Figure 4. Improving spatial resolution using FRET microscopy.
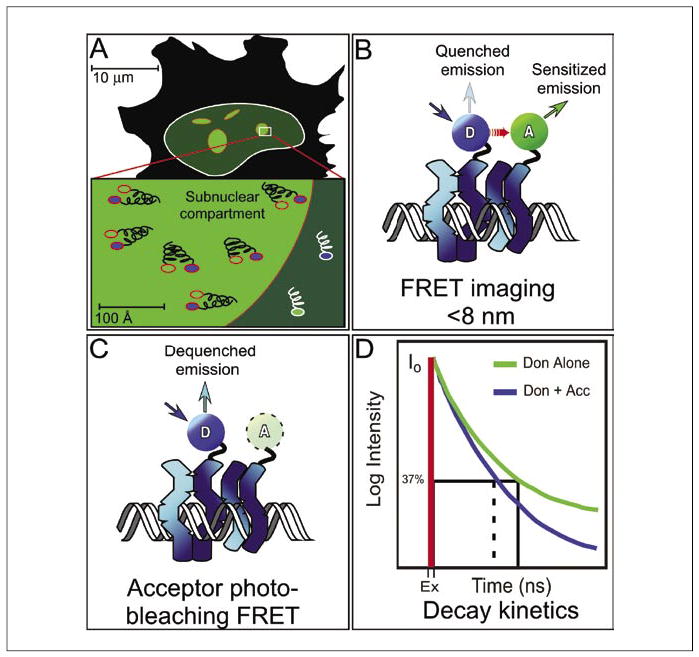
(A) Fluorescence microscopy is limited by the diffraction of light to a resolution of approximately 200 nm, and objects that are closer to-gether will appear as a single object, so considerable distances may actually separate proteins that appear co-localized by fluorescence microscopy. (B) FRET microscopy detects the direct transfer of excitation energy (red arrow) from a donor (D) fluorophore to an acceptor (A) fluorophore that is limited to distances of less than about 8 nm. When energy transfer occurs, the donor fluorescence signal is quenched, and there is sensitized emission from the acceptor. (C) Acceptor photobleaching FRET measures donor quenching by destroying the acceptor, resulting in the elimination of FRET and an increase in the donor signal. (D) Fluorescence decay kinetic measurements determine the time that a probe spends in the excited state prior to returning to the ground state. The excited-state lifetime measurements can be separated into different decay components that provide more detailed information about the environment surrounding the probe. FRET, fluorescence resonance energy transfer; Ex, excited-state lifetime; Don, donor; Don + Acc, donor and acceptor.
