Abstract
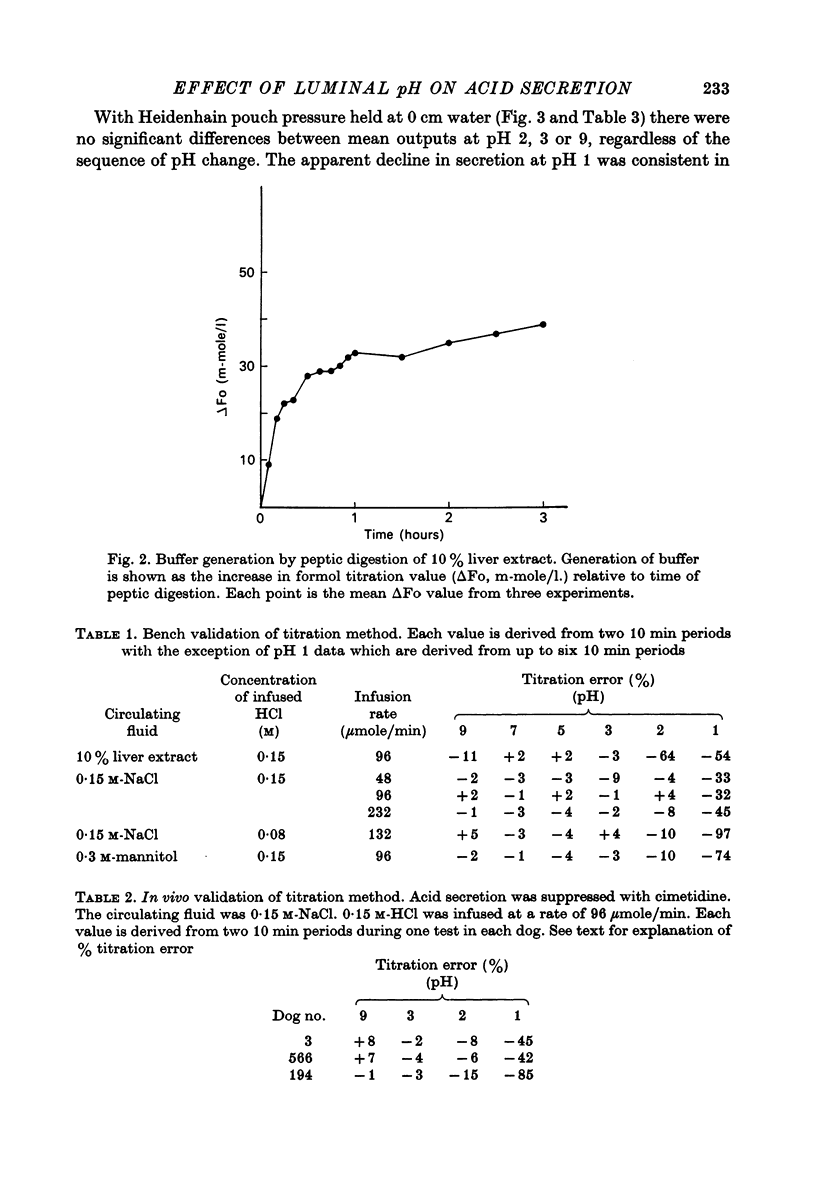
1. An apparatus for intragastric titration has been devised and its validity tested. Both when attached to a beaker simulating a pouch and when attached to a pouch whose secretion was suppressed by infusing cimetidine, the apparatus accurately measured added acid when the endpoint setting was between pH 3·0 and 9·0. At pH 2·0 and 1·0 with liver extract and at pH 1·0 with saline, the amount of acid added was markedly underestimated.
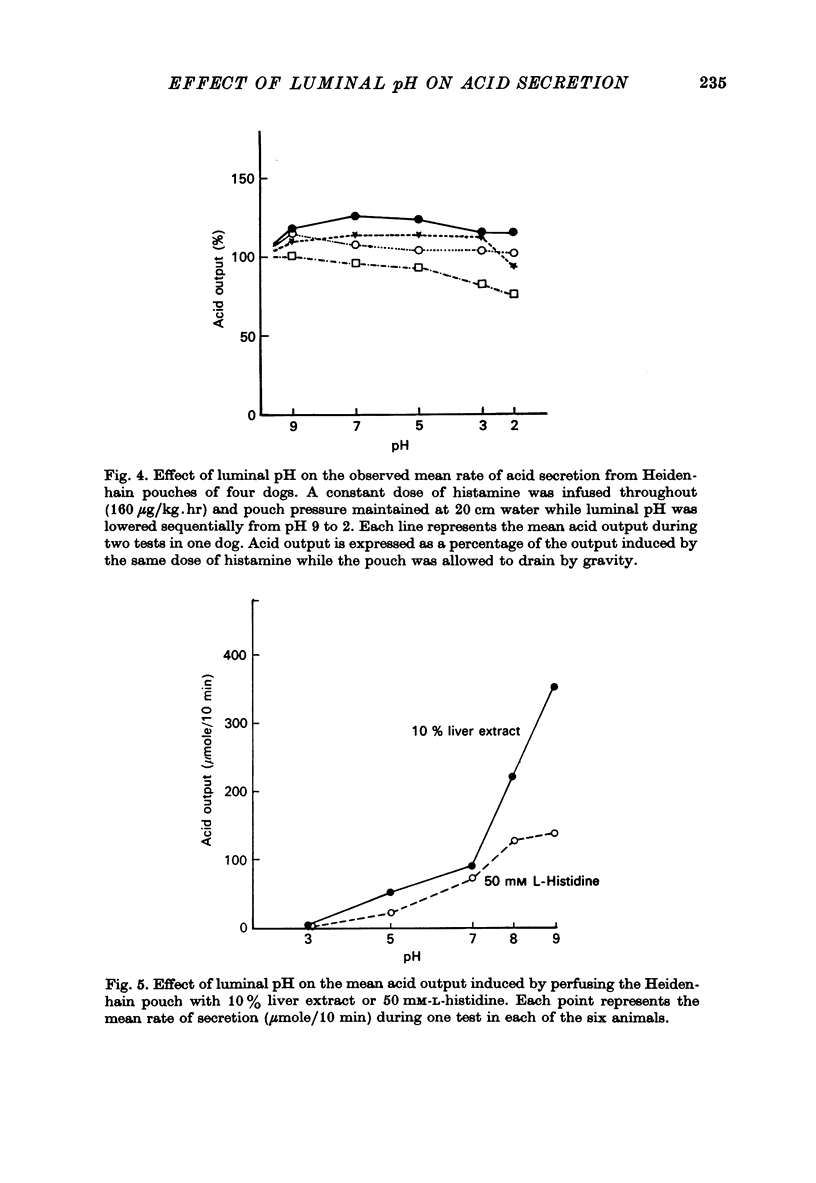
2. In dogs with vagally denervated pouches, during stimulation by I.V. infusion of histamine or pentagastrin, the rate of acid secretion as measured by intrapouch titration was uninfluenced by changes in luminal pH between 2·0 and 9·0. The apparent decrease in acid secretion at pH 1·0 could be shown to be due entirely to artifact in that no change in acid secretion was found when the gain in mass of acid was simultaneously measured by using a non-absorbable dilution indicator to measure volume gain and titration of samples to pH 7·0 to measure acid concentration.
3. During stimulation of acid secretion by solutions of liver extract or of L-histidine instilled into the pouch, the rate of acid secretion was found to increase markedly as pH was increased from 3·0 to 9·0 thus confirming our earlier findings.
4. We conclude that while stimulation of acid secretion by topical stimulants is highly dependent on luminal pH, secretion increasing as pH increases, stimulation by parenteral agents such as histamine and pentagastrin is not influenced by luminal pH in the range from pH 1·0 to 9·0.
Full text
PDF








Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Debas H. T., Grossman M. I. Chemicals bathing the oxyntic gland area stimulate acid secretion in dog. Gastroenterology. 1975 Sep;69(3):654–659. [PubMed] [Google Scholar]
- Fordtran J. S., Walsh J. H. Gastric acid secretion rate and buffer content of the stomach after eating. Results in normal subjects and in patients with duodenal ulcer. J Clin Invest. 1973 Mar;52(3):645–657. doi: 10.1172/JCI107226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gregory R. A. Some factors influencing the passage of fluid through intestinal loops in dogs. J Physiol. 1950 Apr 15;111(1-2):119–137. doi: 10.1113/jphysiol.1950.sp004468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones R. S., Yee T. K., Michielsen C. E. A modified Thomas cannula for gastric and intestinal fistulas. J Appl Physiol. 1971 Mar;30(3):427–428. doi: 10.1152/jappl.1971.30.3.427. [DOI] [PubMed] [Google Scholar]
- Konturek S. J., Obtulowicz W., Tasler J. Characteristics of gastric inhibition by acidification of oxyntic gland area. J Physiol. 1975 Oct;251(3):699–709. doi: 10.1113/jphysiol.1975.sp011117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konturek S. J., Tasler J., Obtulowicz W., Cieszkowski M. Comparison of amino acids bathing the oxyntic gland area in the stimulation of gastric secretion. Gastroenterology. 1976 Jan;70(1):66–69. [PubMed] [Google Scholar]


