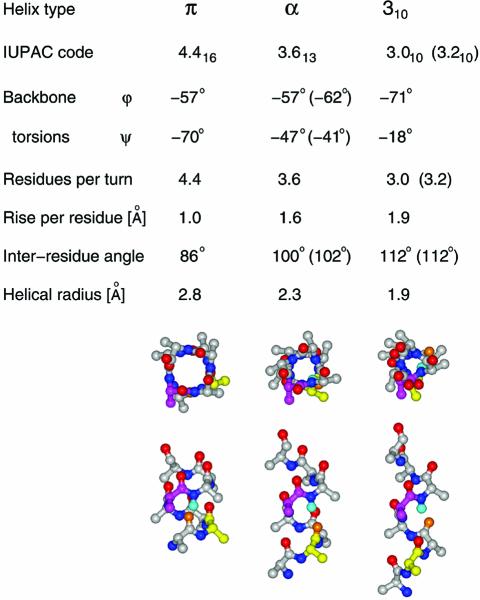
Table 1. Parameters characterizing non-canonical conformations.
See Pauling et al. (19) and Barlow and Thornton (20) for explanations of theoretical (–57, –47) and natural (–62, –41) α-helical backbone torsions, and the theoretical (3.0) and the actual (3.2) ‘3.0’ helix. Backbone stick diagrams are those of nonaalanine peptide in π, α and 310 conformations, with residues 2 and 5 color coded yellow and magenta. The amide hydrogen of residue 6 is shown in cyan and the closest carbonyl oxygen that can form a backbone hydrogen bond is shown in orange.