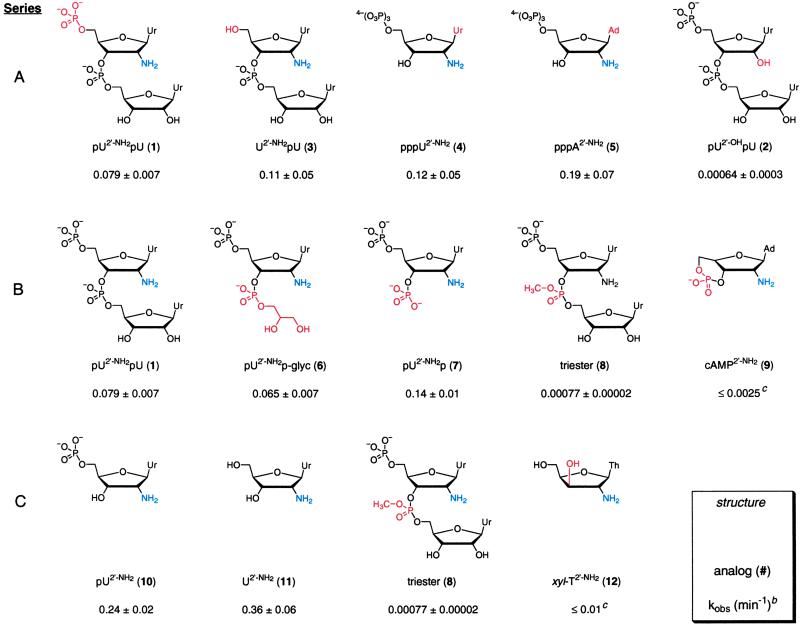
Table 1.
RNA analog structures and acylation ratesa
The amine nucleophile is blue, and structural variation in a series is highlighted in red. Ur, uracil; Ad, adenine; Th, thymine; U, uridine; A, adenosine; T, thymidine; P, phosphate; glyc, glyceryl. For clarity, some analogs are included in more than one series.
kobs are reported at 20 mM succinimidyl ester.
Rate is an upper limit; no product formation was detected. Two unreactive analogs, cAMP2′-OH and U2′-OH, were also tested but are not shown (kobs ≤ 0.002).