 2016;29:277–314. doi: 10.1016/j.echo.2016.01.011. [DOI] [PubMed] [Google Scholar]
- 25. Alter P, Jörres RA, Watz H, Welte T, Gläser S, Schulz H, et al. Left ventricular volume and wall stress are linked to lung function impairment in COPD. Int J Cardiol . 2018;261:172–178. doi: 10.1016/j.ijcard.2018.02.074. [DOI] [PubMed] [Google Scholar]
- 26. McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Böhm M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: developed by the Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC), developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail . 2022;24:4–131. doi: 10.1002/ejhf.2333. [DOI] [PubMed] [Google Scholar]
- 27. Lang RM, Badano LP, Mor-Avi V, Afilalo J, Armstrong A, Ernande L, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr . 2015;28:1–39.e14. doi: 10.1016/j.echo.2014.10.003. [DOI] [PubMed] [Google Scholar]
- 28. Grevious SN, Fernandes MF, Annor AK, Ibrahim M, Saint Croix GR, de Marchena E, et al. Prognostic assessment of right ventricular systolic dysfunction on post–transcatheter aortic valve replacement short‐term outcomes: systematic review and meta‐analysis. J Am Heart Assoc . 2020;9:e014463. doi: 10.1161/JAHA.119.014463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Pieske B, Tschöpe C, de Boer RA, Fraser AG, Anker SD, Donal E, et al. How to diagnose heart failure with preserved ejection fraction: the HFA-PEFF diagnostic algorithm: a consensus recommendation from the Heart Failure Association (HFA) of the European Society of Cardiology (ESC) Eur Heart J . 2019;40:3297–3317. doi: 10.1093/eurheartj/ehz641. [DOI] [PubMed] [Google Scholar]
- 30. Boussuges A, Pinet C, Molenat F, Burnet H, Ambrosi P, Badier M, et al. Left atrial and ventricular filling in chronic obstructive pulmonary disease: an echocardiographic and Doppler study. Am J Respir Crit Care Med . 2000;162:670–675. doi: 10.1164/ajrccm.162.2.9908056. [DOI] [PubMed] [Google Scholar]
- 31. Funk G-C, Lang I, Schenk P, Valipour A, Hartl S, Burghuber OC. Left ventricular diastolic dysfunction in patients with COPD in the presence and absence of elevated pulmonary arterial pressure. Chest . 2008;133:1354–1359. doi: 10.1378/chest.07-2685. [DOI] [PubMed] [Google Scholar]
- 32. Schäfer M, Humphries S, Stenmark KR, Kheyfets VO, Buckner JK, Hunter KS, et al. 4D-flow cardiac magnetic resonance-derived vorticity is sensitive marker of left ventricular diastolic dysfunction in patients with mild-to-moderate chronic obstructive pulmonary disease. Eur Heart J Cardiovasc Imaging . 2018;19:415–424. doi: 10.1093/ehjci/jex069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Maclagan LC, Croxford R, Chu A, Sin DD, Udell JA, Lee DS, et al. Quantifying COPD as a risk factor for cardiac disease in a primary prevention cohort. Eur Respir J . 2023;62:2202364. doi: 10.1183/13993003.02364-2022. [DOI] [PubMed] [Google Scholar]
- 34. Finkelstein J, Cha E, Scharf SM. Chronic obstructive pulmonary disease as an independent risk factor for cardiovascular morbidity. Int J Chron Obstruct Pulmon Dis . 2009;4:337–349. doi: 10.2147/copd.s6400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Pfeffer MA, Shah AM, Borlaug BA. Heart failure with preserved ejection fraction in perspective. Circ Res . 2019;124:1598–1617. doi: 10.1161/CIRCRESAHA.119.313572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Tschöpe C, Kasner M, Westermann D, Gaub R, Poller WC, Schultheiss H-P. The role of NT-proBNP in the diagnostics of isolated diastolic dysfunction: correlation with echocardiographic and invasive measurements. Eur Heart J . 2005;26:2277–2284. doi: 10.1093/eurheartj/ehi406. [DOI] [PubMed] [Google Scholar]
- 37. Zhao H, Kang Y, Pickle J, Wang J, Han Y. Tricuspid annular plane systolic excursion is dependent on right ventricular volume in addition to function. Echocardiography . 2019;36:1459–1466. doi: 10.1111/echo.14439. [DOI] [PubMed] [Google Scholar]
- 38. Vogel-Claussen J, Schönfeld C-O, Kaireit TF, Voskrebenzev A, Czerner CP, Renne J, et al. Effect of indacaterol/glycopyrronium on pulmonary perfusion and ventilation in hyperinflated patients with chronic obstructive pulmonary disease (CLAIM): a double-blind, randomized, crossover trial. Am J Respir Crit Care Med . 2019;199:1086–1096. doi: 10.1164/rccm.201805-0995OC. [DOI] [PubMed] [Google Scholar]
- 39.Washko GR, Nardelli P, Ash SY, Vegas Sanchez-Ferrero G, Rahaghi FN, Come CE, et al. Arterial vascular pruning, right ventricular size, and clinical outcomes in chronic obstructive pulmonary disease: a longitudinal observational study. Am J Respir Crit Care Med. 2019;200:454–461. doi: 10.1164/rccm.201811-2063OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Pelà G, Tagliaferri S, Perrino F, Bussolati G, Longobucco Y, Zerbinati L, et al. Interaction of skeletal and left ventricular mass in older adults with low muscle performance. J Am Geriatr Soc . 2021 doi: 10.1111/jgs.16812. [DOI] [PubMed] [Google Scholar]
- 41. Tinti MG, Scillitani A, Guglielmi G, Nieddu L, Carnevale V. Left ventricular mass and parameters of body composition in older adults. Mayo Clin Proc . 2022;97:626–628. doi: 10.1016/j.mayocp.2022.01.001. [DOI] [PubMed] [Google Scholar]