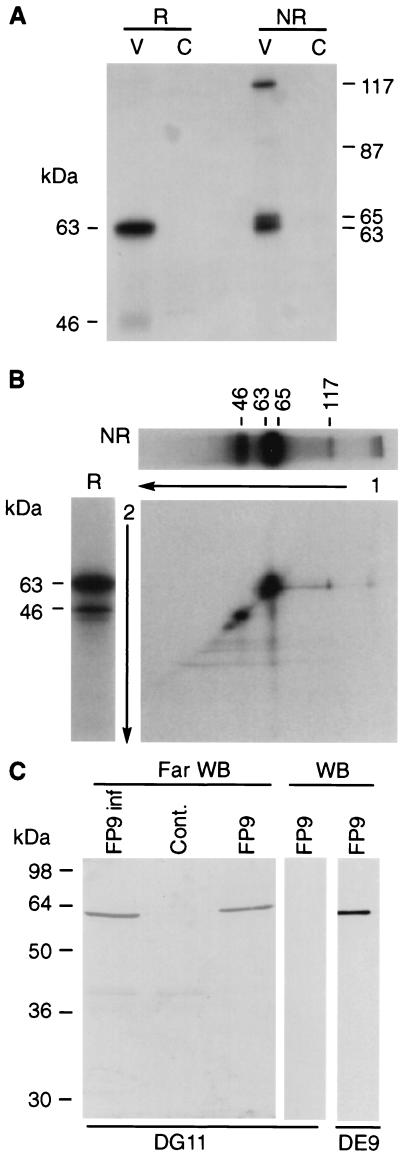
FIG. 2.
(A) Radioimmunoprecipitation of the 63-kDa protein under reducing (R) and nonreducing (NR) conditions. V, FP9 infected cell lysate; C, noninfected cellular control. (B) 2D electrophoresis. The 63-kDa protein was first immunoprecipitated and run on a gel under nonreducing conditions, as in panel A, lane 3. The corresponding lane was excised (a duplicate well is shown horizontally on top of the 2D gel), treated with β-mercaptoethanol, inserted on top of a second gel devoid of wells, and run under reducing conditions (a control sample run treated under reducing conditions is shown on the left of the 2D gel). (C) Far-Western blotting. Purified virus (FP9) and clarified lysates from infected (FP9 inf) and uninfected (Cont.) cells were subjected to SDS-PAGE and then transferred onto a nitrocellulose membrane. The membrane (Far WB) was saturated overnight at 4°C in 5% powdered milk in TN buffer (10 mM Tris, pH 7.5, and 500 mM NaCl) and then probed overnight at 4°C with FP9-infected cellular lysate. MAb DG11, directed against the 63-kDa protein, was then added as for a conventional Western blotting experiment (control Western blots [WB] are shown for DG11 and DE9). The 63-kDa protein used as a probe in the cellular lysate could be distinguished from that run on the gel and transferred to the membrane by revealing the far-Western blot with MAb DG11, which does not recognize the denatured protein (Table 1; also compare lanes 3 and 4). In contrast, DE9 recognizes both forms of the protein.