Abstract
Study design: An investigation of c-fos activation pattern in spinal neurons of intact adult rats after acute bouts of treadmill locomotion.
Objectives: To map spinal neurons that are involved in quadrupedal treadmill stepping of intact adult rats by using c-fos as a marker.
Settings: Los Angeles, CA, USA.
Methods: Spinal cord sections of rats that were not stepped (n = 4) were used to map the FOS-positive (+) neurons under basal conditions. The stepped group (n = 16) was placed on a treadmill to step quadrupedally for varying durations to induce c-fos activity. Spinal cord sections of thoracic and lumbar segments of Stp and Nstp rats were processed using a c-fos antibody, choline acetyl transferase and heat shock protein 27 for identifying motoneurons.
Results: Stepping induced a greater number of FOS+ neurons than was observed in rats that did not step on the treadmill. There was a rostrocaudal and a dorsoventral gradient of FOS labeled neurons. The number of FOS+ neurons increased with the duration of treadmill stepping. Significant increases in FOS+ neurons were in the most medial parts of laminae IV, V, and VII. FOS+ motoneurons increased with treadmill stepping, particularly in large motoneurons ().
Conclusion: These data suggest that FOS can be used to identify activity-dependent neuronal pathways in the spinal cord that are associated with treadmill stepping, specifically in lamina VII and in alpha motoneurons.
Sponsorship: NIH NS16333, NS40917, and the Christopher Reeve Paralysis Foundation (CRPF VEC 2002).
Keywords: locomotion, motoneurons, mapping, interneurons, HSP-27, activity-dependent
Introduction
Little data are available on the location of motoneurons and interneurons within the spinal cord that become active during in vivo locomotion in intact or spinal cord transected animals. Near-normal stepping can be produced by these networks within the spinal cord with the sensory input associated with treadmill locomotion. The general question asks where are these networks of neurons that can generate these movements with or without the connection to the brain. The present study was designed to identify the number and location of locomotor-associated neurons within the lumbosacral spinal cord, using c-fos as a marker of active neurons. We hypothesized that there would be a definable number of neurons restricted to specific anatomical locations that are linked specifically to quadrupedal locomotion.
The FOS protein is a product of an immediate early gene (IEG), c-fos. It is found ubiquitously in the cell nucleus as a result of many types of stimuli such as stress, pain, cutaneous inputs, growth factors, and locomotion.1,2 Production of the FOS protein occurs within minutes following the given stimulus, is maximally expressed approximately 1 h post-stimulus, and can be readily detected using immunohistochemical methods.3 Despite the ubiquitous nature of c-fos activation with various stimuli, it can be found in specific populations dependent on the specific type of stimulus. Other examples which make use of FOS as a marker of neural activity in specific populations include the visual system,4-6 the olfactory system,7-10 taste,10 social stress,11 the endocrine system,12 pharmacological intervention,13 neurotransmitters,14,15 and locomotion.16-21
We examined the distribution pattern of neurons activated by quadrupedal treadmill locomotion in the intact rat. c-fos is activated in spinal neurons after mesencephalic locomotor region (MLR) stimulation to evoke fictive and treadmill locomotion in the cat and fictive locomotion in the neonatal rat spinal cord.22 Assuming that the organization of active neurons in the cat are similar to the rat, we compared the pattern of activated neurons reported by other groups following MLR-evoked stepping and fictive locomotion to actual or voluntary quadrupedal treadmill stepping in an intact rat.22,36,44 We observed FOS-positive (+) interneurons and motoneurons in intact adult rats+ after a single bout of quadrupedal treadmill stepping. FOS+ staining was examined relative to motoneuronal soma size, which is related to the frequency of motoneuron activation. Finally, we characterized the pattern of neuronal activation as a function of voluntary quadrupedal treadmill stepping duration. Preliminary results have been published as abstracts in several conferences.23-25
Methods
Quadrupedal stepping on a treadmill
Two groups of rats were studied; one group was not stepped on a treadmill but had normal cage activity, while the second group was stepped quadrupedally on a treadmill. The stepped rats (Stp, n = 16) were placed on a rat treadmill for one bout of stepping for durations of 6, 23, 35, 45, or 60 min (n=3, 3, 3, 3, and 4, respectively) at an average speed of 13.5 cm/s (Table 1). The most consistent stepping occurred at this averaged speed and thus was chosen for all stepping durations. The Stp rats did not step consistently at higher speeds and for this reason duration of treadmill stepping was chosen over the intensity of stepping. In our study, at the slowest speed of 6 cm/s, rats were unmotivated to step consistently, whereas at the highest treadmill speed of 21 cm/s, rats became too fatigued to continue stepping for the desired amount of time and were prone to injury. At the onset of fatigue the rats would not step at the required pace, which caused friction between their dragging feet and the moving treadmill belt. This in turn resulted in injury to the toe nails of two rats, presumably causing activation of pain pathways. Past research shows that noxious stimuli such as electrical stimulation and inflammation induce c-fos specifically only in laminae I–III and not in lamina IX of the ventral horn (VH).26,27
Table 1.
Summary of experimental groups
| Labeling |
|||||||
|---|---|---|---|---|---|---|---|
| Rat ID | N | Speed (cm/s) | One-bout step duration (min) | FOS | HSP-27 | Adjacent | Double IF |
| Nstp | 4 | — | 0 | X | |||
| Stp | 16 | 13.5 | — | ||||
| 3 | 13.5 | 6 | X | ||||
| 3 | 13.5 | 23 | X | ||||
| 3 | 13.5 | 35 | X | ||||
| 3 | 13.5 | 45 | X | ||||
| 4 | 13.5 | 60 | X | X | X | X | |
Nstp, nonstepped (cage activity) rats; Stp, treadmill-stepped rats; n, number of animals per group; adjacent, each trial was performed on two sections chosen adjacently and one section treated with c-fos the other treated with HSP-27; double IF, sections were stained with both c-fos and HSP-27 antibody using the IF method
Therefore, the rats that underwent noxious stimuli were used only to examine locomotor-associated c-fos activation in the VH. Fatigue-induced c-fos studies in the past have identified c-fos+ neurons in the dorsal horn (DH) only.28 We minimized fatigue and noxious stimuli by optimizing the treadmill speed. Over several trials, all other rats were able to step for at least 60 min at a constant speed of 13.5 cm/s.
Nonstepped rats (Nstp, n = 4) were left in their cages, where their physical activity was limited to the confines of their cage. These rats served as controls for basal locomotor activity when compared with treadmill stepped rats. All procedures were performed according to institutional and governmental regulations, and in accordance with the guidelines set and delineated by the University of California, Los Angeles (UCLA) Animal Research Committee (ARC) protocol concerning the ethical use of animals.
Tissue preparation
To ensure time for maximal FOS staining to occur, rats were returned to their cages and perfused 60 min after the treadmill-stepping bout. Perfusions of nonstepped and stepped rats were matched with respect to the time of day. Animals were deeply anesthetized with Eutha-6 (80 mg/kg i.p.) approximately 5 min before perfusion. All animals were perfused transcardially with a fixative solution of cold 4% paraformaldehyde (PF) in phosphate buffer under deep anesthesia with approximately 450–500 ml of 4% PF for 20 min. The dissected vertebral column containing the spinal cord was post-fixed in 4% PF overnight at 4°C. Before the spinal cords were dissected, they were rinsed four times (30 min each) in phosphate buffer and then left in phosphate buffer overnight at 4°C.
The spinal cords were dissected from the vertebral column and stored in 30% sucrose at 4°C. The spinal cord segments were sectioned into specific blocks and mounted on cork as follows: (block 1) T13–L2, (block 2) L3–L4, and (block 3) L5–L6. These regions were determined by locating the dorsal root ganglion (DRG) of L5. The blocks were mounted on pieces of cork with Polyfreeze (Polysciences, Inc., Warrington, PA, USA) then frozen with powdered dry ice and stored in −80°C. Blocks were later removed for sectioning on a cryostat. Free-floating 30 μm cross sections of spinal cord were cut with a cryostat and stored in 1 × PBS at 4°C. For long-term storage, these sections were later transferred into a cryoprotectant solution along with sodium azide to prevent fungal growth.
Immunohistochemistry (IHC)
Spinal cord sections were handled while free-floating in net wells (Corning Inc., MA, USA). Rinsing was carried out in net wells, while antibody and color reactions were performed in well plates. Sections were first rinsed in 1 × PBS for 30 min, and then incubated in blocking solution (3% normal goat serum in PBS and 0.3% Triton X-100) for 30 min to 1 h. These sections were then incubated in rabbit polyclonal c-fos antibody (Oncogene Research Products, San Diego, CA, USA) at 1:8000 in 1 × PBS overnight at room temperature (RT) on an orbital shaker with shaking for approximately 16–20 h. The following day, free-floating sections were again rinsed in PBS as follows: two quick rinses, two rinses of 5 min each, and two rinses of 10 min each. The sections were then placed in 1:300 secondary antibody (biotinylated goat anti-rabbit; Vector Laboratories, Burlingame, CA, USA) for 1 h at RT on a shaker and rinsed as described above for 30 min. The sections were incubated at RT for 1 h in 1:200 Vectastain Elite Avidin-Biotin Peroxidase Complex (ABC) solution (Vector Laboratories, Burlingame, CA, USA), then rinsed again in PBS and placed in diaminobenzidine (DAB) solution (Sigma-Aldrich Corp., St Louis, MO, USA) for 15 min for the chromogen reaction to occur. Then, sections were rinsed for at least 30 min to stop the color reaction. Next, stained sections were mounted on microscope slides and air-dried overnight. The sections were then counterstained with 2% methyl green and mounted with Permount (Fisher Scientific, Fair Lawn, New Jersey).
c-fos antibody specificity
The specificity of c-fos immunostaining in spinal cord sections was demonstrated by the absence of staining in the nuclei of the spinal cord cells carrying out the IHC protocol with the c-fos antibody which had been incubated in an excess of c-fos-blocking peptide at both 4°C and at RT (data not shown). c-fos antibody was incubated for 2 h in excess of blocking peptide as recommended by the manufacturer (10 μg blocking peptide to 100 μl of serum; Oncogene Research Products, San Diego, CA, USA).
Motoneuron labeling
Choline acetyl transferase (ChAT) and heat shock protein 27 (HSP-27) antibodies were used to identify motoneurons. Adjacent sections were incubated overnight at 4°C with c-fos antibody (1:8000) or ChAT goat antibody (1:200; Chemicon International, Inc., Temecula, CA, USA). IHC was carried out as described earlier, with the exception of an additional blocking step (3% normal horse serum in 0.3% Triton X-100) before incubation in the secondary antibody. Adjacent sections of c-fos antibody (1:8000) and HSP-27 (1:100) goat antibody (Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA) were incubated overnight at RT and overnight at 4°C, respectively. An additional blocking step (3% normal horse serum in 0.3% Triton X-100) was included before incubation in secondary antibody. For double immunofluorescence (IF), sections were incubated in c-fos antibody (1:8000) and HSP-27 antibody (1:100) at 4°C overnight. After rinsing and blocking as described previously, sections were incubated in a mixture of anti-rabbit IgG (1:300) conjugated to FITC (Jackson ImmunoResearch Laboratories, Inc., West Grove, PA, USA) and anti-goat IgG conjugated to Rhodamine (Jackson ImmunoResearch Laboratories, Inc., 1:300).
Digital imaging for data analysis
Digital images were acquired using an Apogee KX-85 CCD camera (Apogee Instruments, Auburn, CA, USA), a Sony 3CCD-IRIS camera (Sony Corporation, Japan), or Insight QE camera (Diagnostic Instruments, Inc., Sterling Heights, MI, USA). A Zeiss Axiophot Microscope was used to acquire bright-field and fluorescent images. All images were taken at a low magnification of 31.25 × and a high magnification of 500 ×. C-Imaging Simple32 (Compix Inc., Cranberry Township, PA, USA), Image-Pro Plus (Media Cybernetics, Silvery Spring, MD, USA), and SPOT Advanced software (Diagnostic Instruments Inc., Sterling Heights, MI, USA) were used to acquire images with the SONY, Apogee, and SPOT InSight QE digital cameras for bright field image capture, respectively.
Quantification of FOS+ neurons
To quantify FOS+ neurons, a manual tagging feature of Image-Pro Plus was used to count the neurons. FOS+ neurons were quantified within each spinal cord lamina that was manually drawn according to Molander's map of the adult rat spinal cord.29 C-Imaging Simple32 software was used to measure soma size and the diameter of motoneurons.
Adjacently labeled sections with c-fos and either ChAT or HSP-27 using DAB or DAB + NiCl2 color reactions were counted by Image-Pro Plus using the Manual Tag settings. For double-labeled IF study of c-fos and HSP-27 neurons, composite images were generated and FOS+ motoneurons were identified and counted using the Fluoro-Pro option of the Image-Pro Plus software. FOS+ motoneuron sizes were measured using the C-Imaging software.
Statistical analysis
A Mixed model, using the PROC MIXED program with the statistical analysis software (SAS) program, was used to study the differences in FOS staining among the experimental groups. The mixed linear model is a widely used statistical method. The main difference between mixed model and ANOVA (or general linear model) is that mixed model can handle nonindependent observations. In this study, the outcome was measured for each rat at different laminae in spinal cord sections from T13 to L6. These measurements may be correlated; therefore, the mixed model is the more appropriate method than ANOVA. The model used was: Yijg=ai+bg, where Yijg is the jth measurement of FOS staining for subject i in group g, ai is the intercept for subject i and bg is the effect o of group g.30,31 The significance level was set at P <0.05. Treadmill step duration, spinal cord level, spinal cord laminae, and their interactions were included in the model, assuming that the measurements from the same rat were correlated and the covariance structure was compounded symmetrically. Since there were seven spinal cord-specific segments (T13, L1–L6) and no significant difference was observed among T13–L2 and L3–L6, the spinal cord levels were combined as rostral (T13–L2) and caudal (L3–L6). The statistical analyses were performed in two ways. First, the treadmill step duration was treated as a categorical variable and the mean of the outcome was estimated for each stepped group, each spinal cord section, and each lamina. Comparisons for trained versus nontrained, rostral versus caudal spinal cord, laminae I–III versus IV, V, and VII, and laminae I–III versus IX were performed. Second, the treadmill step duration was considered as a continuous variable and the relationship between the outcome and the stepping duration was studied. The outcome versus stepping duration plotted with the Loess fit (SAS Institute, Inc., Cary, NC, USA) showed that the relationship between these two variables was approximately linear. The slope was estimated and compared at different segments and different laminae of spinal cord. SAS was used to carry out all analyses.
Results
Stepping increases FOS
Compared to Nstp rats, Stp rats had more FOS+ neurons in all regions of the spinal cord (Figures 1, 2a, b). Figure 1 shows FOS staining in subregions of the DH, central canal (CC), and VH of lumbar spinal cord in nonstepped (Nstp), stepped (Stp) rats, and in two rats that sustained skin lesions on the paw while stepping.
Figure 1.
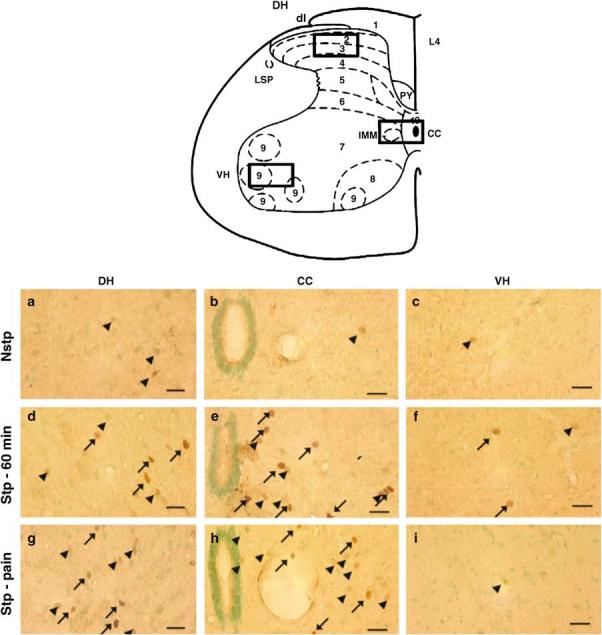
Immunohistochemistry of FOS+ neurons in the DH, CC, and VH (boxed areas on the schematic of spinal cord) of the L4 spinal cord in nonstepped (first row, a–c), stepped for 60 min (second row, d–f), and pain-associated stepping for 35 min (third row, g–i), rats. FOS+ cells found in the DH, CC, and VH. Scale bar = 10 μm. Arrows = high-intensity staining, arrowheads = low-intensity staining
Figure 2.
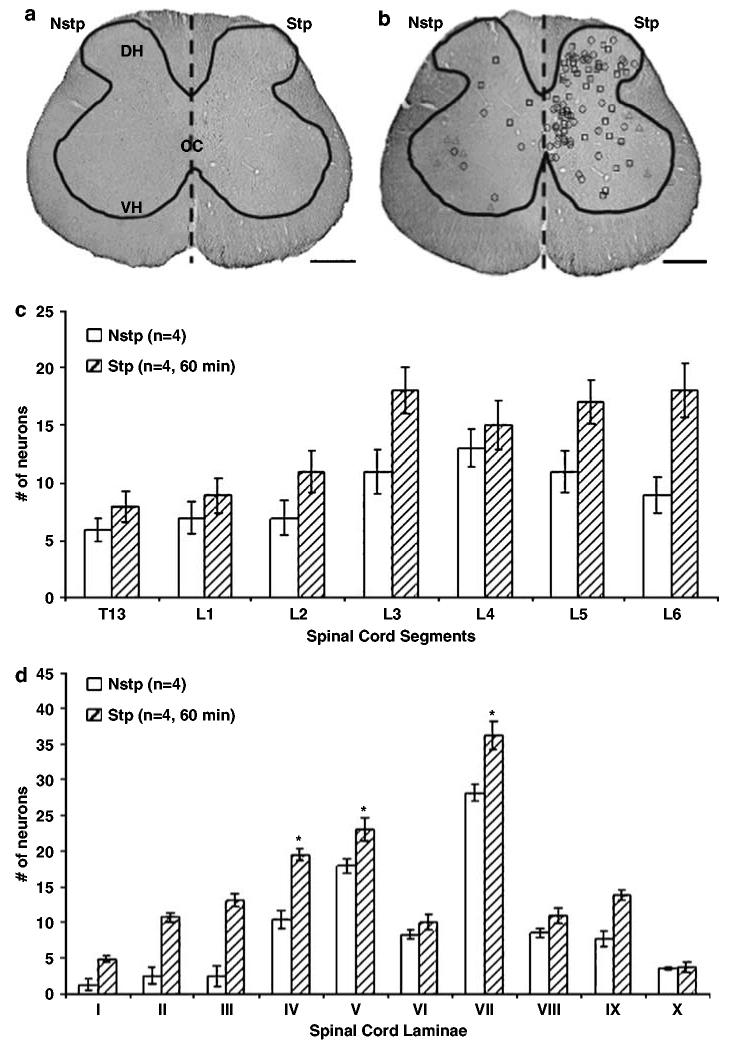
Quadrupedal treadmill stepping increases the number of FOS+ neurons. Immunostained L5 spinal cord section with c-fos antibody (a). Dashed line designates the contrast between the nonstepped (Nstp) and stepped (Stp) rat spinal cord. DH = dorsal horn, CC = central canal, VH = ventral horn. Scale bar = 100 μm. FOS+ neurons tagged in sections shown in (a) with ImagePro Plus software (b). The symbols indicate intensity of FOS staining; high (○), medium (□), low (△). The black line outlines the gray matter. Scale bar = 100 μm. Treadmill stepping for 60 min increased the number of FOS+ neurons of all segments of spinal cord T13–L6 (c), and in all spinal cord laminae I–X (d) of Stp rats compared to Nstp rats and laminae IV, V, and VII are significantly higher than other laminae within Stp rats (asterisks)
FOS-stained nuclei were evident in several regions of rat spinal cord under basal and locomotor activated conditions (Figure 1). However, in the Nstp rats, few FOS+ neurons were found within the superficial DH (Figure 1a, laminae I–III) and the area immediately outside the CC (Figure 1b). In the Stp rats, FOS+ neurons were found in all regions of the lumbar spinal cord (Figure 1d-f). The majority of FOS+ cells were observed in the DH (Figure 1b; laminae I–III) and the medial aspects of laminae IV, V, and VII (Figure 1e) with a small number of FOS+ motoneurons observed in the VH region of the lumbar spinal cord (Figure 1f, lamina IX). In the rat with the injured toe-nail during acute stepping (Figure 1g-i), the number of FOS+ neurons were slightly higher in laminae I–III (Figure 1g). More FOS+ neurons were seen in both the DH and VHs of Stp rats over Nstp rats (Figure 2a, b). Treadmill stepping increased the number of FOS+ neurons within all spinal cord segments (Figure 2c, T13–L6) and in all laminae (Figure 2d, I–X).
In Stp animals FOS increases rostrocaudally
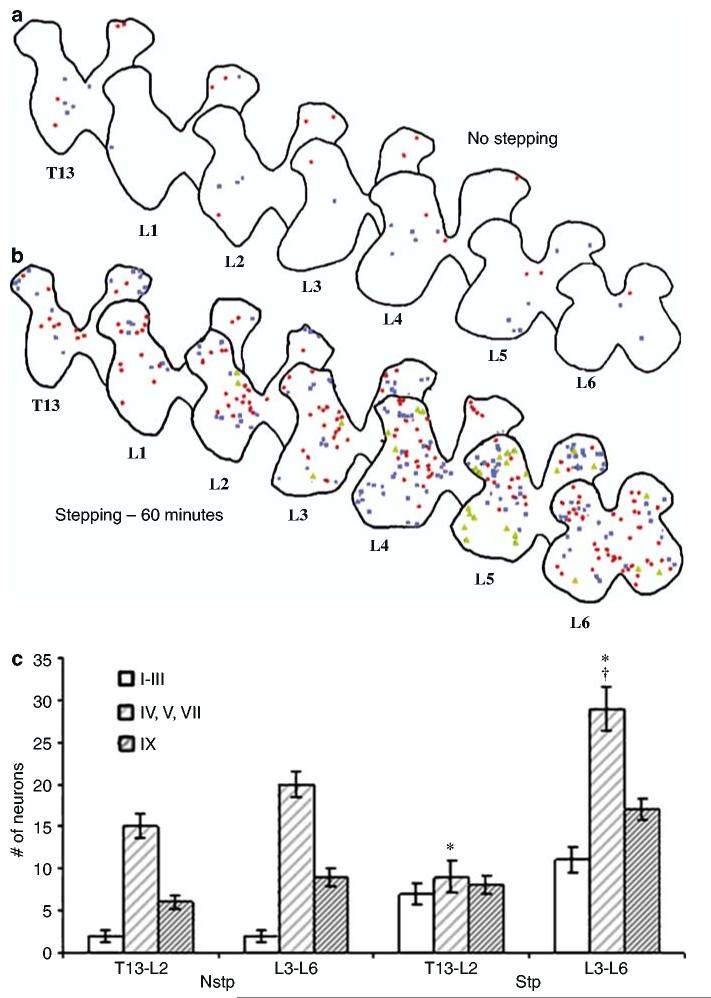
From each rat, four sections were evenly selected from the rostral to caudal spinal cord (T13–L6) for a total of 480 sections. The total number of FOS+ neurons increased in the rostrocaudal direction with treadmill stepping (Figure 3b) whereas, in the Nstp rats, FOS+ neurons were found evenly dispersed rostrocaudally (Figure 3a). The mean numbers of FOS+ neurons in spinal cord segments T13 and L1–L6 were compared. In Stp rats, there was no significant difference in the number of FOS+ neurons between spinal cord segments T13 and L1 (P = 0.533). The number of FOS+ neurons in spinal cord segment L2 was significantly higher than in T13 and L1 (P = 0.005, 0.026, respectively), but significantly lower than in segments L3–L6 (P < 0.0001). There were no significant differences between segments L3, L4, L5, and L6, but the values for L3–L6 were significantly higher than the values of T13–L2 (P < 0.0001). Given these results, we grouped T13–L2 as rostral and L3–L6 as caudal level. We then statistically compared FOS+ neurons in specific laminae in rostral and caudal spinal segments (Figure 3c, Table 2). In the rostral cord, laminae IV, V, and VII were significantly higher than laminae I–III and IX (P < 0.0001). There was no significant difference between laminae I–III and IX (P = 0.813). Similarly, in the caudal cord laminae IV, V, and VII, the number of FOS+ cells was significantly higher than in laminae I–III and IX (P < 0.0001). Again, there was no significant difference between laminae I–III and IX (P = 0.090). In laminae I–III, there were no rostrocaudal differences in numbers of FOS+ neurons (P = 0.169). However, in laminae IV, V, and VII, FOS+ neuron numbers significantly increased rostrocaudally (P < 0.0001). The number of FOS+ neurons in lamina IX also was higher in the rostral than in the caudal region (P = 0.026). Quadrupedal treadmill stepping increased the number of FOS+ neurons not only in the rostrocaudal direction but also in the dorsoventral direction within each section (Figure 4a), and this effect was most pronounced in lumbar segments L3–L5.
Figure 3.
Overall distribution of FOS+ neurons in the adult rat lumbar spinal cord. FOS+ neurons in Nstp rats appear to be diffuse, with no definitive organization seen in the rostrocaudal direction (a). After 60 min of treadmill stepping, the number of FOS+ neurons gradually increased in the rostrocaudal direction (b). Symbols indicate intensity of FOS staining: high (red circles), medium (blue squares), low (green triangles). Scale bar for (a) and (b) = 300 μm. Histogram of Nstp and Stp rats with respect to the number of neurons in the DH (laminae I–III), CC (laminae IV, V, VII), and VH (lamina IX) regions (c). FOS+ neurons were significantly higher in laminae IV, V, and VII in Stp rats only (cross). Within the Stp rats FOS+ neurons significantly increased in the rostral to caudal spinal cord in laminae IV, V, VII, and IX (asterisks)
Table 2.
Statistical analysis of rostrocaudal FOS+ neuronal distribution
| Laminae |
IV, V, VII compared to |
||||
|---|---|---|---|---|---|
| Segment level | I – III | IV, V, VII | IX | I – III | IX |
| aRostral versus bCaudal | 0.169 | <0.0001 | 0.026 | ||
| Within Rostral | <0.0001 | <0.0001 | |||
| Within Caudral | <0.0001 | <0.0001 | |||
The rostral cord is designated here as T13–L2
caudal cord as L3–L6
Segments were compared using a mixed model using the least-square means adjusting for treadmill step duration with respect to the following groups of spinal cord laminae: dorsal horn (I–III), central canal region (medial IV, V, VII), and ventral horn (IX). The P-values were calculated to determine significant relationships of the pairwise comparisons
Figure 4.
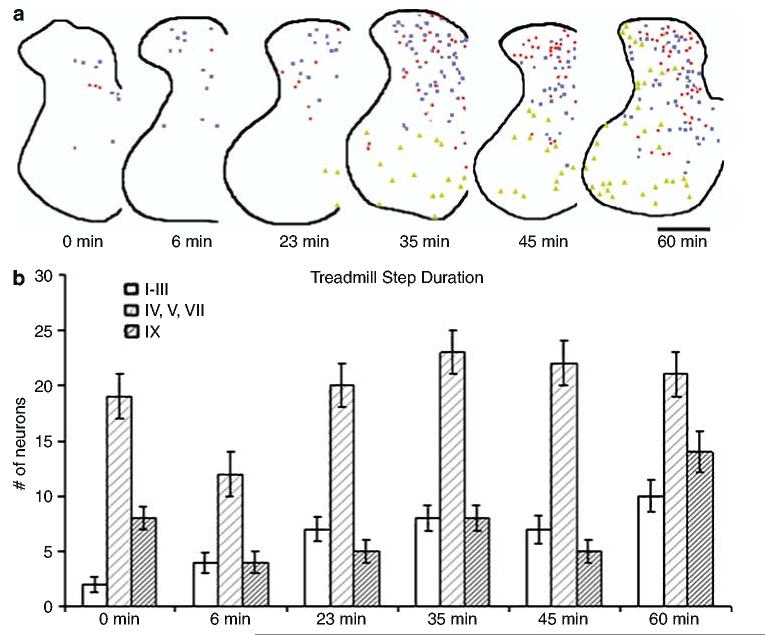
Dorsoventral distribution of FOS+ neurons relative to duration of treadmill stepping. With an increasing duration of treadmill stepping there was not only an increase in number of FOS+ neurons in the DH but also a gradual appearance of VH cells positive for FOS (a). Symbols indicate intensity of FOS staining: high (red circles), medium (blue squares), low (green triangles); black line outlines the gray matter of the spinal cord section. Scale bar = 300 μm. Laminae I–V, VII, and IX were chosen to demonstrate the trend of FOS+ neurons increasing with duration of treadmill stepping in the dorsoventral direction (b)
FOS increases with stepping duration
Overall, quadrupedally stepped rats on the treadmill at varying durations at a constant speed of 13.5 cm/s demonstrated an activity-dependent increase in FOS+ neurons (P = 0.030). Increasing the duration of stepping increased the number of FOS+ neurons in the caudal (L3–L6, P = 0.011), but not in the rostral cord (T13–L2). With increased duration of treadmill stepping, an increased number of FOS+ neurons was observed in most laminae (Figure 4a, b). This activity-dependent trend was particularly visible in lamina IX corresponding to the activation of motoneurons (Figure 4b). Only laminae IV, V, and VII showed a significant increase in the number of FOS+ neurons with increasing duration of treadmill stepping (P = 0.036, Figure 4b). In contrast, both laminae I–III (P = 0.026) and IV, V, and VII (P = 0.003) had a greater number of FOS+ neurons with increasing duration of treadmill stepping (Figure 4b). The number of FOS+ cells found in lamina X did not significantly change with varying duration of stepping. However, lamina X value was also higher than for lamina I (P = 0.0052), but no significant differences were found between laminae X and VI (P = 0.3219), or between lamina I and VI (P = 0.0670).
FOS+ motoneurons in Stp rats
Motoneurons positive for FOS were seen in lamina IX in Stp animals (Figure 5a, b). Adjacent sections stained for c-fos and ChAT showed FOS+ motoneurons to be present in lamina IX (Figure 5c, d). Adjacent sections stained for c-fos and HSP-27 also showed FOS+ motoneurons in lamina IX (Figure 5e, f). Double immunofluorescent labeling confirmed the colocalization of FOS and HSP-27 (Figure 5g-j). ChAT labeled both motoneurons and interneuron, so identifying FOS+ motoneurons by this method was difficult. Sections counterstained with methyl green showed the size, shape, and location of motoneurons, but was also not specific enough to identify FOS+ motoneurons. HSP-27-labeled motoneurons gave the most confident result in identifying FOS+ motoneurons in this study.
Figure 5.
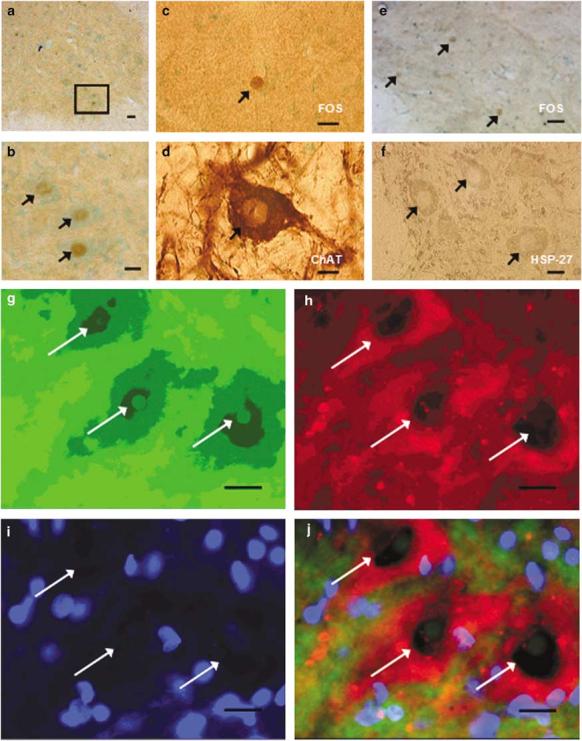
FOS+ motoneurons. Adjacent sections from L4 spinal cord of a Stp rat (60 min) labeled with c-fos, c-fos, and ChAT or HSP-27 and counterstained with methyl green for lamina IX. Distribution of FOS-stained motoneurons in the VH: brown (FOS nuclear stain with DAB), green (cytoplasmic counterstain with methyl green). Scale bar = 40 μm (a). Higher magnification of the same region from (a) with three distinct FOS+ neurons (arrows, b). Scale bar = 20 μm. FOS-stained adjacent section to (d), depicting a clearly labeled nuclear stain of neurons (arrow, c). ChAT section depicting a clear cytoplasmic stain of motoneurons (arrow, d). FOS section stained adjacent to (f) depicting a nuclear stain in three clearly labeled motoneurons (arrows, e). HSP-27 section depicting three motoneurons with clear cytoplasmic staining (arrows, f). Scale bar = 10 μm. Double IF shows FOS+ motoneurons in lamina IX (g–j). FITC – FOS (g); Rhodamine – HSP-27 (h); Dapi – nuclear stain (i); Composite – colocalization of c-fos and HSP-27 (j). Scale bar = 10 μm
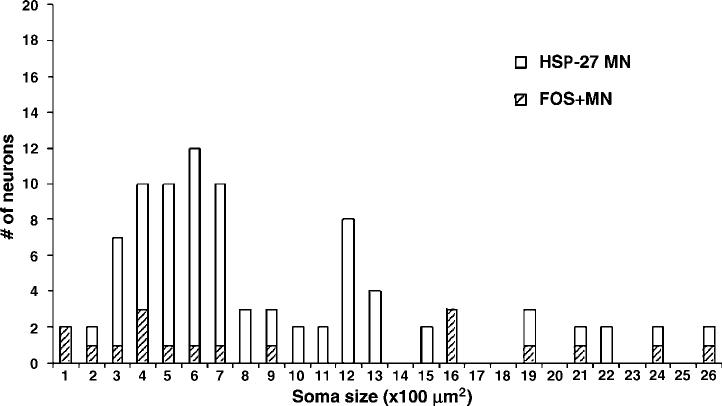
Soma sizes and diameters of labeled neurons by HSP-27 showed a bimodal distribution of large (; 65%) and small (; 35%) motoneurons (Figure 6). Soma size of HSP-27 motoneurons ranged from 175 to 3171 μm2, and the mean soma size was 761 μm2 for all measured HSP-27 neurons. The diameter ranged from 15 to 64 μm and the mean was 29 μm for all measured HSP-27 neurons. Treadmill stepping induced FOS+ neurons in 20% of HSP-27-labeled motoneurons. FOS+ HSP-27 motoneurons displayed a bimodal distribution in soma size and diameter that ranged from 160 to 3171 μm2 and from 6 to 64 μm, respectively, which was also similar to the frequency observed in FOS+ motoneurons counterstained by methyl green. The mean soma size and diameter of FOS+ HSP-27 motoneurons were 1003 μm2 and 32 μm for the Stp rats. There were too few FOS+ motoneurons observed in the Nstp rats, and thus were not included in this study.
Figure 6.
FOS+ motoneurons increase with treadmill stepping. Percentage of FOS+ motoneurons activated by stepping. Distribution of FOS+ motoneurons with respect to their soma sizes ( × 100 μm2) are shown. Motoneurons were identified using HSP-27 as a marker
Discussion
Stepping increases FOS
Following quadrupedal treadmill stepping, the number of FOS+ neurons was greater in the Stp than Nstp rats. Since nonstepped rats have few FOS+ neurons, sensory and motor inputs associated with stepping must have induced the increase in the number of FOS+ spinal neurons. Induction of FOS+ neurons occurs in decerebrate cats after electrically induced fictive locomotion22,32 and in normal intact rats after Rota-rod walking.32 However, no previous studies have addressed c-fos activation in the lumbar spinal cord after normal quadrupedal locomotion. Our preliminary study on treadmill-stepped spinally transected rats demonstrates that FOS+ neurons cluster in the region immediately outside the CC (medial laminae IV, V, and VII) and in segments L3, L4, and L5 (data not shown). These data demonstrate a pattern of activity that is similar to the last-order premotor interneurons that have been suggested to be responsible for producing locomotion.33
Step-induced FOS pattern progressed rostrocaudally in the lumbar spinal cord
The number of FOS+ neurons increased in the rostrocaudal direction with increasing duration of quadrupedal treadmill stepping. The largest number of FOS+ neurons were observed in the lumbar enlargement segments L3–L5.34,35 A similar observation was observed in MLR-evoked fictive locomotion in cats.36 Finally, a large number of FOS+ neurons were also found in the L6 segment, which innervate leg muscles and the bladder.37 This may reflect some neurally mediated link between locomotion and bladder control. In fact, it has been suggested that human spinal cord-injured patients may have improved bladder function after undergoing treadmill step training.38,39 This improvement could be due to the activation of alpha motoneurons that innervate the external urethral sphincter.40
Step-induced FOS increases in all laminae and dorsoventrally in the lumbar spinal cord
Quadrupedal treadmill stepping increased FOS numbers in neurons in all laminae (I–X) of the lumbar spinal cord. This is consistent with the presence of various types of stimuli that occur during treadmill locomotion, such as proprioception, cutaneous inputs from the plantar surface of the rats' paw, and the action of locomotion itself.2,3 FOS+ neurons were first observed in the DH region and at longer durations of treadmill stepping more neurons became activated near the CC, and after 60 min of treadmill stepping, activated neurons appeared in the VH. With increased duration of treadmill stepping, more FOS+ neurons appeared in the dorsoventral direction. This pattern may reflect a progression of dorsally derived sensory stimulation projected to the more ventral motoneurons. An alternative explanation is that the neurons are progressively less responsive to those signals that induce c-fos from a dorsal to a ventral perspective.
Activity-dependent increase of FOS
The number of FOS+ neurons was directly related to the duration of treadmill stepping; more FOS+ neurons were seen with increasing duration of stepping. This is similar to what was observed in decerebrate cats after fictive locomotion22 and in walking on Rota-rods in control and deafferented rats.32 Activity-dependent increases in FOS have been observed in other systems as well, particularly in the well-defined visual system. For example, c-fos activity dependency has been demonstrated after varying durations of light stimulation, preceded by 3 days of dark in the rat visual cortex.41 In their study, longer durations of light stimulation produces greater number of FOS+ cells.
FOS in laminae I–III
In Stp rats, FOS+ neurons in laminae I–III increased in the rostrocaudal direction in segments L3–L6 (P = 0.026) with increased duration of treadmill stepping, again demonstrating that there were more sensory-related neurons activated with increasing duration of treadmill stepping. In order to identify locomotor-activated neurons with minimum contribution from noxious stimulation, we stepped our rats at a moderate speed of 13.5 cm/s to avoid friction-related injuries to hind paws. For example, pain-related c-fos induction has been observed in laminae I–III, VII–VIII and X after somatic noxious stimulation and the superficial dorsal laminae (I–III) after visceral noxious stimulation to the rat hind paw.42
FOS in laminae IV, V, and VII
The greatest number and increase of FOS+ neurons occurred in laminae IV, V, and VII, with the most significant increase in the medial aspect of lamina VII and just adjacent to lamina X in the lumbar spinal cord. Presumably, these laminae are associated with last-order premotor interneurons activated during a locomotor task.43 The appearance of FOS+ neurons surrounding the CC can also be the result of somatic and visceral afferents associated with cutaneous inputs from the plantar surface of rats' feet and visceral organ activation, such as the bladder, during treadmill stepping.40 Following MLR-evoked fictive locomotion in cats, laminae VI–IX had the largest increases in FOS+ neurons.44 After MLR-evoked fictive and treadmill locomotion in decerebrated cats, laminae VII–IX had the largest number of FOS+ neurons.22,36 Interneurons in laminae V–VIII and X have been shown to project to the L7 motor nuclei in cats.45 In the Rota-rod-evoked locomotion, significant numbers of FOS+ neurons were found in the medial aspects of laminae II, V–VII, and in the lateral aspect of lamina IX.32 In all of these studies FOS neurons were observed in lamina VII. However, none of these studies report an increase in lamina IV FOS+ neurons. These increases in lamina IV are consistent with afferent inputs from quadrupedal treadmill stepping playing an important role in eliciting the activation of lamina VII interneurons and in turn activating alpha motoneurons, as seen in lamina IX of this study. This also suggests that the activation of locomotor interneurons in lamina VII can be elicited with quadrupedal treadmill stepping, and that afferent inputs to the DH are important for producing stepping in intact adult rats.
FOS in lamina IX
ChAT and HSP-27 were used to identify motoneurons. ChAT is a marker of cholinergic motoneurons and interneurons,46 while HSP-27 labels motoneurons only.47 There were approximately 30 neurons per section stained with HSP-27, whereas ChAT-labeled sections averaged around 60 neurons per section. The number of FOS+ motoneurons increased with increasing duration of treadmill locomotion. A third of the ChAT-positive neurons also stained for FOS following 7–9 h of MLR stimulation.22 After cat fictive locomotion, 1–2% of the motoneurons in the lumbar spinal cord stained positive with FOS.44 Quantification of cells using Image-Pro Plus and C-imaging show that 20% of HSP-27-labeled motoneurons were FOS+ after a single 60-min bout of treadmill stepping. The soma sizes were measured to determine the type of motoneurons that were being activated during quadrupedal treadmill stepping. The bimodal distribution of FOS+ motoneuron soma sizes indicates that there was an activation of both large (alpha) and small (alpha and gamma) motoneurons with this motor task. With respect to the activation of motor columns after stepping, we observe FOS+ cells largely within the dorsolateral region in L3–L5 segments, which we speculate as the innervations to the quadriceps, interior foot (medial plantar), and posterior accessory muscles, respectively. This observation however, is qualitative and there are still considerable amounts of FOS+ cells found in the lateral region of the VH as well, which correlate with gluteal and hamstring (semitendinosus, semimembranosus, and biceps femoris) muscles.48
Application of FOS to other future studies
Identification of activated neurons by c-fos mapping can guide future studies designed to elucidate the pathways related to spinal learning in spinally transected animals. For example, our preliminary studies (unpublished) from a parallel study of stepping in spinal cord transected rats show that the FOS+ neurons during locomotion in the lumbar spinal cord of intact and spinally transected rats overlap with those neurons that express molecules such as phosphorylated cyclic AMP response element binding protein (pCREB), N-methyl-D-aspartate (NMDA) receptor subunits, and calcium calmodulin kinase (CaMKII), all of which are involved in hippocampal learning.49,50
Conclusion
The number of FOS+ neurons increased with the duration of treadmill stepping. There was a rostrocaudal and a dorsoventral increase in FOS-labeled neurons, which was proportional to the duration of stepping. The most significant increases in FOS+ neurons were found in the region immediately lateral to the CC in laminae IV, V, and VII. This suggests that the interneurons in this region play an important role in the production of quadrupedal locomotor behavior. The number of FOS+ motoneurons clearly increased with treadmill stepping. After midthoracic spinal cord transection, all supraspinal input becomes absent and thus it is no longer possible to produce voluntary movement. However, the local spinal cord neural circuitry can still respond to sensory inputs independent of supraspinal control. Future experiments will allow us to compare the neurons that are involved in the local spinal cord neural circuitry, which can generate involuntary locomotion.
Acknowledgements
We thank Maynor Herrera for help in animal care and Charlotte Liu, Matthias Ziegler, Dr Noore Ali and Joseph Raven for assisting treadmill stepping of the animals. We thank Drs Robert Elashoff and He-Jing Wang of the UCLA Biomath Department for statistical analysis of the data.
References
- 1.Alberini CM, Ghirardi M, Huang YY, Nguyen PV, Kandel ER. A molecular switch for the consolidation of long-term memory: cAMP-inducible gene expression. Ann N Y Acad Sci. 1995;758:261–286. doi: 10.1111/j.1749-6632.1995.tb24833.x. [DOI] [PubMed] [Google Scholar]
- 2.Chiasson BJ, Hong MG, Robertson HA. Putative roles for the inducible transcription factor c-fos in the central nervous system: studies with antisense oligonucleotides. Neurochem Int. 1997;31:459–475. doi: 10.1016/s0197-0186(96)00115-5. [DOI] [PubMed] [Google Scholar]
- 3.Morgan JI, Curran T. Stimulus–transcription coupling in the nervous system: involvement of the inducible proto-oncogenes fos and jun. Annu Rev Neurosci. 1991;14:421–451. doi: 10.1146/annurev.ne.14.030191.002225. [DOI] [PubMed] [Google Scholar]
- 4.Craner SL, Hoffman GE, Lund JS, Humphrey AL, Lund RD. cFos labeling in rat superior colliculus: activation by normal retinal pathways and pathways from intracranial retinal transplants. Exp Neurol. 1992;117:219–229. doi: 10.1016/0014-4886(92)90131-9. [DOI] [PubMed] [Google Scholar]
- 5.Hannibal J, Vrang N, Card JP, Fahrenkrug J. Light-dependent induction of cFos during subjective day and night in PACAP-containing ganglion cells of the retino-hypothalamic tract. J Biol Rhythms. 2001;16:457–470. doi: 10.1177/074873001129002132. [DOI] [PubMed] [Google Scholar]
- 6.Kaczmarek L, Chaudhuri A. Sensory regulation of immediate-early gene expression in mammalian visual cortex: implications for functional mapping and neural plasticity. Brain Res Rev. 1997;23:237–256. doi: 10.1016/s0165-0173(97)00005-2. [DOI] [PubMed] [Google Scholar]
- 7.Kippin TE, Cain SW, Pfaus JG. Estrous odors and sexually conditioned neutral odors activate separate neural pathways in the male rat. Neuroscience. 2003;117:971–979. doi: 10.1016/s0306-4522(02)00972-7. [DOI] [PubMed] [Google Scholar]
- 8.Lau YE, Cherry JA, Baum MJ, Mani SK. Induction of Fos in the accessory olfactory system by male odors persists in female mice with a null mutation of the aromatase (cyp19) gene. Brain Res Bull. 2003;60:143–150. doi: 10.1016/s0361-9230(03)00024-8. [DOI] [PubMed] [Google Scholar]
- 9.Amir S, Cain S, Sullivan J, Robinson B, Stewart J. Olfactory stimulation enhances light-induced phase shifts in free-running activity rhythms and Fos expression in the suprachiasmatic nucleus. Neuroscience. 1999;92:1165–1170. doi: 10.1016/s0306-4522(99)00222-5. [DOI] [PubMed] [Google Scholar]
- 10.Jahng JW, Choi SH, Kim DG, Houpt TA. Central Nomega-nitro-L-arginine methyl ester does not influence lithium-induced c-Fos and conditioned taste aversion. Yonsei Med J. 2003;44:869–874. doi: 10.3349/ymj.2003.44.5.869. [DOI] [PubMed] [Google Scholar]
- 11.Serova LI, Saez E, Spiegelman BM, Sabban EL. c-Fos deficiency inhibits induction of mRNA for some, but not all, neurotransmitter biosynthetic enzymes by immobilization stress. J Neurochem. 1998;70:1935–1940. doi: 10.1046/j.1471-4159.1998.70051935.x. [DOI] [PubMed] [Google Scholar]
- 12.Rachman IM, Unnerstall JR, Pfaff DW, Cohen RS. Estrogen alters behavior and forebrain c-fos expression in ovariectomized rats subjected to the forced swim test. Proc Natl Acad Sci USA. 1998;95:13941–13946. doi: 10.1073/pnas.95.23.13941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bennett HJ, Semba K. Immunohistochemical localization of caffeine-induced c-Fos protein expression in the rat brain. J Comp Neurol. 1998;401:89–108. doi: 10.1002/(sici)1096-9861(19981109)401:1<89::aid-cne6>3.0.co;2-x. [DOI] [PubMed] [Google Scholar]
- 14.Chen T, Dong YX, Li YQ. Fos expression in serotonergic neurons in the rat brainstem following noxious stimuli: an immunohistochemical double-labelling study. J Anat. 2003;203:579–588. doi: 10.1046/j.1469-7580.2003.00242.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hu M, Liu QS, Chang KT, Berg DK. Nicotinic regulation of CREB activation in hippocampal neurons by glutamatergic and nonglutamatergic pathways. Mol Cell Neurosci. 2002;21:616–625. doi: 10.1006/mcne.2002.1202. [DOI] [PubMed] [Google Scholar]
- 16.Brudzynski SM, Wang D. C-Fos immunohistochemical localization of neurons in the mesencephalic locomotor region in the rat brain. Neuroscience. 1996;75:793–803. doi: 10.1016/0306-4522(96)00284-9. [DOI] [PubMed] [Google Scholar]
- 17.Ichiyama RM, Gilbert AB, Waldrop TG, Iwamoto GA. Changes in the exercise activation of diencephalic and brainstem cardiorespiratory areas after training. Brain Res. 2002;947:225–233. doi: 10.1016/s0006-8993(02)02929-3. [DOI] [PubMed] [Google Scholar]
- 18.Iwamoto GA, Wappel SM, Fox GM, Buetow KA, Waldrop TG. Identification of diencephalic and brainstem cardiorespiratory areas activated during exercise. Brain Res. 1996;726:109–122. [PubMed] [Google Scholar]
- 19.Liste I, Guerra MJ, Caruncho HJ, Labandeira-Garcia JL. Treadmill running induces striatal Fos expression via NMDA glutamate and dopamine receptors. Exp Brain Res. 1997;115:458–468. doi: 10.1007/pl00005715. [DOI] [PubMed] [Google Scholar]
- 20.Roberts AC, Glanzman DL. Learning in aplysia: looking at synaptic plasticity from both sides. Trends Neurosci. 2003;26:662–670. doi: 10.1016/j.tins.2003.09.014. [DOI] [PubMed] [Google Scholar]
- 21.Tillakaratne NJ, de Leon RD, Hoang TX, Roy RR, Edgerton VR, Tobin AJ. Use-dependent modulation of inhibitory capacity in the feline lumbar spinal cord. J Neurosci. 2002;22:3130–3143. doi: 10.1523/JNEUROSCI.22-08-03130.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Huang A, Noga BR, Carr PA, Fedirchuk B, Jordan LM. Spinal cholinergic neurons activated during locomotion: localization and electrophysiological characterization. J Neurophysiol. 2000;83:3537–3547. doi: 10.1152/jn.2000.83.6.3537. [DOI] [PubMed] [Google Scholar]
- 23.Shah RR, Liu CC, Bigbee AJ, Roy RR, Tobin AJ, Edgerton VR, Tillakaratne NJK. Does Stepping Similarly Activate the Lumbar Locomotor Circuitry in Spinal Cord Transected and Intact Rats? University of California; Los Angeles: 2002. [Google Scholar]
- 24.Ahn SN, Guu JJ, Tobin AJ, Edgerton VR, Tillakaratne NJK. Use of cfos to Map Spinal Neurons Associated with Locomotion in Rats. University of California; Los Angeles: 2003. [Google Scholar]
- 25.Ahn SN, Guu JJ, Tobin AJ, Edgerton VR, Tillakaratne NJK. Use of c-fos to Identify Locomotor Associated Spinal Neurons Activated During Treadmill Stepping in Adult Rats. University of California; Los Angeles: 2004. [Google Scholar]
- 26.Zhang RX, Wang H, Ruda M, Iadarola MJ, Qiao JT. c-Fos expression in NMDA receptor-contained neurons in spinal cord in a rat model of inflammation: a double immunocytochemical study. Brain Res. 1998;795:282–286. doi: 10.1016/s0006-8993(98)00300-x. [DOI] [PubMed] [Google Scholar]
- 27.Schadrack J, Castro-Lopes JM, Avelino A, Zieglgansberger W, Tolle TR. Modulated expression of c-Fos in the spinal cord following noxious thermal stimulation of monoarthritic rats. J Neurosci Res. 1998;53:203–213. doi: 10.1002/(SICI)1097-4547(19980715)53:2<203::AID-JNR9>3.0.CO;2-B. [DOI] [PubMed] [Google Scholar]
- 28.Pilyavskii AI, et al. c-fos Expression and NADPH-d reactivity in spinal neurons after fatiguing stimulation of hindlimb muscles in the rat. Brain Res. 2001;923:91–102. doi: 10.1016/s0006-8993(01)03049-9. [DOI] [PubMed] [Google Scholar]
- 29.Molander C, Xu Q, Grant G. The cytoarchitectonic organization of the spinal cord in the rat. I. The lower thoracic and lumbosacral cord. J Comp Neurol. 1984;230:133–141. doi: 10.1002/cne.902300112. [DOI] [PubMed] [Google Scholar]
- 30.Tillakaratne NJ, Mouria M, Ziv NB, Roy RR, Edgerton VR, Tobin AJ. Increased expression of glutamate decarboxylase (GAD(67)) in feline lumbar spinal cord after complete thoracic spinal cord transection. J Neurosci Res. 2000;60:219–230. doi: 10.1002/(SICI)1097-4547(20000415)60:2<219::AID-JNR11>3.0.CO;2-F. [DOI] [PubMed] [Google Scholar]
- 31.Tibshirani BE, Tibshirani RJ. An Introduction to the Bootstrap. Chapman & Hall; New York: 1993. [Google Scholar]
- 32.Jasmin L, Gogas KR, Ahlgren SC, Levine JD, Basbaum AI. Walking evokes a distinctive pattern of Fos-like immunoreactivity in the caudal brainstem and spinal cord of the rat. Neuroscience. 1994;58:275–286. doi: 10.1016/0306-4522(94)90034-5. [DOI] [PubMed] [Google Scholar]
- 33.Morales FR, Sampogna S, Yamuy J, Chase MH. c-fos expression in brainstem premotor interneurons during cholinergically induced active sleep in the cat. J Neurosci. 1999;19:9508–9518. doi: 10.1523/JNEUROSCI.19-21-09508.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nicolopoulos-Stournaras S, Iles JF. Motor neuron columns in the lumbar spinal cord of the rat. J Comp Neurol. 1983;217:75–85. doi: 10.1002/cne.902170107. [DOI] [PubMed] [Google Scholar]
- 35.Vanderhorst VG, Holstege G. Organization of lumbosacral motoneuronal cell groups innervating hindlimb, pelvic floor, and axial muscles in the cat. J Comp Neurol. 1997;382:46–76. [PubMed] [Google Scholar]
- 36.Dai XNB, Douglas JR, Jordan LM. Localisation of spinal neurons activated during locomotion using the c-fos immunohistochemical method. J Neurophysio. 2005;93:3442–3452. doi: 10.1152/jn.00578.2004. [DOI] [PubMed] [Google Scholar]
- 37.Marson L. Identification of central nervous system neurons that innervate the bladder body, bladder base, or external urethral sphincter of female rats: a transneuronal tracing study using pseudorabies virus. J Comp Neurol. 1997;389:584–602. [PubMed] [Google Scholar]
- 38.Harkema SJ, Dobkin B, Edgerton VR. Pattern generators in locomotion: implications for recovery of walking after spinal cord injury. Top Spinal Cord Injury Rehabil. 2000;6:82–96. [Google Scholar]
- 39.Sadowsky CL. Electrical stimulation in spinal cord injury. NeuroRehabilitation. 2001;16:165–169. [PubMed] [Google Scholar]
- 40.Honda CN. Visceral and somatic afferent convergence onto neurons near the central canal in the sacral spinal cord of the cat. J Neurophysiol. 1985;53:1059–1078. doi: 10.1152/jn.1985.53.4.1059. [DOI] [PubMed] [Google Scholar]
- 41.Zangenehpour S, Chaudhuri A. Differential induction and decay curves of c-fos and zif268 revealed through dual activity maps. Brain Res Mol Brain Res. 2002;109:221–225. doi: 10.1016/s0169-328x(02)00556-9. [DOI] [PubMed] [Google Scholar]
- 42.Menetrey D, Gannon A, Levine JD, Basbaum AI. Expression of c-fos protein in interneurons and projection neurons of the rat spinal cord in response to noxious somatic, articular, and visceral stimulation. J Comp Neurol. 1989;285:177–195. doi: 10.1002/cne.902850203. [DOI] [PubMed] [Google Scholar]
- 43.Puskar Z, Antal M. Localization of last-order premotor interneurons in the lumbar spinal cord of rats. J Comp Neurol. 1997;389:377–389. doi: 10.1002/(sici)1096-9861(19971222)389:3<377::aid-cne2>3.0.co;2-y. [DOI] [PubMed] [Google Scholar]
- 44.Carr PA, Huang A, Noga BR, Jordan LM. Cytochemical characteristics of cat spinal neurons activated during fictive locomotion. Brain Res Bull. 1995;37:213–218. doi: 10.1016/0361-9230(94)00271-2. [DOI] [PubMed] [Google Scholar]
- 45.Hoover JE, Durkovic RG. Retrograde labeling of lumbosacral interneurons following injections of red and green fluorescent microspheres into hindlimb motor nuclei of the cat. Somatosens Mot Res. 1992;9:211–226. doi: 10.3109/08990229209144772. [DOI] [PubMed] [Google Scholar]
- 46.Barber RP, Phelps PE, Houser CR, Crawford GD, Salvaterra PM, Vaughn JE. The morphology and distribution of neurons containing choline acetyltransferase in the adult rat spinal cord: an immunocytochemical study. J Comp Neurol. 1984;229:329–346. doi: 10.1002/cne.902290305. [DOI] [PubMed] [Google Scholar]
- 47.Kalmar B, Burnstock G, Vrbova G, Greensmith L. The effect of neonatal nerve injury on the expression of heat shock proteins in developing rat motoneurones. J Neurotrauma. 2002;19:667–679. doi: 10.1089/089771502753754127. [DOI] [PubMed] [Google Scholar]
- 48.Peyronnard JM, Charron LF, Lavoie J, Messier JP. Motor, sympathetic and sensory innervation of rat skeletal muscles. Brain Res. 1986;373:288–302. doi: 10.1016/0006-8993(86)90343-4. [DOI] [PubMed] [Google Scholar]
- 49.Ali NJ, Guu JJ, Liu CC, Roy RR, Tobin AJ, Edgerton VR, Tillakaratne NJK. Increased Expression of N-Methyl-D-Aspartate Subunits in Spinal Dendrites After a Single Bout of Bipedal Stepping Following Complete Spinal Transection. University of California; Los Angeles: 2004. [Google Scholar]
- 50.Melikian RLC, et al. Increased Levels of PCREB in Motoneurons of Spinal Cord Transected Rats Following Step Training. University of California; Los Angeles: 2004. [Google Scholar]