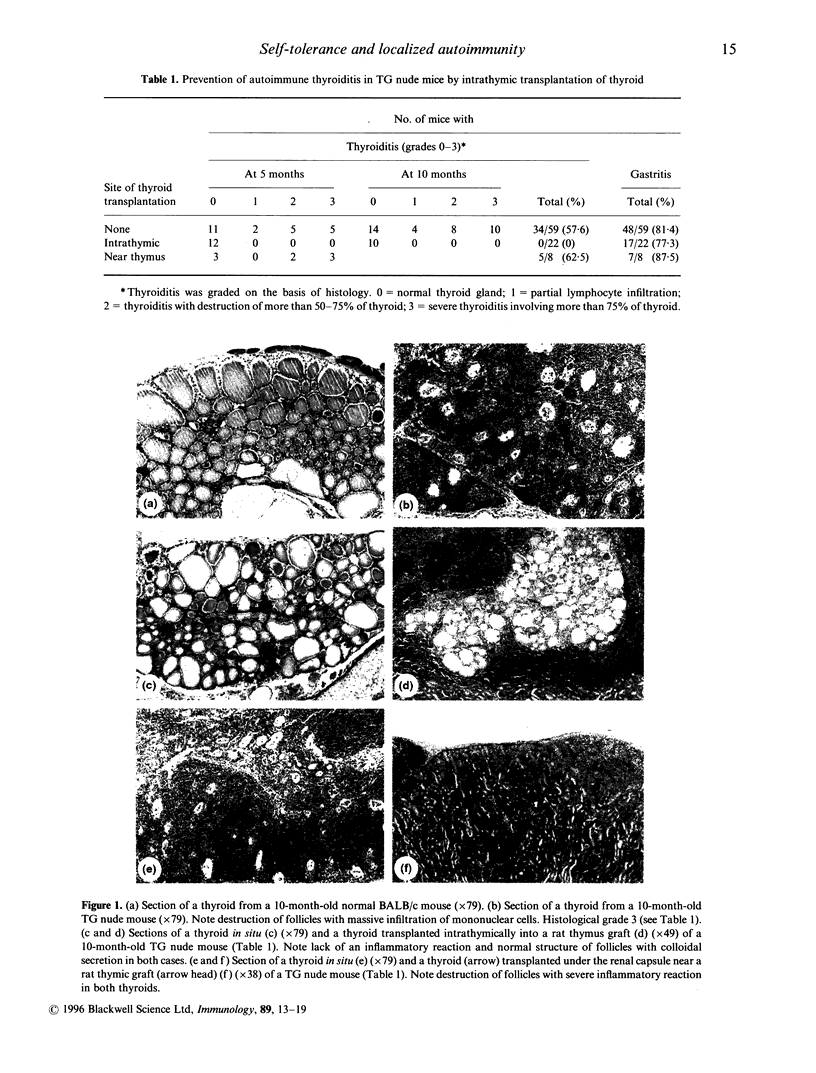
Abstract
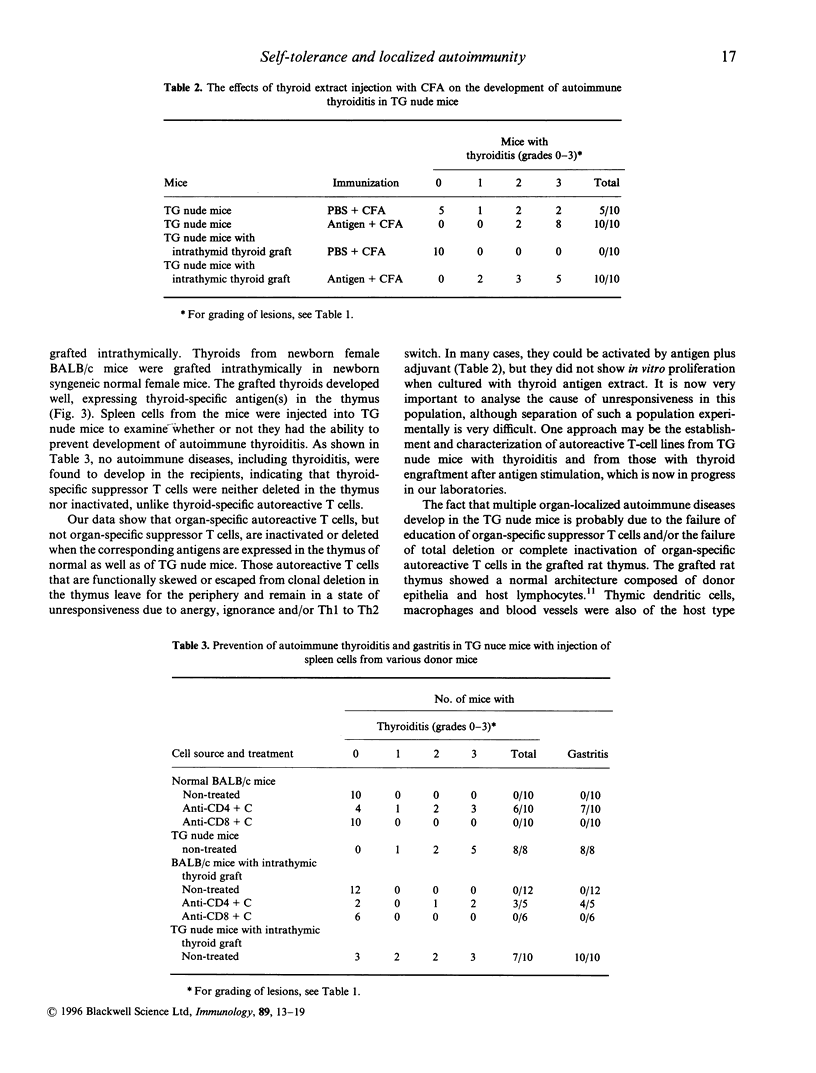
Multiple organ-localized autoimmune diseases, such as thyroiditis and gastritis, spontaneously develop in BALB/c nu/nu (nude) mice receiving embryonic rat thymus grafts (TG) under their renal capsules (TG nude mice). When thyroid was grafted into the rat thymus of TG nude mice, development of autoimmune thyroiditis, but not other diseases, was completely prevented. However, when such mice received thyroid antigen plus complete Freund's adjuvant (CFA), severe autoimmune thyroiditis developed, suggesting that some thyroid-specific autoreactive T cells migrate into the periphery, but remain unresponsive. Development of autoimmune diseases, including thyroiditis, in TG nude mice was prevented by a single intraperitoneal injection of splenic CD4+ cells from normal BALB/c mice and also from mice with intrathymic thyroid grafts, indicating that thyroid-specific suppressor T cells are present in normal mice and that such T cells are neither deleted nor inactivated by the intrathymic thyroid grafts, in contrast to autoreactive T cells. Thus clonal deletion in the thymus, and clonal anergy and/or ignorance in the periphery, of autoreactive cells is important to maintain immune tolerance to organ-specific antigen, but CD4 suppressor T cells may play a more important role in tolerance, and the failure of education of this population may cause autoimmune diseases in the TG nude mouse model.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Burkly L. C., Lo D., Kanagawa O., Brinster R. L., Flavell R. A. T-cell tolerance by clonal anergy in transgenic mice with nonlymphoid expression of MHC class II I-E. Nature. 1989 Nov 30;342(6249):564–566. doi: 10.1038/342564a0. [DOI] [PubMed] [Google Scholar]
- Cunningham A. J. Active suppressor mechanism maintaining tolerance to some self components. Nature. 1975 Mar 13;254(5496):143–144. doi: 10.1038/254143a0. [DOI] [PubMed] [Google Scholar]
- Herold K. C., Montag A. G., Buckingham F. Induction of tolerance to autoimmune diabetes with islet antigens. J Exp Med. 1992 Oct 1;176(4):1107–1114. doi: 10.1084/jem.176.4.1107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ichikawa T., Taguchi O., Takahashi T., Ikeda H., Takeuchi M., Tanaka T., Usui M., Nishizuka Y. Spontaneous development of autoimmune uveoretinitis in nude mice following reconstitution with embryonic rat thymus. Clin Exp Immunol. 1991 Oct;86(1):112–117. doi: 10.1111/j.1365-2249.1991.tb05782.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikeda H., Taguchi O., Takahashi T., Itoh G., Nishizuka Y. L3T4 effector cells in multiple organ-localized autoimmune disease in nude mice grafted with embryonic rat thymus. J Exp Med. 1988 Dec 1;168(6):2397–2402. doi: 10.1084/jem.168.6.2397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kappler J. W., Roehm N., Marrack P. T cell tolerance by clonal elimination in the thymus. Cell. 1987 Apr 24;49(2):273–280. doi: 10.1016/0092-8674(87)90568-x. [DOI] [PubMed] [Google Scholar]
- Kisielow P., Blüthmann H., Staerz U. D., Steinmetz M., von Boehmer H. Tolerance in T-cell-receptor transgenic mice involves deletion of nonmature CD4+8+ thymocytes. Nature. 1988 Jun 23;333(6175):742–746. doi: 10.1038/333742a0. [DOI] [PubMed] [Google Scholar]
- Koevary S. B., Blomberg M. Prevention of diabetes in BB/Wor rats by intrathymic islet injection. J Clin Invest. 1992 Feb;89(2):512–516. doi: 10.1172/JCI115614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kojima A., Tanaka-Kojima Y., Sakakura T., Nishizuka Y. Prevention of postthymectomy autoimmune thyroiditis in mice. Lab Invest. 1976 Jun;34(6):601–605. [PubMed] [Google Scholar]
- Mackay C. R. Homing of naive, memory and effector lymphocytes. Curr Opin Immunol. 1993 Jun;5(3):423–427. doi: 10.1016/0952-7915(93)90063-x. [DOI] [PubMed] [Google Scholar]
- Marrack P., Lo D., Brinster R., Palmiter R., Burkly L., Flavell R. H., Kappler J. The effect of thymus environment on T cell development and tolerance. Cell. 1988 May 20;53(4):627–634. doi: 10.1016/0092-8674(88)90578-8. [DOI] [PubMed] [Google Scholar]
- Ohashi P. S., Oehen S., Buerki K., Pircher H., Ohashi C. T., Odermatt B., Malissen B., Zinkernagel R. M., Hengartner H. Ablation of "tolerance" and induction of diabetes by virus infection in viral antigen transgenic mice. Cell. 1991 Apr 19;65(2):305–317. doi: 10.1016/0092-8674(91)90164-t. [DOI] [PubMed] [Google Scholar]
- Oldstone M. B., Nerenberg M., Southern P., Price J., Lewicki H. Virus infection triggers insulin-dependent diabetes mellitus in a transgenic model: role of anti-self (virus) immune response. Cell. 1991 Apr 19;65(2):319–331. doi: 10.1016/0092-8674(91)90165-u. [DOI] [PubMed] [Google Scholar]
- Posselt A. M., Barker C. F., Friedman A. L., Naji A. Prevention of autoimmune diabetes in the BB rat by intrathymic islet transplantation at birth. Science. 1992 May 29;256(5061):1321–1324. doi: 10.1126/science.1598576. [DOI] [PubMed] [Google Scholar]
- Pullen A. M., Marrack P., Kappler J. W. The T-cell repertoire is heavily influenced by tolerance to polymorphic self-antigens. Nature. 1988 Oct 27;335(6193):796–801. doi: 10.1038/335796a0. [DOI] [PubMed] [Google Scholar]
- Rammensee H. G., Kroschewski R., Frangoulis B. Clonal anergy induced in mature V beta 6+ T lymphocytes on immunizing Mls-1b mice with Mls-1a expressing cells. Nature. 1989 Jun 15;339(6225):541–544. doi: 10.1038/339541a0. [DOI] [PubMed] [Google Scholar]
- Ramsdell F., Lantz T., Fowlkes B. J. A nondeletional mechanism of thymic self tolerance. Science. 1989 Nov 24;246(4933):1038–1041. doi: 10.1126/science.2511629. [DOI] [PubMed] [Google Scholar]
- Sha W. C., Nelson C. A., Newberry R. D., Kranz D. M., Russell J. H., Loh D. Y. Positive and negative selection of an antigen receptor on T cells in transgenic mice. Nature. 1988 Nov 3;336(6194):73–76. doi: 10.1038/336073a0. [DOI] [PubMed] [Google Scholar]
- Staerz U. D., Rammensee H. G., Benedetto J. D., Bevan M. J. Characterization of a murine monoclonal antibody specific for an allotypic determinant on T cell antigen receptor. J Immunol. 1985 Jun;134(6):3994–4000. [PubMed] [Google Scholar]
- Taguchi O., Kontani K., Ikeda H., Kezuka T., Takeuchi M., Takahashi T., Takahashi T. Tissue-specific suppressor T cells involved in self-tolerance are activated extrathymically by self-antigens. Immunology. 1994 Jul;82(3):365–369. [PMC free article] [PubMed] [Google Scholar]
- Taguchi O., Takahashi T., Nishizuka Y. Self-tolerance and localized autoimmunity. Curr Opin Immunol. 1989;2(4):576–581. doi: 10.1016/0952-7915(90)90013-7. [DOI] [PubMed] [Google Scholar]
- Taguchi O., Takahashi T., Seto M., Namikawa R., Matsuyama M., Nishizuka Y. Development of multiple organ-localized autoimmune diseases in nude mice after reconstitution of T cell function by rat fetal thymus graft. J Exp Med. 1986 Jul 1;164(1):60–71. doi: 10.1084/jem.164.1.60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Twarog F. J., Rose N. R. The production of thyroid autoantibodies in mice. J Immunol. 1968 Aug;101(2):242–250. [PubMed] [Google Scholar]