Figure 3. Activation of NOX2 is driven by p47phox S-379 phosphorylation leading to the interaction between regulatory and membrane components.
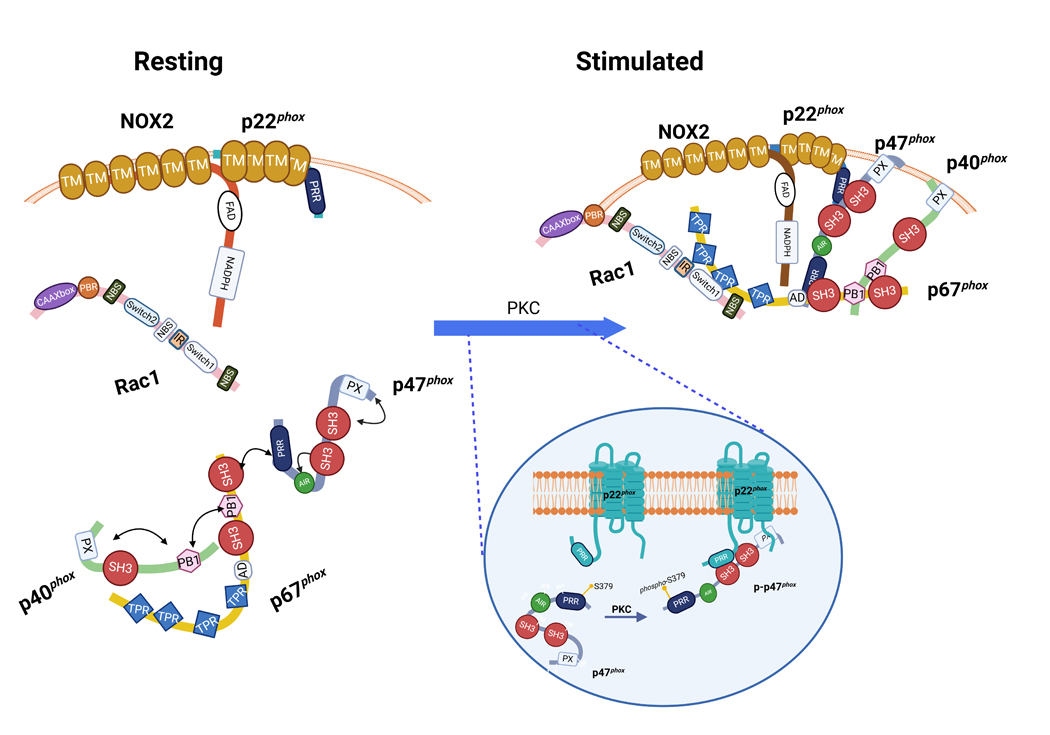
In the resting state, NOX2 oxidase is disassociated into its subunits and cofactors, while NOX2 and p22phox are in the membrane, p47phox–p67phox – p40phox are in an associated, yet dephosphorylated, state in a soluble trimeric complex, in which the p67phox and p40phox PB1 domains bind and a SH3 domain at the C-terminus of p67phox binds to the PRR of p47phox. At this time, the SH3 super-groove and PX domain of p47phox are masked by the polybasic auto-inhibitory region (AIR) which restricts p47phox to its folded and inactive state. Upon activation by diverse stimuli, protein kinase C (PKC) gets activated and phosphorylates p47phox on critical serines, this in turn disrupts the hydrogen bonds linking the C-terminal AIR and the tandem SH3 domains and exposes the supergroove pocket. This allows for the secondary phosphorylation of serine residues (S379) (see insert) permitting translocation and binding of p47phox to p22phox’s PRR domain. p47phox and p40phox acting through hallmark binding domains outlined in Figure 2 function to engage p67phox and Rac1 and scaffold the entire complex in place for NOX2 activation.
