Abstract
Objectives: Metabolic complications have been associated with HIV-1 infection and with long-term use of antiretroviral (ARV) medications. In some studies, such complications have been linked to cardiovascular events, yet limited data exist concerning metabolic complications and dementia. The objective of this study was to examine the relationship between HIV-associated dementia (HAD) and diabetes among patients with HIV-1 infection.
Design: Cross-sectional analysis of entry data for a longitudinal cohort study.
Methods: A total of 203 participants who were enrolled in the Hawaii Aging with HIV Cohort between October 2001 and November 2003 served as the study population. Research case definitions of HAD were determined in consensus conferences by a panel that included neurologists, neuropsychologists, and a geriatrician. Diabetes was determined by self-report or a fasting glucose level >125 mg/dL.
Results: Participants' ages ranged between 20–76 years at enrollment with approximately one-half aged ≥50 years. After adjustment for important covariates including age, education, ethnicity, CD4 lymphocyte count, duration of HIV infection, and protease inhibitor–based ARV therapy, we found a statistically significant association of diabetes with HAD (odds ratio 5.43, 1.66–17.70). A significant association remained after adjustment for other vascular risk factors. Among participants without diabetes, fasting glucose levels were higher with increasing impairment category.
Conclusions: Within the Hawaii Aging with HIV Cohort, a longitudinal study enriched with older HIV-1–infected individuals, diabetes is associated with prevalent dementia. This finding is not fully explained by age or coexisting vascular risk factors. Evaluation of underlying mechanisms is warranted.
Keywords: dementia, diabetes mellitus, insulin resistance, HIV-1
Among HIV-1–infected individuals, both HIV infection itself and the use of antiretroviral (ARV) medications contribute to a greater incidence of metabolic abnormalities known to be risk factors for cardiovascular events. Studies conducted before the widespread use of ARVs suggest that, while glucose homeostasis is not markedly affected, HIV-1 infection is associated with a proatherogenic lipid profile characterized by an increase in triglyceride levels, a decrease in HDL cholesterol levels, and the presence of small, dense low-density lipoprotein (LDL) particles.1-3 The addition of ARV medications, particularly the use of protease inhibitors (PIs), has had further deleterious effects on metabolic risk factors in this already vulnerable population. Specifically, among HIV-1–seropositive patients, the initiation of PI-based ARV therapy is associated with the development of insulin resistance in 25–62% and the development of overt new-onset diabetes mellitus in 6–7%.4-6 Increases in LDL cholesterol and triglyceride levels following ARV therapy have also been observed.7,8
The presence of such metabolic complications correlates with both increased risk of cerebrovascular disease and an increased rate of dementia among seronegative populations.9 This raises concerns that the increased prevalence of metabolic abnormalities currently observed in the HIV-1–infected population may not only increase the risk for future vascular complications but may also contribute to a greater risk for the development of neurocognitive abnormalities. While still controversial, a corresponding increase in cardiovascular events has been described in some studies; however, our understanding of a potential correlation to cerebrovascular events and dementia among HIV-1–infected patients is incomplete.10-13
We have recently reported findings from the Hawaii Aging with HIV Cohort suggesting that older age is associated with a higher risk of dementia among HIV-1–infected individuals.14 The mean duration of infection among our older participants is nearly twice that of our younger participants; however, neither duration of infection itself nor age alone appears to be sufficient to explain our findings. We hypothesize that prolonged infection, extended exposure to ARV, and thus, greater likelihood of metabolic abnormalities may contribute to the observed increase in cognitive abnormalities. Thus, a cohort of older HIV-1–infected individuals may be at greatest risk for these clinically significant cognitive complications. In such a model, younger individuals may have early or subclinical disease that is too mild to result in a measurable cognitive effect. To test this hypothesis, we examined whether diabetes, fasting glucose, and insulin resistance contribute to the relative risk of HIV-associated dementia (HAD) among HIV-1–infected participants in the Hawaii Aging with HIV Cohort.
METHODS
Subject Population Studied
The Hawaii Aging with HIV Cohort is a longitudinal prospective cohort study designed to investigate the fundamental neuroepidemiology of HIV infection in older individuals. Briefly, ongoing enrollment of the cohort began in October 2001 to compare older (≥50 years) and younger (20–40 years) HIV-1–infected individuals. Participants are recruited from all islands of Hawaii using broad community-based methods to maximize representation of the HIV-1–seropositive population living in Hawaii. To date, the basic demographic parameters, including the distribution of participants by island, are roughly similar to those reported to the Hawaii State Department of Health for the HIV-1–infected population.
Exclusion criteria include major neurologic or psychiatric illness, learning disability, major head injury, brain opportunistic infection, and primary language other than English. Several individuals with transient ischemic attack or minor stroke with near full resolution of findings have been allowed to enroll in the cohort; however, these participants were excluded from the current analysis (n = 3 older and 1 younger). While all participants must be living in Hawaii at the time of enrollment, 62% reported being born and raised on the continental United States and, on average, each participant has spent only 41% of their lives in Hawaii. This current analysis was based on available cases for entry assessments of participants enrolled through November 2003.
Neurocognitive Assessments
Baseline evaluations included an 80-minute neuropsychological test battery assessing visual and verbal memory, visuospatial and executive functioning, motor/psychomotor speed, working memory, attention/concentration, and information processing. This battery closely matches that used in the Northeast AIDS Dementia (NEAD) cohort.15 Published age and education-adjusted normative data were used to calculate z-scores used in clinical characterization. A neurologic examination (macroneurologic examination as used in the Adult AIDS Clinical Trials Group) with emphasis on motor speed and extrapyramidal signs was also completed. Individual cases were discussed weekly in collaboration with colleagues at Johns Hopkins University in a consensus conference involving 2 neurologists, 2 neuropsychologists, and a geriatrician. Research case definitions of cognitive status—HAD, Minor Cognitive Motor Disorder (MCMD), neuropsychological (NP) testing abnormality not meeting MCMD or HAD criteria—were assigned based on American Academy of Neurology criteria and using all clinical data obtained during the study visit. Panel members were not blinded to any clinical data including diabetes status.16 A full description of this diagnostic process is provided elsewhere.14
In general, a diagnosis of HAD required the following: an abnormality in at least 2 cognitive domains including attention/concentration, speed of information processing, abstraction/reasoning, visuospatial skills, memory/learning, or speech/language, and either an abnormality in motor function or decline in motivation/emotional control. For a diagnosis of MCMD, requirements included at least 2 of the following symptoms: impaired attention/concentration, mental slowing, impaired memory, slowed movements, incoordination, or personality change; and a cognitive or motor abnormality on examination or testing.
Metabolic Parameters
Trained personnel obtained medical history information using a structured interview to identify metabolic risk factors. Participants were classified as diabetic if they reported such a diagnosis. The remaining individuals were classified as diabetic based on published standards of having either a fasting glucose level >125 mg/dL (when available) or a nonfasting glucose level >200 mg/dL.17 Consequently, 2 participants were classified as having diabetes due to laboratory findings alone; each had a fasting glucose level >125 mg/dL while no participants met diabetes criteria based solely on nonfasting glucose evaluations. There were 7 individuals (4%) in the non-diabetic group who had nonfasting glucose levels between 125–200 mg/dL. Hypertension and hypercholesterolemia were defined as reporting a current diagnosis of either, identified from the medical history and medication intake.
Laboratory parameters were obtained on all participants who were encouraged (but not required) to arrive after an 8-hour fast (52% compliant with fasting at baseline). Blood specimens for glucose and insulin levels were drawn in serum separator tubes, allowed to clot, and then spun within 1 hour to separate serum. The separated serum was sent to a local reference laboratory where glucose levels were determined in real time. Serum specimens were frozen to −70°C and sent in batches to Quest Diagnostic Laboratory (Baltimore, MD) for quantitative insulin levels. Estimates of insulin resistance were calculated using the homeostasis model assessment (HOMA).18
Statistical Analysis
All participants signed institutional review board–approved consent forms. While participants in the parent cohort were either <40 or >50 years of age at entry by design, the cohort was examined as one group for the purpose of this set of analyses, using age in the statistical models as a continuous covariate. We estimated the independent (type III) risk of HAD associated with each glucoregulatory predictor after controlling for other known risk factors using logistic regression models. All analyses were carried out on the SAS v.8.2 (Cary, NC) platform. Inferential statistical analyses were carried out using Proc LOGISTIC and Proc GENMOD. Basic descriptive statistical analyses were carried out using Proc FREQ and Proc MEANS.
RESULTS
Baseline Demographics of the Cohort
After excluding individuals with transient ischemic attack or minor stroke, data from 96 younger and 103 older HIV-1–infected participants were analyzed. Diabetes criteria were met in 17/199 individuals (8.6%) in the cohort. The average age among patients with diabetes was 53.3 years, reflecting an age-associated risk for diabetes. Only 1 participant in our younger group met diabetes criteria. Diabetic and nondiabetic participants were similar in most parameters; however, diabetic participants were more likely to have hypertension after adjustment for age (Table 1). There were no differences in major HIV parameters including viral load, CD4 lymphocyte counts, and self-reported duration of infection between diabetic and non-diabetic participants. There were no differences in the percentages of participants on a potent ARV regimen or currently taking PIs.
TABLE 1.
Baseline Demographic Variables by Diabetes Status and Age Group
| Variable | All Participants Without DM | Participants With DM | All DM vs. All Non-DM P Value | P Value After Additional Adjustment for Age |
|---|---|---|---|---|
| n | 182 | 17 | ||
| Age | 44.97 (11.55) | 53.26 (6.18) | P < 0.01 | |
| Sex (% male) | 82.42% | 88.24% | P = 0.55 | P = 0.82 |
| BMI | 25.12 (4.82) | 27.64 (6.32) | P < 0.05 | P = 0.03 |
| Ethnicity | P = 0.39 | P = 0.27 | ||
| % White | 55.49% | 47.06% | ||
| % API | 30.22% | 41.18% | ||
| % Other | 14.29% | 11.76% | ||
| Education (y) | 14.43 (2.50) | 14.24 (2.73) | P = 0.89 | P = 0.25 |
| Years in Hawaii | 17.87 (14.21) | 21.94 (16.50) | P = 0.27 | P = 0.55 |
| On ARV | 72.93% | 76.47% | P = 0.75 | P = 0.78 |
| On ARV containing PI | 43.96% | 52.94% | P = 0.48 | P = 0.68 |
| % Undetectable VL | 56.59% | 52.94% | P = 0.77 | P < 0.99 |
| Duration of infection (y) | 9.8 (5.7) | 10.1 (5.1) | P = 0.82 | P = 0.24 |
| Mean CD4 | 444.15 (244.11) | 543.82 (342.44) | P = 0.12 | P = 0.16 |
| % with CD4 >200 | 85.16% | 82.35% | P = 0.76 | P = 0.72 |
| Smoking status | P = 0.50 | P = 0.82 | ||
| Current | 31.32% | 35.29% | ||
| Past | 38.46% | 29.41% | ||
| Plasma glucose* | 88.86 (12.74) | 154.30 (55.18) | P < 0.01 | P < 0.001 |
| Total cholesterol* | 191.02 (48.70) | 204.80 (81.27) | P = 0.59 | P = 0.72 |
| Hypertension | 9.34% | 64.71% | P < 0.0001 | P < 0.0001 |
| SBP | 123.36 (16.06) | 145.00 (16.07) | P < 0.0001 | P < 0.0001 |
| DBP | 78.50 (9.39) | 86.88 (7.94) | P < 0.001 | P < 0.01 |
| Estimated IQ (NAART) | 108.77 (8.91) | 107.20 (9.64) | P = 0.49 | P = 0.08 |
| Current substance dependence at baseline | 9.89% | 0.00% | P = 0.07 | P = 0.08 |
Fasting only.
API, Asian-Pacific Islander; BMI, body mass index; DBP, diastolic blood pressure; NAART, North American Adult Reading Test; SBP, systolic blood pressure.
Diabetes and Dementia
A diagnosis of diabetes significantly correlated to our research case definition of HAD when compared with all other diagnostic categories combined; OR = 4.33 (1.55–12.10), P < 0.01 (Fig. 1). After adjustment for important covariates including age, education, ethnicity, CD4 lymphocyte count, self-reported duration of HIV, and PI-based ARV, the significance remained; OR = 5.43 (1.66–17.70), P < 0.01 (Table 2). This association remained significant after further adjustment for important cardiovascular variables including hypertension, hypercholesterolemia, and pack-years of smoking; OR = 5.73 (1.52–21.62), P < 0.01. To maximize specificity of our non-diabetic group, we reevaluated the ORs excluding participants who had denied a diagnosis of diabetes but had a nonfasting glucose level >125 mg/dL. We found little change in the results.
FIGURE 1.
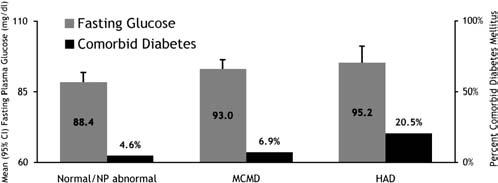
Diabetes was associated with HAD vs. any other category (P < 0.01); however, glucose was not (P = 0.14). However, the effect of glucose was significant across all diagnostic categories in an ordinal logistic regression (P < 0.05).
TABLE 2.
Prediction of HAD by Markers of Glucose Metabolism
| Marker of Glucose Metabolism | Research Diagnosis |
Unadjusted Odds Ratio (95% CI) P Value | Adjusted* Odds Ratio (95% CI) P Value | Further Adjusted† Odds Ratio (95% CI) P Value | ||
|---|---|---|---|---|---|---|
| Normal | MCMD | HAD | ||||
| Diabetes mellitus, n (%) | 4/8 (4.55%) | 5/72 (6.94%) | 8/39 (20.51%) | 4.33 (1.55–12.10) | 5.43 (1.66–17.70) | 5.73 (1.52–21.62) |
| <0.01 | <0.01 | <0.01 | ||||
| ‡Glucose (mg/dL), mean ± SD | 88.35 ± 12.86 | 93.00 ± 10.74 | 95.19 ± 10.80 | 1.04 (0.99–1.08) | 1.03 (0.98–1.08) | 1.03 (0.98–1.08) |
| n | 55 | 45 | 16 | =0.14 | =0.22 | =0.29 |
| §Insulin resistance, mean ± SD | 2.24 ± 1.40 | 2.80 ± 1.45 | 1.541 (0.63–4.06) | |||
| n | 12 | 3 | =0.34 | |||
The outcome tested was HAD vs. any other diagnostic category. Normal category includes participants categorized as NP abnormal. For insulin resistance, exact logistic regression was used due to the small sample size.
Adjusted for demographic and disease severity variables: age, education, ethnicity, on ARV with PI, duration of infection, and CD4 count.
Additionally adjusted for the following cardiovascular risk variables: hypercholesterolemia, hypertension, and pack-years of smoking.
Participants without diabetes mellitus for whom fasting glucose levels were available.
Participants without diabetes mellitus for whom insulin resistance could be estimated via HOMA. Normal and MCMD grouped together.
Since a correlation between diabetes and HAD existed independently of other cardiovascular risk factors, we investigated a possible association between glucose metabolism and HAD among diabetic and non-diabetic participants in the cohort. This was completed only among participants who had been fasting at the time of specimen collection. To maximize the number of participants available for this analysis, we used entry specimens whenever the participant was fasting at entry (n = 84) and, for participants who did not have fasting specimens at entry but did have a fasting specimen at the year 1 follow-up visit, the latter was used and correlated to the participant's year 1 follow-up diagnosis (n = 32). There were no significant differences in key variables between participants who were fasting compared with nonfasting, including age, education, ARV status, CD4 count, and percent with detectable viral load.
We found a stepwise pattern whereby fasting glucose levels were higher with increasing cognitive compromise; normal and NP abnormal combined: 88.35 ± 12.86 mg/dL, MCMD: 93.00 ± 10.78 mg/dL, and HAD: 95.19 ± 10.80 mg/dL (P < 0.05). However, a univariate analysis of the association between glucose and an outcome of HAD vs. any other diagnosis was not significant; OR = 1.04 (0.99–1.08) P = 0.14 (Table 2).
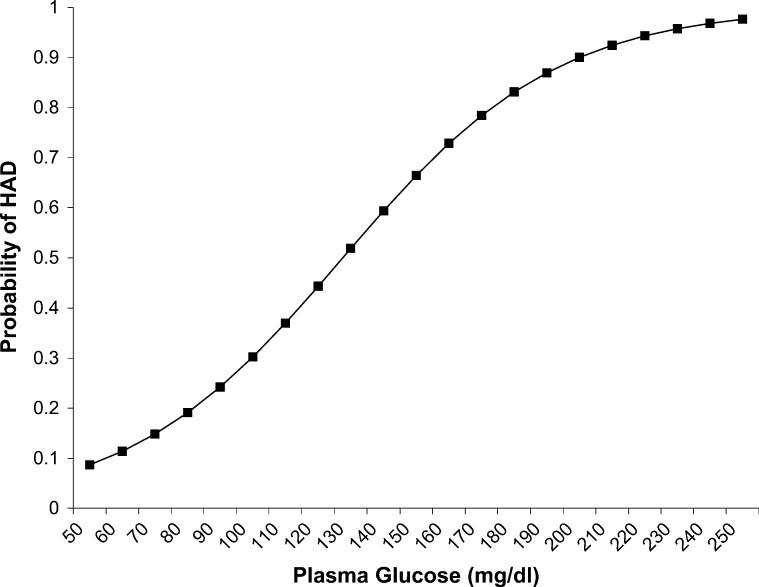
Using nondiabetic participants for whom we had fasting glucose levels, we calculated a probability curve by scoring a clinically relevant range of glucose values using the parameter estimate for glucose obtained in a univariate logistic regression with HAD as the outcome. We then converted the resulting logits (log of the odds) into probabilities (Fig. 2). The exclusion of diabetes mellitus participants makes this a conservative picture of the association of glucose with HAD because it excludes the upper end of the range of glucose values.
FIGURE 2.
Probability curve for HAD by fasting glucose level. (MD/MD participants excluded.)
To explore the association between insulin resistance and HAD, we obtained insulin levels for participants who had stored fasting serum samples at year 1 (n = 15). Here, the mean insulin levels by diagnostic category were 9.45 μU/mL (n = 11) for normal and NP abnormal combined, 16.0 μU/mL (n = 1) for MCMD, and 13.7 μU/mL (n = 3) for HAD. Since only one participant met MCMD criteria, we combined this participant with the normal group and calculated the insulin resistance using the HOMA to determine risk for HAD. The mean insulin resistance level was 2.8 for participants with HAD compared with 2.2 for participants without HAD (P = 0.34, Table 2). The small number of specimens available for this preliminary analysis limits our clear understanding of this intriguing finding.
DISCUSSION
Our findings suggest that dysregulation of glucose homeostasis may be a risk factor for cognitive impairment among HIV-1–infected individuals. Specifically, in the Hawaii Aging with HIV Cohort, having a diagnosis of diabetes is a risk factor for HAD. This finding appears to be principally related to diabetes occurring among older participants. Since early diabetes is often unrecognized in adults, it is likely that we have under-identified the disorder in our group, which would tend to decrease our ability to identify relationships. However, when this correlation is assessed further by analyzing subclinical disease (fasting glucose levels), our findings indicate an association between glucose homeostasis and cognitive dysfunction among older and younger patients combined. Our test of association between fasting glucose and HAD was especially strict in that it excluded participants with diabetes. Thus, the risk associated with increasing fasting glucose reported here excludes participants with levels sufficient to meet diabetes criteria and should be considered a conservative estimate of risk. Our exploratory investigation of insulin resistance resulted in an interesting pattern; however, a larger sample size is required to determine differences. Based on these preliminary data, we should have 80% power to detect a difference once data are available on 82 participants.
To our knowledge, this is the first published report indicating a correlation between cognition and abnormalities of glucoregulation among HIV-1–infected patients. Since our findings are based on a relatively small number of cases, particularly among younger HIV patients, this work deserves confirmation in larger cohorts. Other limitations to this work include the post hoc aspect to the analysis, the modest neuro-psychological battery used to diagnose impairment, and the use of self-report and fasting glucose levels rather than true measures of diabetes and insulin resistance. Evaluation of the longitudinal data in our cohort will further clarify these relationships as the current data are based solely on cross-sectional data. Nevertheless, this work should encourage others to consider glucose homeostasis in a model for cognitive dysfunction in HIV patients, particularly patients in their 6th decade of life or older.
The underlying mechanisms associated with our finding remain to be clarified. Comigration of risk factors, including elevated lipid levels and hypertension, suggest that the effect of hyperglycemia or hyperinsulinemia may be partially mediated by their combined atherogenic effect. Proatherogenic lipid profiles occur with HIV-1 infection and other chronic infections such as Chlamydia pneumoniae and cytomegaloviral infections and are likely to represent a cytokine-mediated, acute-phase response to chronic infection.19 Such proatherogenic tendencies may be further compounded by metabolic abnormalities of insulin resistance, elevated LDL cholesterol, and triglyceride levels commonly induced by various potent ARV medications.
Additional findings that are relevant to the metabolic syndrome have also been reported in the HIV-infected population. Elevated blood pressure has been reported in association with lipodystrophy.20 Elevation of blood pressure may occur in the context of either PI-based or nonnucleoside reverse transcriptase inhibitor–based ARV regimens.21
Our data suggest that the risk of cognitive impairment associated with diabetes may not be fully mediated through an atherogenic mechanism. We adjusted for key vascular risk factors including pack-years of smoking, hypertension, and hypercholesterolemia in our model and an increased risk was identified independently of these markers. However, our adjustment variables were also limited by self-report and may have underestimated the frequency of disease states. A correlation with fasting glucose level and a suggestion for a possible association with insulin resistance were noted, further indicating that glucoregulatory-specific mechanisms may be involved.
Population-based studies among HIV-negative patients indicate a correlation between diabetes and cognition,22,23 including relationships for Alzheimer dementia22-24 and the cognitive impairment associated with vascular disease.22 While some discrepant reports suggest that other factors may be involved and controversy remains as to whether their role is independent and causative,25,26 these associations appear firmly established.27 While diabetes is associated with atherosclerosis of cerebral arteries, lacunar infarcts, and decreased cerebral blood flow and the frequency of insulin resistance and diabetes is associated with other cardiovascular risk factors, adjusted models typically evidence an independent relative risk for cognitive abnormalities associated with diabetes and insulin resistance themselves.22 Further, early investigations using positron emission tomography (PET) support a causative role whereby changes in brain glucose metabolism are identified early in Alzheimer disease patients,28,29 with PET changes often preceding detectable cognitive abnormalities.30 A decreased brain glucose metabolism has also been reported in biopsy specimens of Alzheimer patients.31
In seronegative populations, several possible pathogenic mechanisms have been hypothesized to explain the relationship between changes in cognition and glucoregulation. These pathways include harmful interactions with amyloid beta leading to greater deposition of aggregate insoluble proteins; competitive inhibition of insulin degradation enzyme contributing to an increased amyloid burden; blood-brain barrier insulin resistance leading to decreased available insulin in the brain, subsequently attenuating protective insulin mechanisms; and increased production of detrimental advanced glycation end products associated with impaired neurosignaling.22,32,33 These potential contributing factors have not been explored in HIV-1–infected populations. Further evaluation of these mechanisms could identify modifiable pathways and factors, which might ultimately improve the burden of cognitive impairment in this vulnerable population.
Current PI use did not correlate with a diagnosis of diabetes in our cohort. However, our data did not allow us to fully identify the duration of past exposure. Our findings are also limited by the absence of data for individuals in their 5th decade of life (due to the design of the parent cohort). This cohort is enriched with participants who are ≥50 years of age, in whom applicability of these risk factors may be greatest.
In summary, our results suggest that diabetes is independently associated with HAD in the Hawaii Aging with HIV Cohort. Further, we contend that dysregulation of glucose may pose a risk for cognitive impairment among HIV-1–infected nondiabetic individuals regardless of age. These findings are based on a relatively small number of cases; if confirmed, they could have important implications for clinical care of HIV-infected individuals, particularly older patients. A vigilant and aggressive approach to the management of metabolic abnormalities may be important to the long-term morbidity for HIV-1–infected adults. Further work to identify underlying mechanisms and subsequent modifiable pathways is warranted.
ACKNOWLEDGMENTS
The authors thank our participants and community physicians.
Footnotes
Supported by NIH grant 1U54NS43049 with additional support from P20 RR11091 and G12 RR/AI 03061.
REFERENCES
- 1.Feingold KR, Krauss RM, Pang M, et al. The hypertriglyceridemia of acquired immunodeficiency syndrome is associated with an increased prevalence of low density lipoprotein subclass pattern B. J Clin Endocrinol Metab. 1993;76:1423–1427. doi: 10.1210/jcem.76.6.8501146. [DOI] [PubMed] [Google Scholar]
- 2.Grunfeld C, Kotler DP, Hamadeh R, et al. Hypertriglyceridemia in the acquired immunodeficiency syndrome. Am J Med. 1989;86:27–31. doi: 10.1016/0002-9343(89)90225-8. [DOI] [PubMed] [Google Scholar]
- 3.Shor-Posner G, Basit A, Lu Y, et al. Hypocholesterolemia is associated with immune dysfunction in early human immunodeficiency virus-1 infection. Am J Med. 1993;94:515–519. doi: 10.1016/0002-9343(93)90087-6. [DOI] [PubMed] [Google Scholar]
- 4.Walli R, Goebel FD, Demant T. Impaired glucose tolerance and protease inhibitors. Ann Intern Med. 1998;129:837–838. doi: 10.7326/0003-4819-129-10-199811150-00022. [DOI] [PubMed] [Google Scholar]
- 5.Carr A, Samaras K, Thorisdottir A, et al. Diagnosis, prediction, and natural course of HIV-1 protease-inhibitor-associated lipodystrophy, hyperlipidaemia, and diabetes mellitus: a cohort study. Lancet. 1999;353:2093–2099. doi: 10.1016/S0140-6736(98)08468-2. [DOI] [PubMed] [Google Scholar]
- 6.Dever LL, Oruwari PA, Figueroa WE, et al. Hyperglycemia associated with protease inhibitors in an urban HIV-infected minority patient population. Ann Pharmacother. 2000;34:580–584. doi: 10.1345/aph.19231. [DOI] [PubMed] [Google Scholar]
- 7.Calza L, Manfredi R, Farneti B, et al. Incidence of hyperlipidaemia in a cohort of 212 HIV-infected patients receiving a protease inhibitor-based antiretroviral therapy. Int J Antimicrob Agents. 2003;22:54–59. doi: 10.1016/s0924-8579(03)00100-6. [DOI] [PubMed] [Google Scholar]
- 8.Nolan D. Metabolic complications associated with HIV protease inhibitor therapy. Drugs. 2003;63:2555–2574. doi: 10.2165/00003495-200363230-00001. [DOI] [PubMed] [Google Scholar]
- 9.DeCarli C. The role of cerebrovascular disease in dementia. Neurology. 2003;9:123–136. doi: 10.1097/00127893-200305000-00001. [DOI] [PubMed] [Google Scholar]
- 10.Barbaro G. HIV infection, highly active antiretroviral therapy and the cardiovascular system. Cardiovasc Res. 2003;60:87–95. doi: 10.1016/s0008-6363(02)00828-3. [DOI] [PubMed] [Google Scholar]
- 11.Holmberg SMA, Tong T, Ward D, et al. 698-T: protease inhibitor use and adverse cardiovascular outcomes in ambulatory HIV patients. Paper presented at: 9th Conference on Retroviruses and Opportunistic Infections; Seattle, WA. February 24–28, 2002.2002. [Google Scholar]
- 12.Mary-Krause L, Cotte L, Partisani M, et al. T-657: impact of treatment with protease inhibitor on myocardial infarction occurrence in HIV-infected men. Paper presented at: 8th Conference on Retroviruses and Opportunistic Infections; Chicago, IL. February 4–8, 2001.2001. [Google Scholar]
- 13.Bozzette SA, Ake CF, Tam HK, et al. Cardiovascular and cerebrovascular events in patients treated for human immunodeficiency virus infection. N Engl J Med. 2003;348:702–710. doi: 10.1056/NEJMoa022048. [DOI] [PubMed] [Google Scholar]
- 14.Valcour V, Shikuma C, Shiramizu B, et al. Higher frequency of dementia in older HIV+ individuals. The Hawaii Aging with HIV Cohort. Neurology. 2004;63:822–827. doi: 10.1212/01.wnl.0000134665.58343.8d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Marder K, Liu X, Stern Y, et al. Neurologic signs and symptoms in a cohort of homosexual men followed for 4.5 years. Neurology. 1995;45:261–267. doi: 10.1212/wnl.45.2.261. [DOI] [PubMed] [Google Scholar]
- 16.Nomenclature and research case definitions for neurologic manifestations of human immunodeficiency virus-type 1 (HIV-1) infection. Report of a Working Group of the American Academy of Neurology AIDS Task Force. Neurology. 1991;41:778–785. doi: 10.1212/wnl.41.6.778. [DOI] [PubMed] [Google Scholar]
- 17.Genuth S, Alberti KG, Bennett P, et al. Follow-up report on the diagnosis of diabetes mellitus. Diabetes Care. 2003;26:3160–3167. doi: 10.2337/diacare.26.11.3160. [DOI] [PubMed] [Google Scholar]
- 18.Matthews DR, Hosker JP, Rudenski AS, et al. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28:412–419. doi: 10.1007/BF00280883. [DOI] [PubMed] [Google Scholar]
- 19.Khovidhunkit W, Memon RA, Feingold KR, et al. Infection and inflammation-induced proatherogenic changes of lipoproteins. J Infect Dis. 2000;181(Suppl 3):S462–S472. doi: 10.1086/315611. [DOI] [PubMed] [Google Scholar]
- 20.Gazzaruso C, Bruno R, Garzaniti A, et al. Hypertension among HIV patients: prevalence and relationships to insulin resistance and metabolic syndrome. J Hypertens. 2003;21:1377–1382. doi: 10.1097/01.hjh.0000059071.43904.dc. [DOI] [PubMed] [Google Scholar]
- 21.Chow DC, Souza SA, Chen R, et al. Elevated blood pressure in HIV-infected individuals receiving highly active antiretroviral therapy. HIV Clin Trials. 2003;4:411–416. doi: 10.1310/5E7Q-PGWB-16UE-J48U. [DOI] [PubMed] [Google Scholar]
- 22.Peila R, Rodriguez BL, Launer LJ. Type 2 diabetes, APOE gene, and the risk for dementia and related pathologies: the Honolulu-Asia Aging Study. Diabetes. 2002;51:1256–1262. doi: 10.2337/diabetes.51.4.1256. [DOI] [PubMed] [Google Scholar]
- 23.Leibson CL, Rocca WA, Hanson VA, et al. The risk of dementia among persons with diabetes mellitus: a population-based cohort study. Ann NY Acad Sci. 1997;826:422–427. doi: 10.1111/j.1749-6632.1997.tb48496.x. [DOI] [PubMed] [Google Scholar]
- 24.Kuusisto J, Koivisto K, Mykkanen L, et al. Association between features of the insulin resistance syndrome and Alzheimer's disease independently of apolipoprotein E4 phenotype: cross sectional population based study. BMJ. 1997;315:1045–1049. doi: 10.1136/bmj.315.7115.1045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Craft S, Watson GS. Insulin and neurodegenerative disease: shared and specific mechanisms. Lancet Neurol. 2004;3:169–178. doi: 10.1016/S1474-4422(04)00681-7. [DOI] [PubMed] [Google Scholar]
- 26.Vanhanen M, Koivisto K, Kuusisto J, et al. Cognitive function in an elderly population with persistent impaired glucose tolerance. Diabetes Care. 1998;21:398–402. doi: 10.2337/diacare.21.3.398. [DOI] [PubMed] [Google Scholar]
- 27.Gasparini L, Netzer WJ, Greengard P, et al. Does insulin dysfunction play a role in Alzheimer's disease? Trends Pharmacol Sci. 2002;23:288–293. doi: 10.1016/s0165-6147(02)02037-0. [DOI] [PubMed] [Google Scholar]
- 28.Friedland RP, Budinger TF, Ganz E, et al. Regional cerebral metabolic alterations in dementia of the Alzheimer type: positron emission tomography with [18F]fluorodeoxyglucose. J Comput Assist Tomogr. 1983;7:590–598. doi: 10.1097/00004728-198308000-00003. [DOI] [PubMed] [Google Scholar]
- 29.Foster NL, Chase TN, Fedio P, et al. Alzheimer's disease: focal cortical changes shown by positron emission tomography. Neurology. 1983;33:961–965. doi: 10.1212/wnl.33.8.961. [DOI] [PubMed] [Google Scholar]
- 30.Haxby JV, Grady CL, Friedland RP, et al. Neocortical metabolic abnormalities precede nonmemory cognitive impairments in early dementia of the Alzheimer type: longitudinal confirmation. J Neural Transm Suppl. 1987;24:49–53. [PubMed] [Google Scholar]
- 31.Sims NR, Finegan JM, Blass JP, et al. Mitochondrial function in brain tissue in primary degenerative dementia. Brain Res. 1987;436:30–38. doi: 10.1016/0006-8993(87)91553-8. [DOI] [PubMed] [Google Scholar]
- 32.Watson GS, Peskind ER, Asthana S, et al. Insulin increases CSF Abeta42 levels in normal older adults. Neurology. 2003;60:1899–1903. doi: 10.1212/01.wnl.0000065916.25128.25. [DOI] [PubMed] [Google Scholar]
- 33.Craft S, Asthana S, Cook DG, et al. Insulin dose-response effects on memory and plasma amyloid precursor protein in Alzheimer's disease: interactions with apolipoprotein E genotype. Psychoneuroendocrinology. 2003;28:809–822. doi: 10.1016/s0306-4530(02)00087-2. [DOI] [PubMed] [Google Scholar]