FIGURE 4. Effect of phosphorylation on Ser/Thr-Pro sites on the PA phos-phatase activity of Pah1p.
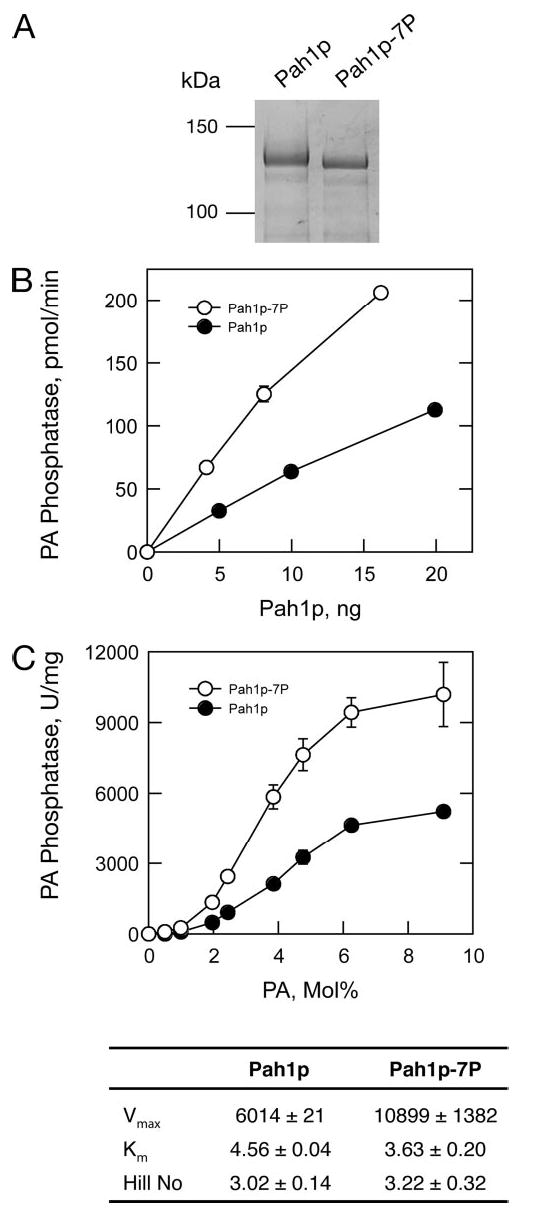
A, SDS-PAGE of the purified untagged Pah1p and Pah1p-7P from wild-type yeast cells, stained with Coomassie Blue. The positions of the molecular mass standards are indicated. B, the Mg2+-dependent PA phosphatase activity of the purified Pah1p and Pah1p-7P proteins was measured with the indicated protein contents. The values shown were determined from triplicate enzyme determinations (errors fall within the size of the circles). C, effect of PA surface concentration on Pah1p- and Pah1p-7P-dependent PA phosphatase activity. The indicated purified Pah1p and Pah1p-7P proteins from yeast were assayed for Mg2+-dependent PA phos-phatase activity at the indicated surface concentrations (mol %) of PA. The molar concentration was held constant at 0.2 mM. The values shown were determined from triplicate enzyme determinations ± S.D. Lower panel, the kinetic parameters for the Pah1p and Pah1p-7P samples analyzed in B and C are shown.
