TABLE 4.
Partition coefficients determined from the apparent first-order rate constants for the reaction between MePEG-OPSS reagents and Cys-135 in L135C7 αHL pores
| MePEG-OPSS* | 1000 mM KCl | 300 mM KCl† |
|---|---|---|
| PEG-0.85k | 0.34 ± 0.06 | 0.32 ± 0.04 |
| PEG-1.7k | 0.14 ± 0.03 | 0.13 ± 0.03 |
| PEG-2.5k | 0.069 ± 0.009 | 0.058 ± 0.008 |
| PEG-5.0k | 0.0040 ± 0.0008 | 0.0032 ± 0.0007 |
Values are the mean ± SD of four experiments. The buffer solutions also contained 10 mM Tris·HCl and 100 μM EDTA, pH 8.5. The polymer concentration in the trans bath was 4 mM. The applied potential was −40 mV for the experiments at 300 mM KCl and +100 mV for the experiments at 1000 mM KCl. The partition coefficients (Π) were calculated from the reaction rates by applying a scaling law, which has been shown to apply to the αHL pore (Movileanu and Bayley, 2001), 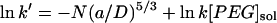 , where k′ is the apparent first-order reaction rate constant; a, the persistence length of the polymer (3.5 Å) (Rex et al., 1998; Kienberger et al., 2000); D, the diameter of the transmembrane barrel; k, the second-order reaction rate constant; and [PEG]sol, the PEG concentration in the bulk aqueous phase. The slope of a plot of ln k′ versus N was used to obtain (a/D)5/3, which was used to obtain Π for the various polymers from
, where k′ is the apparent first-order reaction rate constant; a, the persistence length of the polymer (3.5 Å) (Rex et al., 1998; Kienberger et al., 2000); D, the diameter of the transmembrane barrel; k, the second-order reaction rate constant; and [PEG]sol, the PEG concentration in the bulk aqueous phase. The slope of a plot of ln k′ versus N was used to obtain (a/D)5/3, which was used to obtain Π for the various polymers from 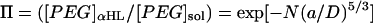 .
.
Mn values are given for the MePEG part of the molecules only.
These values are from Movileanu and Bayley (2001).
