Abstract
Background
The mechanism of aluminum-induced neurotoxicity is not clear. The involvement of glutamate in the aluminium-induced neurocomplications has been suggested. Brain glutamate levels also found to be altered in protein malnutrition. Alterations in glutamate levels as well as glutamate-α-decarboxylase in different regions of rat brain has been reported in response to aluminum exposure. Thus the study of glutamate metabolising enzymes in different brain regions of rats maintained on either normal or restricted protein diet may be of importance for understanding the neurotoxicity properties of aluminium.
Results
Dietary protein restrictions does not have an significant impact on regional aluminum content of the brain. The interaction of aluminum intoxication and protein restriction is significant in the thalamic area and the midbrain-hippocampal region in cases of glutamate oxaloacetate transaminase. In the case of gluatmate pyruvate transaminase, this interaction is significant only in thalamic area.
Conclusion
The metabolism of amino acids, as indicated by activities of specific transaminases, of brain is altered in response to aluminum exposure. These alterations are region specific and are dependent on dietary protein intake or manipulation of the brain amino acid homeostasis.
Background
The ubiquity of numerous aluminum compounds found in soil, water and air ensure that we live in an aluminum-rich environment. The entrance aluminum to human body is carefully refrained; however, the metal gains access to the body. The so-called 'biological inertness' and other chemical properties of aluminum render maximum use of this metal in our every day living. The toxic consequences in human after aluminum exposure are now well established [1,2]. The brain is the most sensitive organ to show aluminum-induced disorders [3]. Despite several hypotheses, the mechanism of aluminum-induced neurotoxicity is not clear [4,5]. There are suggestions that glutamate may be involved in the aluminum-induced neurocomplications. The excitotoxic effect of glutamate is believed to be the cause of several neurodegenerative processes [6] and several enzymes with the capacity to degrade glutamate have been suggested as possible neuroprotectants [7]. It has been reported that aluminum impairs the glutamatergic neurotransmission [8] and mediates glutamate-induced neurotoxicity in organotypic cultures [9]. On the other hand, glutamate is an important metabolic intermediate of the brain and it has potential aluminum binding capacity [10]. In our earlier studies, we have shown that aluminum causes alteration in glutamate levels and glutamate α-decarboxylase activities of different brain regions [11]. In addition to this, we had also shown dietary protein restrictions to alter the aluminum-induced neurological responses in several brain regions [3,12]. Thus the present investigation is aimed to study the impact of aluminum as well as dietary protein restriction on transaminases of the brain, which are related to the glutamate metabolism.
Results
Body weights
The body weights of the rats during the period of treatment (0–4th week) are presented in figure 1. A slightly higher gain in body weight (on the 4th week) was observed in the aluminum-treated rats of both the normal and low protein dietary groups. However, these differences in body weight are not statistically significant. During the course of the treatment, a decrement in food intake was observed in the aluminum-treated rats of both dietary groups (Figure 2).
Figure 1.
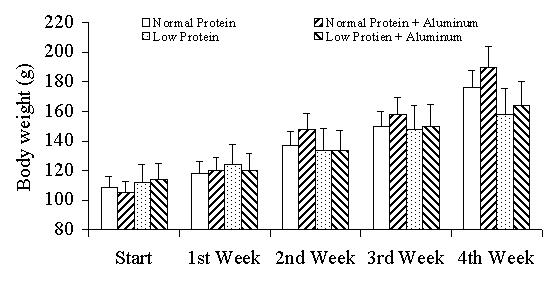
Week wise body weight of rats during the period of treatment. Each data point represents the mean of six observations ± SEM.
Figure 2.
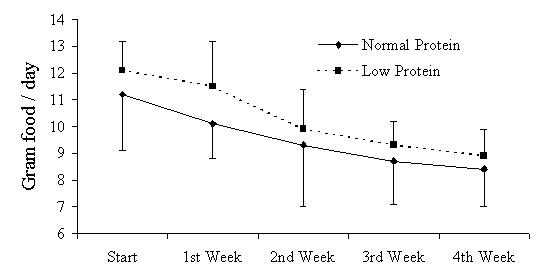
Week wise food intakes of rats during the period of treatment. Each data point represents the mean of six observations ± SEM.
Aluminum content
Changes in the aluminum content in different brain regions on exposure to aluminum are given in table 1. The present treatment increases the aluminum content of the cerebrum, thalamic area, midbrain-hippocampal region and cerebellum by 70.6%, 115.6%, 123.3% and 53.5% respectively in normal protein-fed group of rats, whereas 59.4%, 93.3%, 107.7% and 53.2% respectively in low protein-fed group of rats. Though there is significant amount of added components for treatment effects in all of the tested brain regions, Scheffe's F test for multiple comparisons showed that aluminum exposure significantly increased the aluminum content of cerebrum, thalamic area and midbrain-hippocampal region of normal protein group and thalamic area and midbrain-hippocampal region of rats maintained on low protein diet. However, two factor ANOVA (with replication) showed that only effects of aluminum treatment were significant in all the four brain regions.
Table 1.
Regional aluminum content (μmoles / 100 mg tissue) in rat brain.
| Brain regions | Groups of animals | Statistical calculation (F) | ||||
| Normal protein | Low protein | ANOVA Single factor | ANOVA Two factors (with replication) | |||
| Control | Aluminum exposed | Control | Aluminum exposed | |||
| Cerebrum | 2.86 ± 0.47 | 4.88 ± 0.44* | 2.91 ± 0.54 | 4.64 ± 0.21 | 6.30 # | Protein : 0.05 Aluminum : 18.75 # Interaction : 0.11 |
| Thalamic area | 4.16 ± 0.52 | 8.97 ± 0.71* | 4.96 ± 0.42 | 9.59 ± 0.52* | 24.69 # | Protein : 1.65 Aluminum : 72.39 # Interaction : 0.03 |
| Midbrain-hippocampal region | 4.17 ± 0.42 | 9.31 ± 0.62* | 4.13 ± 0.25 | 8.58 ± 0.68* | 28.47 # | Protein : 0.55 Aluminum : 84.39# Interaction : 0.45 |
| Cerebellum | 3.10 ± 0.52 | 4.76 ± 0.64 | 2.82 ± 0.34 | 4.32 ± 0.44 | 3.56# | Protein : 0.51 Aluminum : 10.14# Interaction : 0.03 |
Data are means of six observations ± SEM. * indicates significant difference (p < 0.05) in comparison to respective control. # indicates calculated F is higher than the critical F (α = 0.05).
Glutamate oxaloacetate transaminase
The glutamate oxaloacetate transaminase (GOT) activities of different brain regions of all groups of rats are presented in table 2. The cerebrum, thalamic area, midbrain-hippocampal region and cerebellum of low protein-fed group of rats showed 9.7%, 17.9%, 20.8% and 13.6% increase in GOT activities in response to aluminum exposure. On the other hand, only the cerebellum of the normal protein-fed group of rats showed 1.6% increase in GOT activity when exposed to aluminum. Regional GOT activities of thalamic area and midbrain-hippocampal region of rats maintained on normal protein diet were reduced by 10% and 11.5% respectively. Changes in the GOT activity of only the cerebrum and midbrain-hippocampal regions of inadequately protein-fed rats were found to be statistically significant by Scheffe's F test for multiple comparisons. However, significant impacts of treatment were observed in all the brain regions (ANOVA single factor). Two factor ANOVA with replication showed that aluminum exposure contributed significantly on the changes of the GOT activity of cerebellum only, but the interactions of impact of dietary protein deficiency and aluminum exposure were significant in thalamic area as well as midbrain-hippocampal region.
Table 2.
Regional glutamate oxaloacetate transaminase activity (μg pyruvate produced / hr / mg tissue) in rat brain.
| Brain regions | Groups of animals | Statistical calculation (F) | ||||
| Normal protein | Low protein | ANOVA Single factor | ANOVA Two factors (with replication) | |||
| Control | Aluminum exposed | Control | Aluminum exposed | |||
| Cerebrum | 8.42 ± 0.37 | 8.38 ± 0.35 | 7.69 ± 0.25 | 8.44 ± 0.25* | 9.03 # | Protein : 1.14 Aluminum : 1.31 Interaction : 1.62 |
| Thalamic area | 8.48 ± 0.16 | 7.64 ± 0.28 | 7.39 ± 0.32 | 8.71 ± 0.52 | 3.41 # | Protein : 0.00 Aluminum : 0.49 Interaction : 9.75 # |
| Midbrain-hippocampal region | 8.50 ± 0.18 | 7.52 ± 0.31 | 7.25 ± 0.27 | 8.76 ± 0.20* | 9.03 # | Protein : 0.00 Aluminum : 1.15 Interaction : 25.95 # |
| Cerebellum | 8.08 ± 0.25 | 8.21 ± 0.21 | 7.67 ± 0.23 | 8.71 ± 0.28 | 3.12# | Protein : 0.04 Aluminum : 5.86# Interaction : 3.46 |
Data are means of six observations ± SEM. * indicates significant difference (p < 0.05) in comparison to respective control. # indicates calculated F is higher than the critical F (α = 0.05).
Glutamate pyruvate transaminase
Table 3 represents the regional GPT activities of the different groups of rats. Elevated activities of GPT were observed in the cerebellum (24.6%), thalamic area (51.4%) and cerebellum of aluminum-exposed animals of normal protein-fed group. In the protein-restricted group, the GPT activities were found to be increased in the cerebellum (10.5%) and decreased in midbrain-hippocampal region (14%). Though a significant impact of treatments were observed in all the brain regions except the cerebrum (single factor ANOVA), only changes in the thalamic area of normal protein diet group was found to significant by Scheffe's F test. Two factor ANOVA (with replication) showed that impact of aluminum is significant in thalamic area and cerebellum, impact of protein restriction is significant in midbrain-hippocampal region and cerebellum, and the interaction of both protein restriction and aluminum treatment is significant only in thalamic area.
Table 3.
Regional glutamate pyruvate transaminase activity (μg pyruvate produced / hr / mg tissue) in rat brain.
| Brain regions | Groups of animals | Statistical calculation (F) | ||||
| Normal protein | Low protein | ANOVA Single factor | ANOVA Two factors (with replication) | |||
| Control | Aluminum exposed | Control | Aluminum exposed | |||
| Cerebrum | 3.41 ± 0.21 | 4.25 ± 0.30 | 3.68 ± 0.25 | 3.69 ± 0.20 | 2.07 | Protein : 0.35 Aluminum : 2.97 Interaction : 2.88 |
| Thalamic area | 2.49 ± 0.19 | 3.77 ± 0.19* | 3.41 ± 0.16 | 3.48 ± 0.18 | 9.23# | Protein : 2.96 Aluminum : 13.78# Interaction : 10.96# |
| Midbrain-hippocampal region | 3.34 ± 0.19 | 3.34 ± 0.14 | 3.15 ± 0.07 | 2.71 ± 0.15 | 4.10# | Protein : 7.71# Aluminum : 2.29 Interaction : 2.29 |
| Cerebellum | 4.91 ± 0.09 | 5.43 ± 0.22 | 4.58 ± 0.10 | 5.06 ± 0.13 | 5.88# | Protein : 5.84# Aluminum : 11.80# Interaction : 0.02 |
Data are means of six observations ± SEM. * indicates significant difference (p < 0.05) in comparison to respective control. # indicates calculated F is higher than the critical F (α = 0.05).
The correlations between the region wise changes in the level of aluminum and activities of transaminases in response to aluminum exposure (of both adequately protein-fed rats and inadequately protein-fed rats) have been shown in table 4. Only significantly negative correlation was observed between changes in aluminum level and alteration of GOT activities in adequately protein-fed group of rats.
Table 4.
Correlation between regions of brain in terms of transaminase activity and aluminum content.
| Correlation between | Normal protein | Low protein |
| Aluminum : GOT | -1.00* | 0.91 |
| Aluminum : GPT | 0.16 | 0.90 |
| GOT : GPT | -0.10 | -0.60 |
* Indicates significant correlation (p < 0.05)
Discussion
Aluminum treatment of rats fed on either normal protein or low protein diet increased the aluminum content in all the brain regions. The midbrain-hippocampal region of the aluminum-exposed rat brain showed highest level of accumulation of aluminum. This observation is in agreement with the available reports indicating hippocampus to be the susceptible brain region for accumulation of aluminum [15-17]. However, the regional differences in increment of aluminum level on exposure to aluminum are comparable in both the dietary regimens. Protein deficiency is not significantly impinging on regional aluminum level of brain (Table 1).
The observation that glutamate oxaloacetate transaminase (GOT) is present in rat cerebral homogenate at about the same concentrations as in liver suggests its importance in the brain amino acid pool homeostasis [18]. The GOT activity is found in both mitochondrial and soluble fractions of the brain. The enzyme in mitochondria is latent and can be 'activated' by various procedures that disrupt the mitochondrial structure. The critical role of mitochondrial dysfunction in the pathogenesis of age-related nerve cell degeneration has been suggested in several occasions [19,20]. On the other hand, coexposure of aluminum and glutamate is reported to produce morphological abnormalities of mitochondrial structures in pyramidal neurons of hippocampal formation, which are not observed when exposed to aluminum or glutamate alone [9]. Toninello et al [21] also showed that aluminum is an inducer of mitochondrial permeability transition.
No significant change in GOT activity has been observed in any of the brain regions in response to aluminum exposure when the animals were maintained on normal protein diet. Favarato and Zatta [22] also did not observe any alteration in serum GOT activity in response to aluminum. However, regional specificity was observed in aluminum-induced elevation of brain GOT activity, in the inadequately protein-fed animals. Increased GOT activity may be connected with an increased transport of NADH from the cytosol to mitochondria [23]
In the present investigation the regional variation of normal GOT activity is not prominent but the response to aluminum insult shows significant regional specificity. Thus it may be suggested that aluminum exposure causes region specific alteration in amino acid metabolism. Hence, altered amino acid distribution pattern within the brain might be expected in response to aluminum exposure.
The observed region-specific alterations may also be attributed to the level of accumulated aluminum in the respective regions. Diencephalon is reported to be the most vulnerable portion in case of accumulation in brain [17,24], which is also observed in the present investigation (Table 1).
Though glutamate pyruvate transaminase (GPT) is indicated to be distributed in both mitochondrial and soluble fractions of the brain, unlike GOT, it is less active in brain. However, Matthews et al [7] reported that both the enzymes degrade glutamate; though only GPT was able to reduce toxic (500 μM) levels of glutamate into the physiologic (<20 μM) range. Our earlier observation showed that aluminum exposure causes significant increase in glutamate content in different regions of brain while only thalamic area showed enhanced γ-amino butyrate content [11]. In the present investigation, the GPT activity is increased only in thalamic area of sufficient protein-fed group. These observations support region-wise specific sensitivity to aluminum exposure as indicated in nucleic acid content and nucleic acid metabolism [3]. On the other hand, in continuation with earlier studies [11] it can be suggested that either the enhanced glutamate level is not sufficient enough to trigger the response of GPT in these regions or other removal pathway(s) are activated. In addition to these, direct inhibition of GPT by aluminum and local concentration of pyruvate may also be involved in the present observation of GPT activity. The present investigation has revealed differential response of enzymes in response to aluminum insult in various regions of the brain. This finding corroborates with the earlier observation of regional variation in enzyme activities of brain in response to aluminum exposure [12,25].
These transaminase reactions are reversible, but the equilibrium of the GOT and GPT reactions favour formation of aspartate and alanine respectively [26]. Increased aminotransferase activities might participate in the enhanced synthesis of excitatory amino acid neurotransmitters in the nervous system [24].
Conclusions
These observations are suggestive of the fact that the metabolism of the amino acids in response to aluminum exposure is very much region specific. As amino acids in the brain compose a major part of the neurotransmitters, it is expected that, these alterations might also be involved in the aluminum-induced neurobehavioral impairments. These alterations are region specific and are dependent on dietary protein intake or manipulation of the brain amino acid homeostasis.
Methods
Animal maintenance and aluminum exposure
Male albino rats of Wistar strain weighing 100–120 g were divided into four groups of almost equal average body weight. The animals of two groups were maintained on the diet containing 18% protein (casein) while the remaining two groups were maintained on 6% protein (casein) diet as reported earlier [3]. One group of rats from each of the two dietary regimens received 4.2 mg /Kg body weight / day as aluminum chloride intraperitoneally for four weeks and the animals of the remaining groups serving as pair-fed controls received only the vehicle.
Tissue collection
After the period of treatment, overnight fasted rats were sacrificed by cervical dislocation. The whole brain was removed, washed with ice-cold saline, blotted dry and immediately transferred to the ice chamber. Cerebrum, thalamic area, midbrain-hippocampal region and cerebellum were separated following of the dissection of the frozen brain after thawing (making soft) in cold, under dissecting microscope [3].
Biochemical estimation
The aluminum content of each tissue region was estimated by Atomic Absorption Spectrometer after digested with acid mixture, containing nitric acid, sulphuric acid and perchloric acid in the ratio of 6:1:1, over a regulated heater [12]. The activities of glutamate oxaloacetate transaminase and glutamate pyruvate transaminase were measured through spectrophotometric determination of 2,4 dinitrophenylhydrazone of pyruvate [13].
Authors' contributions
PN carried out the literature survey, experimental works and statistical calculation.
AKC conceived of the study, and participated in its design and coordination.
All authors read and approved the final manuscript.
Contributor Information
Prasunpriya Nayak, Email: nprasunpriya@hotmail.com.
Ajay K Chatterjee, Email: ajayk@cubmb.ernet.in.
References
- US Public Health Science Report Toxicological profile of aluminum and its compounds. 1992. pp. 1–99.
- Nayak P. Aluminum : Impacts and Disease. Environ Res. 2002;89:111–115. doi: 10.1006/enrs.2002.4352. [DOI] [PubMed] [Google Scholar]
- Nayak P, Chatterjee AK. Impact of protein malnutrition on subcellular nucleic acid and protein status of brain of aluminum-exposed rats. J Toxicol Sci. 1998;23:1–14. doi: 10.2131/jts.23.1. [DOI] [PubMed] [Google Scholar]
- Hermenegildo C, Soez R, Minoia C, Manzo L, Felipo V. Chronic exposure to aluminum impairs the glutamate – nitric oxide – cGMP pathway in the rat in vivo. Neurochem Int. 1999;34:245–253. doi: 10.1016/S0197-0186(99)00010-8. [DOI] [PubMed] [Google Scholar]
- Nayak P, Chatterjee AK. Biochemical view of aluminum-induced neurohazards. J Environ Biol. 1999;20:77–84. [Google Scholar]
- Mahy N, Prats A, Riveros A, Andres N, Bernal F. Basal ganglia calcification induced by excytotoxicity : an experimental model characterized by electron microscopy and X – ray microanalysis. Acta Neuropathol (Berl) 1999;98:217–225. doi: 10.1007/s004010051072. [DOI] [PubMed] [Google Scholar]
- Matthews CC, Zielke HR, Wollack JB PS. Fishman Enzymatic degradation protects neurons from glutamate excitotoxicity. J Neurochem. 2000;75:1045–1052. doi: 10.1046/j.1471-4159.2000.0751045.x. [DOI] [PubMed] [Google Scholar]
- Llansola M, Minana MD, Montoliu C, Saez R, Carbalan R, Manzo L, Felipo V. Prenatal exposure to aluminum reduces expression of neuronal nitric oxide synthase and soluble guanylate cyclase and impairs glutamatergic neurotransmission in rat cerebellum. J Neurochem. 1998;73:712–718. doi: 10.1046/j.1471-4159.1999.0730712.x. [DOI] [PubMed] [Google Scholar]
- Matyja E. Aluminum enhances glutamate-mediated neurotoxicity in organotypic cultures of rat hippocampus. Folia Neuropathol. 2000;38:47–53. [PubMed] [Google Scholar]
- de Voto E, Yokel RA. The biological speciation and toxicokinetics of aluminum. Environ Health Perspect. 1994;102:940–951. doi: 10.1289/ehp.94102940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nayak P, Chatterjee AK. Effect of aluminum exposure in brain glutamate and GABA systems : an experimental study in rats. Food Chem Toxicol. 2001;39:1285–1289. doi: 10.1016/S0278-6915(01)00077-1. [DOI] [PubMed] [Google Scholar]
- Nayak P, Chatterjee AK. Differential responses of certain brain phosphoesterases to aluminum in dietary protein adequacy or inadequacy. Food Chem Toxicol. 2001;39:587–592. doi: 10.1016/S0278-6915(00)00173-3. [DOI] [PubMed] [Google Scholar]
- Bergmeyer HU, Bernt E. Glutamate oxaloacetate transaminase, Glutamate pyruvate transaminase. In: Bergmeyer HU, editor. Methods of Enzymatic Analysis. Academic Press, New York; 1963. pp. 837–852. [Google Scholar]
- Das D. Statistics in Biology and Psychology, Academic Press, Calcutta. 1981.
- Crapper DR, Krishnan SS, Quitkat S. Aluminum, neurofibrillary degeneration and Alzheimer's disease. Brain. 1976;99:67–80. doi: 10.1093/brain/99.1.67. [DOI] [PubMed] [Google Scholar]
- Thorne BM, Donhoe T, Lin KN, Lyon S, Mdediros DM, Weaver ML. Aluminum ingestion and behaviour in Long-Evans rats. Physiol Behav. 1986;36:63–67. doi: 10.1016/0031-9384(86)90074-0. [DOI] [PubMed] [Google Scholar]
- Julka D, Vasistha RK, Gill KD. Distribution of aluminum in different brain regions and body organs of rats. Biol Trace Elem Res. 1996;52:181–192. doi: 10.1007/BF02789460. [DOI] [PubMed] [Google Scholar]
- Streecher HJ. Transaminases. In: A Lajtha, editor. In : Handbook of Neurochemistry. III. Plenum Press, New York; 1970. pp. 173–192. [Google Scholar]
- Beal MF, Brovillet E, Jenkins B, Henshaw R, Rosen B, Hyman BT. Age-dependent striatal excitotoxic lesion produced by the endogenous mitochondrial inhibitor malanate. J Neurochem. 1993;61:1147–1150. doi: 10.1111/j.1471-4159.1993.tb03633.x. [DOI] [PubMed] [Google Scholar]
- Schulz JB, Mathews RT, Henshaw DR, Beal MF. Neuroprotective strategies for treatment of lesions produced by mitochondrial toxins : implication for neurodegenerative diseases. Neuroscience. 1996;71:1043–1048. doi: 10.1016/0306-4522(95)00527-7. [DOI] [PubMed] [Google Scholar]
- Toninello A, Clari G, Mancon M, Tognon G, Zatta P. Aluminum as an inducer of the mitochondrial permeability transition. J Biol Inorg Chem. 2000;5:612–623. doi: 10.1007/s007750000144. [DOI] [PubMed] [Google Scholar]
- Favarato M, Zatta PF. Differential aluminum lactate toxicity in rabbits using either aqueous solutions or liposomal suspensions. Toxicol Lett. 1993;66:133–146. doi: 10.1016/0378-4274(93)90088-F. [DOI] [PubMed] [Google Scholar]
- Netopilova M, Haugvicova R, Kubova H, Drsata J, Mares P. Influence of convulsants on rat brain activities of alanine aminotransferase and aspartate aminotransferase. Neurochem Res. 2001;26:1285–1291. doi: 10.1023/A:1014386416109. [DOI] [PubMed] [Google Scholar]
- Santos F, Chan JC, Yang MS, Savory J, Wills MR. Aluminum deposition in the central nervous system : Preferential accumulation in the hippocampus in weanling rats. Med Biol. 1987;65:53–55. [PubMed] [Google Scholar]
- Guo-Ros SX, Yang EY, Walsh TJ, Bondy SC. Decrease in glial fibrillary acidic protein in rat frontal cortex following aluminum treatment. J Neurochem. 1999;73:1609–1614. doi: 10.1046/j.1471-4159.1999.0731609.x. [DOI] [PubMed] [Google Scholar]
- Moss DW, Henderson AR. Clinical enzymology. In: Burtis CA, Ashwood ER, editor. In : Teitz Textbook of Clinical Chemistry. Harcourt Brace and Company Asia Pvt. Ltd., Singapore; 1999. pp. 617–721. [Google Scholar]


