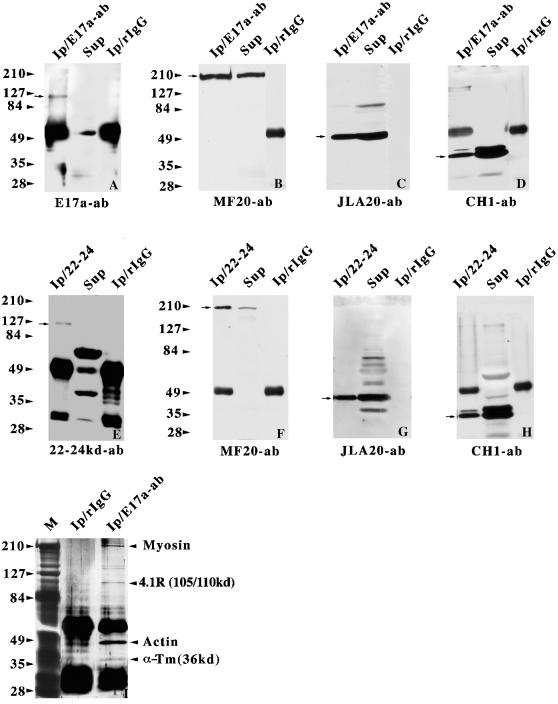
Figure 6.
Coimmunoprecipitation of protein 4.1R and myosin, α-actin, and tropomyosin from adult skeletal muscle homogenates. The 4.1R anti-E17a antibody (A, B, C, D, and I) and anti-22/24 kDa antibody (E, F, G, and H) along with rabbit IgG were used in the immunoprecipitation assays. The immunoprecipitates were analyzed by immunoblotting with anti-E17a (A), anti-22/24 kDa (E), anti-myosin MF20 mAb (B and F), anti-actin JLA20 mAb (C and G), and anti-tropomyosin CH1 mAb (D and H). The arrows denote the migration positions of the ∼105/110-kDa 4.1R protein (A and E), myosin (B and F), α-actin (C and G), and tropomyosin (D and H). One-fourth of the immunoprecipitates (Ip) and one-eighth of the supernatant fractions (Sup) were loaded onto the lanes as indicated. (I) An aliquot of either rabbit IgG or anti-E17a immunoprecipitates was subjected to silver staining. M, silver-stained molecular mass protein standards. The arrowheads point to the positions of protein 4.1R, myosin, α-actin, and α-tropomyosin.