Abstract
In a recent report, [Zhang et al. (2003) N. Engl. J. Med. 348, 203–213], the presence of CD3+ tumor-infiltrating lymphocytes (TILs) was found to correlate with improved survival in epithelial ovarian cancer. We performed immunohistochemical analysis for TILs and cancer testis antigens in 117 cases of epithelial ovarian cancer. The interrelationship between subpopulations of TILs and expression of cancer testis antigens was investigated, as well as between TILs and overall survival. The median follow-up of the patients was 31 months. Patients with higher frequencies of intraepithelial CD8+ T cells demonstrated improved survival compared with patients with lower frequencies [median = 55 versus 26 months; hazard ratio = 0.33; confidence interval (C.I.) = 0.18–0.60; P = 0.0003]. No association was found for CD3+ TILs or other subtypes of intraepithelial or stromal TILs. However, the subgroups with high versus low intraepithelial CD8+/CD4+ TIL ratios had median survival of 74 and 25 months, respectively (hazard ratio = 0.30; C.I. = 0.16–0.55; P = 0.0001). These results indicate that CD4+ TILs influence the beneficial effects of CD8+ TIL. This unfavorable effect of CD4+ T cells on prognosis was found to be due to CD25+forkhead box P3 (FOXP3)+ regulatory T cells (Treg; suppressor T cells), as indicated by survival of patients with high versus low CD8+/Treg ratios (median = 58 versus 23 months; hazard ratio = 0.31; C.I. = 0.17–0.58; P = 0.0002). The favorable prognostic effect of intraepithelial CD8+ TILs did not correlate with concurrent expression of NY-ESO-1 or MAGE antigens. We conclude that intraepithelial CD8+ TILs and a high CD8+/Treg ratio are associated with favorable prognosis in epithelial ovarian cancer.
Keywords: cancer testis antigen, CD8+ T cell
Several attempts have been made to evaluate the prognostic significance of tumor-infiltrating lymphocytes (TILs) in human cancers, and pronounced lymphocytic infiltration has been shown to be a prognostic parameter for better survival (1–5). Zhang et al. (6) recently showed that, in epithelial ovarian cancer (EOC), the presence of intratumoral CD3+ T cells was associated with improved survival. On the other hand, Curiel et al. (7) reported that tumor infiltration by a subpopulation of CD3+CD4+ T cells with immunosuppressive properties (suppressor or regulatory T cells, Treg) predicted reduced survival in EOC. Because the overall survival for patients with EOC remains poor despite improved diagnostic and treatment strategies (8, 9), these observations suggest that precise evaluation of the immune response at the primary tumor site could be useful for understanding the development and monitoring of immune therapies for this disease.
Over the past decade, there has been major progress in defining the targets for CD8+ and CD4+ T cell recognition of human cancers (10, 11). One category of tumor antigens, the cancer testis (CT) antigens, are promising targets for the development of human cancer vaccines (12). The defining characteristics of CT antigens are high expression levels in adult male germ cells, absence of expression in other normal adult tissues, and aberrant expression in a variable proportion of a wide range of different cancer types. Among CT antigens, NY-ESO-1 (13), MAGE-A1 (14), and MAGE-A4 (15) have been reported to be expressed frequently in EOC. The aims of the present study were to examine the significance of various subtypes of TILs in patients with ovarian cancer and to evaluate the relationship between TILs and CT antigen expression.
Materials and Methods
Patients and Specimens. Formalin-fixed, paraffin-embedded specimens were obtained from patients who underwent debulking surgery for EOC at Roswell Park Cancer Institute between 1995 and 2002. All tissue specimens were collected under an Institutional Review Board-approved protocol and were classified according to World Health Organization criteria. After surgery, all patients received platinum- and paclitaxel-based first-line chemotherapy for six cycles every 3 weeks. A complete response was defined by a normal physical examination, a normal computed tomographic scan of the abdomen and pelvis, and a normal serum CA-125 level (<35 units/ml). A partial response was defined by a decrease of at least 50% in the sum of the largest dimensions of tumors as measured by computed tomographic scanning. A smaller decrease or any increase in tumor size was considered to indicate a lack of response. Posttreatment surveillance consisted of bimonthly assessments by clinical examination, serum CA-125 measurements, and computed tomographic scan for patients with suspicion of recurrence. Overall survival was defined as the interval between diagnosis and death from ovarian cancer, or the interval between diagnosis and the last observation for surviving patients. No patient was lost to follow-up. Data were censored at the last follow-up for patients who were alive at the time of the analysis in January 2004.
Immunohistochemistry. The mouse monoclonal antibodies used were anti-human CD4 (clone 1F6, Neomarkers, Fremont, CA), anti-human CD8 (clone C8/144B, Neomarkers), anti-human CD20 (clone L26, DAKO), anti-human CD25 (clone 4C9, Vector Laboratories), anti-human NY-ESO-1 (clone ES121) (13), anti-human MAGE-A1 (clone MA454) (16), and anti-human MAGE-A4 (clone 57B) (17, 18). After deparaffinization, sections were immersed into preheated antigen-retrieval solution (DAKO high pH solution), incubated at 95°C for 20 min, and allowed to cool to room temperature. Dextran polymer (EnVision Plus, DAKO) was used as secondary antibody. For anti-human CD3 rabbit polyclonal antibody (DAKO), antigen retrieval was by proteinase digestion (Proteinase type VIII, Sigma-Aldrich). For anti-human forkhead box P3 (FOXP3) rabbit polyclonal antibody (Biolegend, San Diego), deparaffinized sections were immersed into preheated antigen-retrieval solution (DAKO high pH solution), incubated at 95°C for 20 min, and allowed to cool to room temperature. Endogenous peroxidase activity was blocked by 20-min incubation in 0.3% hydrogen peroxide and 0.1% sodium azide containing PBS. 3,3′-diamino-benzidine (BioGenex Laboratories, San Ramon, CA) was used as chromogen, and hematoxylin counterstain was performed. Negative control slides omitting the primary antibody were included in all assays. Triple immunohistochemistry using antibodies to NY-ESO-1, CD8, and CD20 were performed according to the protocol described by Lan et al. (19). Anti-human CD25, CD68, and FOXP3 were used for double immunohistochemistry. Details of the protocol are presented in Table 5, which is published as supporting information on the PNAS web site.
Quantification of TILs. Each entire tumor section was evaluated for TILs by using a ×20 objective lens, and 10 independent areas with the most abundant TILs were selected (5 cancer epithelium and 5 stromal areas), digitally photographed at a size of 0.0625 mm2, and counted manually. The count was performed three times for each photograph by the same investigator (E.S.) without knowledge of earlier results. The average of stromal or intraepithelial TIL counts for each patient was used for statistical analysis. Because CD68+ macrophages occasionally stain with CD4 antibody, CD4+ cells with apparent morphological appearance of macrophages were excluded from the count. For CD20+ TILs, the number of lymphoid aggregates was counted in a low-power (×10) field, and individual CD20+ cells in the absence of aggregation were counted in the same fashion as were T cells. Because intraepithelial CD20+ TILs were rare, only stromal infiltration was counted. For the assessment of CD25/FOXP3 double immunohistochemistry, CD25+ cells were counted first, followed by counts of FOXP3+CD25+ cells. The numbers of CD25+FOXP3+ T cells and CD25+FOXP3- TILs were used for analyses. CD25+FOXP3+ T cells were considered as representing Treg. Only intraepithelial TILs were examined by CD25/FOXP3 double immunohistochemistry. In addition, we calculated CD8+/CD4+ (CD8+ T cell count divided by CD4+ T cell count), CD8+/Treg, and CD8+/CD25+FOXP3- ratios. For CT antigens, sections with positive staining in >5% of cancer cells were defined as positive.
Statistical Analysis. Statistical analyses were performed with spss (SPSS, Chicago) and sas (SAS Institute, Cary, NC) software programs. Cumulative survival time was calculated by the Kaplan–Meier method and analyzed by the log-rank test. Univariate and multivariate Cox proportional hazards models were used to determine the hazard ratio that represents the relative risk of death among patients with higher frequencies of TILs compared with those with lower frequencies. Cutoff values are defined in Results. Two-sided P values <0.05 were judged to be significant.
Results
Study Population. The patients' characteristics are presented in Table 1. Of the 117 patients examined, 60 (51%) were dead before the end of observation period, and 58 of these patients died directly from their disease. The median duration of follow-up for the entire group was 30.6 months (range, 0.7–120.6 months). The median age of the study population was 62 years (range, 33–89 years). The majority of patients presented with grade-3 tumors (90%), at stage IIIc (74%), and with serous histology (78%). The median survival for all patients was 45 months [95% confidence interval (C.I.) = 33–58 months]. The 5-year survival for the entire study population was 13.7%.
Table 1. Patient characteristics.
| Characteristics | |
|---|---|
| No. of evaluable patients | 117 |
| Age, years | |
| Median | 62 |
| Range | 33–89 |
| Median overall survival,* months | 45 |
| FIGO stage | |
| 1A | 4 (4) |
| 1C | 1 (1) |
| IIB | 2 (2) |
| IIC | 5 (4) |
| IIIA | 1 (1) |
| IIIB | 3 (3) |
| IIIC | 87 (74) |
| IV | 12 (10) |
| Missing | 1 (1) |
| Histological type | |
| Papillary serous | 91 (78) |
| Clear cell | 6 (5) |
| Endometroid | 4 (3) |
| Mucinous | 4 (3) |
| Others (e.g., transitional, mixed) | 12 (10) |
| Grade | |
| 1 | 8 (7) |
| 2 | 4 (3) |
| 3 | 105 (90) |
| Response to frontline therapy | |
| Complete response | 56 (48) |
| Partial response | 57 (49) |
| Progression | 3 (3) |
| Unknown | 1 (1) |
| Residual tumor | |
| None | 55 (47) |
| Present | 56 (48) |
| Unknown | 6 (5) |
| Status | |
| Alive with no evidence of disease | 26 (22) |
| Alive with disease | 31 (27) |
| Dead of disease | 58 (50) |
| Dead with disease | 1 (1) |
| Dead with unknown disease status | 1 (1) |
| CT antigen expression | |
| NY-ESO-1 | 50 (43) |
| MAGE-A1 | 18 (15) |
| MAGE-A4 | 13 (11) |
Data in parentheses are percentages.
C.I. = 33–58 months
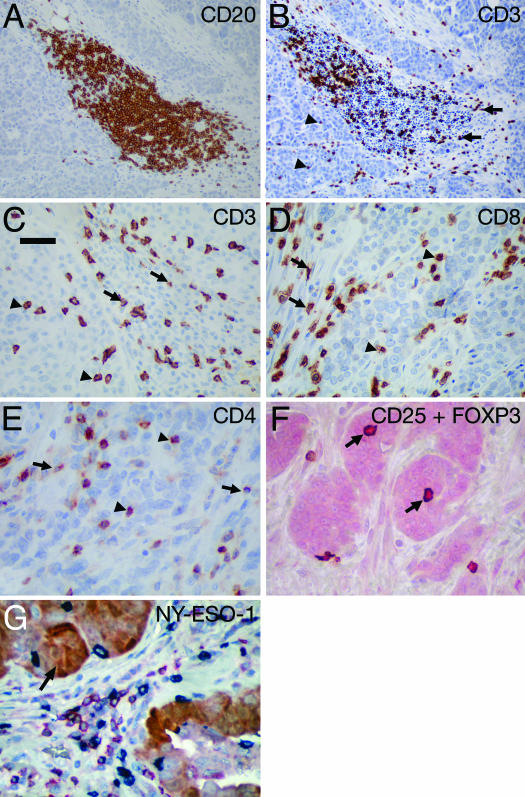
TILs in EOC. TILs were observed both in cancer stroma and within cancer epithelium (i.e., intraepithelial) except for CD20+ TILs, which were almost exclusively present in stroma and frequently in the form of lymphoid aggregates. Representative pictures of lymphocyte infiltration are shown in Fig. 1 A–F. Statistics of averaged TILs counts are shown in Table 2. The number of TILs positively correlated with each other for most subtypes, for both intraepithelial and stromal infiltration (range of correlation coefficient = 0.21–0.85; P < 0.001 for the majority of correlations) (Table 6, which is published as supporting information on the PNAS web site).
Fig. 1.
Representative pictures of lymphocyte infiltration in EOC. TILs were observed both in cancer stroma and within cancer epithelium (i.e., intraepithelial). (A) CD20+ TILs frequently form lymphoid aggregates in cancer stroma. (B) CD3+ TILs in a lymphoid aggregate. (Original magnification: ×20). (C) CD3+ TIL distribution in stromal and intraepithelial areas. (D and E) CD8+ (D) and CD4+ (E) TILs in stromal (arrow) and intraepithelial (arrowhead) areas. (F) Treg cells (CD25+FOXP3+) (arrow) were detected by double immunohistochemistry of CD25 (brown) and FOXP3 (red). (G) Triple immunohistochemistry showing heterogenous expression of NY-ESO-1 (brown/arrow) in ovarian cancer with the presence of CD8+ TILs (blue) and CD20+ TILs (red). (Scale bar, 50 μm.)
Table 2. Descriptive statistics for numbers of TILs.
| Cells | Mean* | Median* | Bottom tertile* | Variance | Range* | Skewness |
|---|---|---|---|---|---|---|
| Stromal CD3+ TILs | 60.20 | 51.90 | 34.60 | 1,802.00 | 3–163.9 | 0.55 |
| Stromal CD4+ TILs | 31.60 | 25.10 | 16.30 | 575.0 | 1.7–128.6 | 1.24 |
| Stromal CD8+ TILs | 37.20 | 35.10 | 23.40 | 606.30 | 0–135.3 | 0.81 |
| Stromal CD20+ TILs | 19.0 | 12.10 | 5.20 | 393.70 | 0–80.5 | 1.19 |
| Intraepithelial CD3+ TILs | 11.0 | 8.00 | 4.50 | 110.60 | 0–64.1 | 2.13 |
| Intraepithelial CD4+ TILs | 3.40 | 2.30 | 1.70 | 17.0 | 0.2–27.0 | 3.73 |
| Intraepithelial CD8+ TILs | 13.20 | 7.60 | 3.30 | 222.80 | 0–89.5 | 2.09 |
| Intraepithelial Treg | 1.01 | 0.67 | 0.40 | 1.26 | 0–5.4 | 1.96 |
| Intraepithelial CD25+FOXP3– TILs | 2.12 | 1.27 | 0.67 | 6.25 | 0–14.09 | 2.24 |
| Stromal CD8/CD4 ratio† | 1.60 | 1.20 | 0.96 | 1.60 | 0–7.3 | 1.74 |
| Intraepithelial CD8/CD4 ratio† | 4.70 | 3.10 | 1.94 | 20.30 | 0–20.3 | 1.53 |
| Intraepithelial CD8/Treg ratio‡ | 18.75 | 9.16 | 4.20 | 593.92 | 0–131.7 | 2.24 |
| Intraepithelial CD25+FOXP3– ratio‡ | 13.00 | 4.29 | 2.60 | 815.30 | 0–246.5 | 5.94 |
Data are cells counted per field
One case and 6 cases of 117 were excluded for the calculation of stromal and intraepithelial CD8/CD4 ratio, respectively
Nineteen cases and 8 cases of 117 were excluded for CD8/Treg ratio and CD8/CD25+FOXP3– ratio, respectively
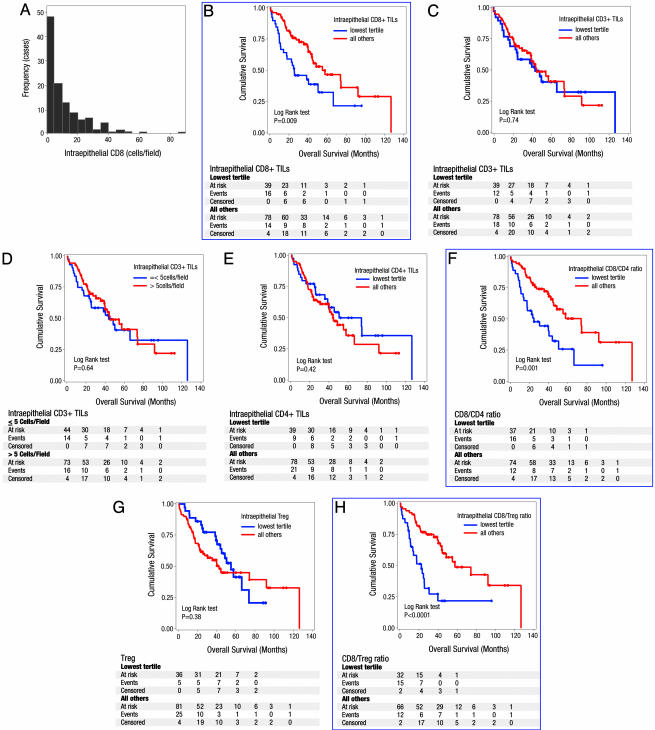
Prognostic Significance of TILs. The distribution of TILs was positively skewed (i.e., did not show a normal distribution curve, Table 2), and Fig. 2A shows the distribution for intraepithelial CD8+ TILs with a heavier left than right tail of the curve. In consideration of this asymmetric distribution, we adopted a classification by tertile (33rd percentile) to compare the lowest tertile subgroup (group 1) to all others (group 2). Five clinicopathological variables (age, stage, grade, histological type, and presence of residual disease) were adopted as covariates for multivariate Cox proportional hazards model because of their relative prognostic significance by univariate Cox proportional hazards model (Table 3) and in previous studies (20, 21). The subgroup with the lowest frequency of intraepithelial CD8+ TILs (≤3.3; see Table 2) consistently showed poorer survival by univariate and multivariate analysis (Fig. 2B and Tables 3 and 4). Median survival for the patients in the lowest tertile of intraepithelial CD8+ T cell was 26 months, whereas other patients had a median survival of 55 months (hazard ratio = 0.33; C.I. = 0.18–0.60; P = 0.0003 for multivariate analysis; see Table 4).
Fig. 2.
Prognostic significance of TILs in ovarian cancer. (A) Frequency distribution of intraepithelial CD8+ TILs showing a heavier left than right tail of the curve (i.e., did not show a normal distribution curve). The x axis shows the average number of intraepithelial CD8+ TILs, and the y axis shows the number of cases in each class. Cumulative survival time was calculated by the Kaplan–Meier method and analyzed by the log-rank test. High frequency of intraepithelial CD8+ TILs was associated with improved survival (B), whereas the presence or absence of intraepithelial CD3+ TILs was not associated with improved survival using the tertile classification (C) or Zhang et al's (6) classification (D). Intraepithelial CD4+ TILs were also not associated with improved survival (E), but a high intraepithelial CD8+/CD4+ ratio was associated with improved overall survival (F). Treg were not prognostic for survival (G), but a high intraepithelial CD8+/Treg ratio was found to be associated with improved survival (H). Significant results are boxed in blue.
Table 3. Univariate analysis of overall survival.
| Variable | Hazard ratio | 95% C.I. | P |
|---|---|---|---|
| Age, years (continuous) | 1.03 | 1.01–1.05 | 0.01 |
| Stage I/II vs. III/IV | 2.72 | 0.85–8.72 | 0.09 |
| Grade 1/2 vs. 3 | 1.26 | 0.50–3.14 | 0.63 |
| Histology, serous vs. others | 0.66 | 0.32–1.34 | 0.25 |
| Residual tumor, absent vs. present | 1.20 | 0.71–2.05 | 0.50 |
| Stromal TILs (lowest tertile vs. all others) | |||
| CD3+ | 0.70 | 0.40–1.20 | 0.19 |
| CD4+ | 0.89 | 0.51–1.56 | 0.68 |
| CD8+ | 0.71 | 0.41–1.22 | 0.22 |
| CD8+/CD4+ ratio | 1.23 | 0.70–2.16 | 0.48 |
| CD20+ | 0.77 | 0.45–1.32 | 0.34 |
| Intraepithelial TILs (lowest tertile vs. all others) | |||
| CD3+ | 0.92 | 0.54–1.55 | 0.74 |
| CD3+ (according to Zhang et al. in ref. 6) | 0.89 | 0.53–1.49 | 0.64 |
| CD4+ | 0.84 | 0.50–1.40 | 0.50 |
| CD8+ | 0.51 | 0.30–0.85 | 0.01 |
| CD8+/CD4+ ratio | 0.42 | 0.25–0.72 | 0.002 |
| Treg | 1.28 | 0.74–2.24 | 0.38 |
| CD25+FOXP3– | 0.61 | 0.35–1.04 | 0.07 |
| CD8+/Treg ratio | 0.33 | 0.18–0.59 | 0.0002 |
| CD8+/CD25+FOXP3– ratio | 0.98 | 0.56–1.71 | 0.94 |
Univariate Cox proportional hazards model. Whereas high intraepithelial CD8+ TILs, CD8+/CD4+ ratio, and CD8+/Treg ratio were associated with improved survival, stromal TIL subtypes and other intraepithelial TILs were not significant. Age, stage, grade, histological type, and presence of residual disease were adopted as covariates. Classification of CD3+ TILs by Zhang et al. (6) was applied but was not associated with improved survival.
Table 4. Median survival and multivariate Cox proportional hazards model.
| Median survival, months
|
|||||
|---|---|---|---|---|---|
| Cells | Lowest tertile | All others | Hazard ratio | 95% C.I. | P |
| Intraepithelial CD8+ TILs | 25.5 | 54.9 | 0.33 | 0.18–0.60 | 0.0003 |
| Intraepithelial CD3+ TILs | 43.9 | 54.9 | 0.71 | 0.36–1.29 | 0.26 |
| CD3+ TILs using Zhang et al.'s classification (6) | 43.9 | 45.2 | 0.72 | 0.41–1.26 | 0.27 |
| Intraepithelial CD8+/CD4+ ratio | 24.7 | 73.8 | 0.30 | 0.16–0.55 | 0.0001 |
| Intraepithelial Treg | 54.9 | 42.4 | 1.31 | 0.72–2.41 | 0.38 |
| Intraepithelial CD8+/Treg ratio | 22.6 | 57.6 | 0.31 | 0.17–0.58 | 0.0002 |
| Intraepithelial CD25+FOXP3– | 39.3 | 57.6 | 0.64 | 0.37–1.11 | 0.11 |
| Intraepithelial CD8/CD25+FOXP3–2 ratio | 48.6 | 44.4 | 0.88 | 0.47–1.66 | 0.69 |
Age, stage, grade, histological type, and presence of residual disease were adopted as covariates. Classification of CD3+ TILs by Zhang et al. (6) was applied but was not associated with improved survival.
CD3+ TILs were not prognostic for survival by the tertile classification (Fig. 2C and Tables 3 and 4). We adopted the classification used by Zhang et al. (6) to classify CD3+ TILs into present or absent based on no more than five cells per high-power field. We first applied this classification to the sum of stromal and intraepithelial CD3+ TILs because these tumor compartments were not considered separately by Zhang et al. (6). This classification resulted in only 4 cases in the CD3- group and 113 cases in the CD3+ group. Applying the classification solely to intraepithelial CD3+ TILs resulted in 44 CD3- cases and 73 CD3+ cases, and improved survival was still not demonstrable for the CD3+ group (Fig. 2D and Tables 3 and 4). The median survival for CD3+ and CD3- patients was 45.2 and 43.9 months, respectively (hazard ratio = 0.72; C.I. = 0.41–1.28; P = 0.27). In the study by Zhang et al. (6), the median survival was 50.3 and 18 months for the CD3+ and CD3- groups, respectively.
Neither intraepithelial nor stromal CD4+ TILs were associated with improved survival (Fig. 2E and Table 3). However, the subgroup of patients with high intraepithelial CD8+/CD4+ ratios (>1.94; see Table 2) demonstrated improved survival (hazard ratio = 0.30; C.I. = 0.16–0.55; P = 0.0001) (Fig. 2F and Tables 3 and 4). This result stresses the importance of CD4+ TILs in modulating the beneficial prognostic influence of CD8+ TILs on survival. Therefore, we next examined the contribution of Tregs (suppressor T cells) by CD25/FOXP3 double immunohistochemistry (Fig. 1F). Although the IL-2 receptor α, CD25, is considered a marker of suppressor T cells, it is also expressed on activated T cells. Recently, FOXP3 has been shown to be a marker for T cells with regulatory activity (22, 23). Suppressor T cells (Treg) were defined as CD25+FOXP3+ T cells in this study, and CD25+FOXP3- cells were also evaluated. Although patients with intraepithelial Treg infiltration had shorter median survival (42.4 months) than did those without Treg (54.9 months), the difference was not significant (Fig. 2G and Tables 3 and 4). However, the median survival for patients with high CD8+/Treg ratios (>4.2, Table 2) was 57.6 months, whereas patients with low ratios had a median survival of 22.6 months (hazard ratio = 0.31; C.I. = 0.17–0.58; P = 0.0002; see Fig. 2H and Tables 3 and 4).
There were no significant differences in survival when patients were classified based on the degree of intraepithelial CD25+FOXP3- TILs (Tables 3 and 4) or CD8/CD25+FOXP3- ratio (Tables 3 and 4). Details of the multivariate Cox proportional hazards model are presented in Table 7, which is published as supporting information on the PNAS web site. Together, the results indicate that patients with low intraepithelial CD8+ TILs, and low ratios of CD8+/CD4+ and CD8+/Treg, consistently demonstrated the lowest overall survival. All of these results remained significant after adjusting for multiple testing. In an alternative analysis, individuals were divided into two equal groups (low and high) by using the median values of TILs in Table 2 as cutoff. Consistent with the tertile classification, high intraepithelial CD8+/CD4+ ratios and high CD8+/Treg ratios were still associated with improved survival (Table 8, which is published as supporting information on the PNAS web site), underlining the adverse effect of Tregs on the beneficial prognostic influence of intraepithelial CD8+ TILs.
Response to chemotherapy was found to be associated with improved survival in multivariate analyses (data not shown). Therefore, we examined its association with TILs. The results indicate that TILs and response to chemotherapy are independent variables. When we included response to chemotherapy as a covariate in multivariate analyses, hazard ratios and C.I.s for the prognostic significance of all TIL subtypes did not change significantly.
TILs and Concurrent Expression of CT Antigens. NY-ESO-1, MAGE-A1, and MAGE-A4 expression were observed in 43% (50 cases), 15% (18 cases), and 11% (13 cases) of 117 samples (Table 1 and Fig. 1G). The relationship between CT antigen expression and TILs was analyzed in the patient population. We found no significant association between TILs in relation to CT antigen expression and survival. The detailed description of these analyses is shown in Table 9, which is published as supporting information on the PNAS web site.
Discussion
The presence of TILs within the tumor microenvironment is considered to be an indication of the host immune response to tumor antigens (1–5) and is thought to reflect the dynamic process of “cancer immunoediting” (24). In the present study, we performed a detailed immunohistochemical evaluation of TILs in EOC. Although most TIL subtypes positively correlated with each other, intraepithelial CD8+ TIL was the only subtype that was associated with improved survival. All of these analyses were confirmed by multivariate statistics using previously established clinicopathologic factors (20, 21).
There are important differences between the results of the present study and a recent report by Zhang et al. (6). The authors examined 174 Italian patients with EOC, and the presence of CD3+ TILs was defined as more than five T cells per high-power field. First, using this classification, we were unable to demonstrate the association between CD3+ TILs and improved survival of ovarian cancer patients. Second, although the report by Zhang et al. (6) indicated that CD3+ TILs both within tumor islets and peritumoral stroma were counted, a distinction was not made regarding the precise location of TILs that were associated with improved survival. In the current study, stromal TILs of all subtypes did not correlate with improved survival in ovarian cancer as reported in other malignancies (2–4). Our results underline the importance of assessing the precise in situ localization of TILs for each tumor type.
Our results clearly indicate that intraepithelial CD8+ TILs were the only subtype associated with favorable prognosis in EOC; this finding raises the important question of why there are differences between the current study and the report of Zhang et al. (6). One possibility is the differences in chemotherapeutic regimens in the two studies. In Zhang et al.'s study (6), patients were treated between 1991 and 1995 with a combination of platinum and/or cyclophosphamide and/or Adriamycin, and between 1995 and 1999 with platinum plus paclitaxel, as in the current study. However, differences in overall response rates to chemotherapy are modest in the two studies. There are also methodologic differences between the two studies. Zhang et al. (6) applied anti-CD3 monoclonal antibody on fresh frozen specimens, whereas rabbit polyclonal anti-CD3 antibody on archived formalin-fixed, paraffin-embedded specimens was used in this study. In addition, for TIL counting, Zhang et al. (6) selected 15–20 areas of high-power field and adopted manual counting (or computerized counting on captured photograph), whereas we selected 10 areas (5 stromal and 5 intraepithelial areas) of abundant TIL infiltration for photography, followed by manual counting. Despite these methodologic issues, we suggest that an important explanation for the discrepancy in results is the limited evaluation of CD4+ TILs by Zhang et al. (6). In the present study, we have shown that a high intraepithelial CD8+/CD4+ T cell ratio results in improved survival in EOC. In contrast, neither intraepithelial CD4+ nor CD3+ TILs alone were associated with survival. In a previous study in colon cancer, a high CD8+/CD4+ T cell ratio was also associated with improved survival (25). This finding indicates that CD4+ TILs influence the prognostic significance of CD8+ TILs and implies that a subpopulation of CD4+ T cells with immunosuppressive properties might modulate the beneficial antitumor effects of CD8+ effector cells.
Consistent with our observations, CD4+CD25+ suppressor T cells (Treg) (26–28), a population of CD4+ T cells with immunosuppressive properties, have been shown to be increased in human ovarian cancer (29) and to be associated with reduced survival (7). We have directly examined intraepithelial Treg infiltration in the current study by detection of FOXP3 (27, 28) expression among CD25+ TILs. Our results indicate that intraepithelial Treg infiltration alone was not associated with worse survival, as reported by Curiel et al. (7), probably a reflection of sample size and methodologic differences. However, we demonstrated that a high intraepithelial CD8+/Treg ratio was associated with improved survival in EOC, corresponding to an almost 70% reduction in the risk of death. We also examined the subpopulation of CD25+FOXP3- TILs, a population considered to be activated T cells, composed mainly of CD4+ helper T and CD8+ T cells, and we found that the intraepithelial CD8+/CD25+FOXP3- ratio was not prognostic for survival. Taken together with the findings of Curiel et al. (7), these results indicate that the modulating influence of CD4+ T cells on the beneficial effects of CD8+ T cells can be ascribed to the number of Tregs in the CD4+ T cell population. Recent evidence indicates that cyclophosphamide reduces the number of Tregs (30, 31), and it is possible that the inclusion of this chemotherapeutic agent in the regimen used to treat patients studied by Zhang et al. (6) contributed to the discrepancies between the current study and the report by Zhang et al. (6).
Our data did not show any relationship between intraepithelial CD8+ TILs and expression of NY-ESO-1 or the MAGE antigens examined. Although spontaneous immune responses to NY-ESO-1 are found in EOC patients (13), it is likely to be only one of many immunogenic antigens. The correlation between the relatively small numbers of TILs that may be specific for each CT antigen and survival could be difficult to demonstrate by statistical methods.
In conclusion, our results demonstrate that the presence of intraepithelial CD8+ TILs and high CD8+/CD4+, CD8+/Treg ratios are associated with improved survival in ovarian cancer. Expanded analyses of immunological parameters at the tumor site, including typing for a larger panel of tumor antigens, antigen specificity of the infiltrating lymphocytes, and MHC expression by the tumor cells will be required to understand the nature and role of TILs in ovarian cancer.
Supplementary Material
Acknowledgments
This work was supported by a Cancer Research Institute/Ludwig Institute for Cancer Research Cancer Vaccine Collaborative Grant. K.O. is supported by the Cancer Research Institute's Anna-Marie Kellen Clinical Investigator Award.
Author contributions: E.S., L.J.O., and K.O. designed research; E.S., H.N., Y.-T.C., F.Q., A.A.J., D.F., S.G., T.O., G.R., K.O., and L.J.O. performed research; S.H.O., J.A., B.B., C.A., J.K., S.L., H.O., E.S., K.O., and L.J.O. analyzed data; and E.S., L.J.O., and K.O. wrote the paper.
Conflict of interest statement: No conflicts declared.
Abbreviations: TIL, tumor-infiltrating lymphocyte; CT, cancer testis; Treg, regulatory T cell; EOC, epithelial ovarian cancer; C.I., confidence interval.
References
- 1.Clemente, C. G., Mihm, M. C., Jr., Bufalino, R., Zurrida, S., Collini, P. & Cascinelli, N. (1996) Cancer 77, 1303-1310. [DOI] [PubMed] [Google Scholar]
- 2.Jass, J. R. (1986) J. Clin. Pathol. 39, 585-589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Naito, Y., Saito, K., Shiiba, K., Ohuchi, A., Saigenji, K., Nagura, H. & Ohtani, H. (1998) Cancer Res. 58, 3491-3494. [PubMed] [Google Scholar]
- 4.Schumacher, K., Haensch, W., Roefzaad, C. & Schlag, P. M. (2001) Cancer Res. 61, 3932-3936. [PubMed] [Google Scholar]
- 5.Nakano, O., Sato, M., Naito, Y., Suzuki, K., Orikasa, S., Aizawa, M., Suzuki, Y., Shintaku, I., Nagura, H. & Ohtani, H. (2001) Cancer Res. 61, 5132-5136. [PubMed] [Google Scholar]
- 6.Zhang, L., Conejo-Garcia, J. R., Katsaros, D., Gimotty, P. A., Massobrio, M., Regnani, G., Makrigiannakis, A., Gray, H., Schlienger, K., Liebman, M. N., et al. (2003) N. Engl. J. Med. 348, 203-213. [DOI] [PubMed] [Google Scholar]
- 7.Curiel, T. J., Coukos, G., Zou, L., Alvarez, X., Cheng, P., Mottram, P., Evdemon-Hogan, M., Conejo-Garcia, J. R., Zhang, L., Burow, M., et al. (2004) Nat. Med. 10, 942-949. [DOI] [PubMed] [Google Scholar]
- 8.McGuire, W. P., Hoskins, W. J., Brady, M. F., Kucera, P. R., Partridge, E. E., Look, K. Y., Clarke-Pearson, D. L. & Davidson, M. (1996) Semin. Oncol. 23, 40-47. [PubMed] [Google Scholar]
- 9.Vaidya, A. P. & Curtin, J. P. (2003) Semin. Oncol. 30, 401-412. [DOI] [PubMed] [Google Scholar]
- 10.Boon, T., Cerottini, J. C., Van den Eynde, B., van der Bruggen, P. & Van Pel, A. (1994) Annu. Rev. Immunol. 12, 337-365. [DOI] [PubMed] [Google Scholar]
- 11.Old, L. J. & Chen, Y. T. (1998) J. Exp. Med. 187, 1163-1167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Scanlan, M. J., Gure, A. O., Jungbluth, A. A., Old, L. J. & Chen, Y. T. (2002) Immunol. Rev. 188, 22-32. [DOI] [PubMed] [Google Scholar]
- 13.Odunsi, K., Jungbluth, A. A., Stockert, E., Qian, F., Gnjatic, S., Tammela, J., Intengan, M., Beck, A., Keitz, B., Santiago, D., et al. (2003) Cancer Res. 63, 6076-6083. [PubMed] [Google Scholar]
- 14.Gillespie, A. M., Rodgers, S., Wilson, A. P., Tidy, J., Rees, R. C., Coleman, R. E. & Murray, A. K. (1998) Br. J. Cancer 78, 816-821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yakirevich, E., Sabo, E., Lavie, O., Mazareb, S., Spagnoli, G. C. & Resnick, M. B. (2003) Clin. Cancer Res. 9, 6453-6460. [PubMed] [Google Scholar]
- 16.Chen, Y. T., Stockert, E., Chen, Y., Garin-Chesa, P., Rettig, W. J., van der Bruggen, P., Boon, T. & Old, L. J. (1994) Proc. Natl. Acad. Sci. USA 91, 1004-1008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kocher, T., Schultz-Thater, E., Gudat, F., Schaefer, C., Casorati, G., Juretic, A., Willimann, T., Harder, F., Heberer, M. & Spagnoli, G. C. (1995) Cancer Res. 55, 2236-2239. [PubMed] [Google Scholar]
- 18.Landry, C., Brasseur, F., Spagnoli, G. C., Marbaix, E., Boon, T., Coulie, P. & Godelaine, D. (2000) Int. J. Cancer 86, 835-841. [DOI] [PubMed] [Google Scholar]
- 19.Lan, H. Y., Mu, W., Nikolic-Paterson, D. J. & Atkins, R. C. (1995) J. Histochem. Cytochem. 43, 97-102. [DOI] [PubMed] [Google Scholar]
- 20.Holschneider, C. H. & Berek, J. S. (2000) Semin. Surg. Oncol. 19, 3-10. [DOI] [PubMed] [Google Scholar]
- 21.Engel, J., Eckel, R., Schubert-Fritschle, G., Kerr, J., Kuhn, W., Diebold, J., Kimmig, R., Rehbock, J. & Holzel, D. (2002) Eur. J. Cancer 38, 2435-2445. [DOI] [PubMed] [Google Scholar]
- 22.Hori, S., Nomura, T. & Sakaguchi, S. (2003) Science 299, 1057-1061.12522256 [Google Scholar]
- 23.Fontenot, J. D., Gavin, M. A. & Rudensky, A. Y. (2003) Nat. Immunol. 4, 330-336. [DOI] [PubMed] [Google Scholar]
- 24.Dunn, G. P., Old, L. J. & Schreiber, R. D. (2004) Immunity 21, 137-148. [DOI] [PubMed] [Google Scholar]
- 25.Diederichsen, A. C., Hjelmborg, J. B., Christensen, P. B., Zeuthen, J. & Fenger, C. (2003) Cancer Immunol. Immunother. 52, 423-428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sakaguchi, S., Sakaguchi, N., Asano, M., Itoh, M. & Toda, M. (1995) J. Immunol. 155, 1151-1164. [PubMed] [Google Scholar]
- 27.Sakaguchi, S. (2004) Annu. Rev. Immunol. 22, 531-562. [DOI] [PubMed] [Google Scholar]
- 28.Shevach, E. M. (2002) Nat. Rev. Immunol. 2, 389-400. [DOI] [PubMed] [Google Scholar]
- 29.Woo, E. Y., Chu, C. S., Goletz, T. J., Schlienger, K., Yeh, H., Coukos, G., Rubin, S. C., Kaiser, L. R. & June, C. H. (2001) Cancer Res. 61, 4766-4772. [PubMed] [Google Scholar]
- 30.Lutsiak, M. E., Semnani, R. T., De Pascalis, R., Kashmiri, S. V., Schlom, J. & Sabzevari, H. (2005) Blood 105, 2862-2868. [DOI] [PubMed] [Google Scholar]
- 31.Ghiringhelli, F., Larmonier, N., Schmitt, E., Parcellier, A., Cathelin, D., Garrido, C., Chauffert, B., Solary, E., Bonnotte, B. & Martin, F. (2004) Eur. J. Immunol. 34, 336-344. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.