FIGURE 5.
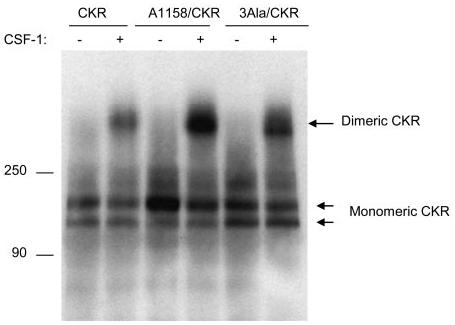
Effect of mutation of leucines 1158, 1161, and 1162 on ligand-induced dimerization of VEGFR-2. An equal number of serum-starved PAE cells expressing wild type chimeric VEGFR-2 (CKR), A1158/CKR, and A1158/1161/1162 (3Ala/CKR) were either not stimulated or stimulated for 10 min with CSF-1 (40 ng/ml). Cells were lysed and total cell lysates were immunoprecipitated with anti-VEGFR-2 antibody. The immunoprecipitated proteins were resolved in a 4–15% gradient non-reducing SDS-PAGE and proteins were transferred to polyvinylidene difluoride membrane and finally subjected to Western blot analysis using an anti-VEGFR-2 antibody. The positions of monomeric and dimeric VEGFR-2s are shown. It is also evident that expression of the mutant VEGFR-2s are slightly higher than the wild type VEGFR-2. This may explain why there is more dimerized mutant of VEGFR-2s compared with that of the wild type VEGFR-2.
