Abstract
Aims: To test the ability of Ki-67 to detect cytological lesions in a screening setting and its use as a surrogate marker of human papillomavirus (HPV) infection.
Methods: A study of liquid based cytology, HPV DNA testing by MY09/MY11 consensus polymerase chain reaction (PCR), type specific PCRs, and Ki-67 immunocytochemistry on a randomly selected series of 147 patients.
Results: Comparison of the number of Ki-67 immunoreactive cells/1000 cells in the different cytological groups showed that the HSIL group yielded a significantly higher mean count than did the other groups. The number of Ki-67 immunoreactive cells/1000 cells was significantly higher in HPV-16 positive samples than in samples containing infections with other high risk types. Receiver operating characteristic curves indicated a test accuracy (area under curve) of 0.68, 0.72, and 0.86 for atypical squamous cells of undetermined significance (ASCUS), low grade squamous intraepithelial lesions (LSIL), and high grade squamous intraepithelial lesions (HSIL), respectively. Thresholds for 95% sensitivity were 0.07, 0.08, and 0.15 Ki-67 immunopositive cells/1000 cells for ASCUS, LSIL and HSIL, respectively. The threshold for 95% specificity was 1.9 Ki-67 immunopositive cells/1000 cells.
Conclusions: Ki-67 immunocytochemistry can be applied to liquid based cytology. The accuracy and diagnostic indices of the test are good when compared with those of other techniques. As part of a panel of screening procedures, it could be used as an adjunct to liquid based cytology to identify HSIL, and as a surrogate marker of HPV-16 infection.
Keywords: cervical cytology, human papillomavirus, polymerase chain reaction, Ki-67
The causal relation between genital human papillomavirus (HPV) infection and cervical dysplasia/carcinoma is well established.1–3 HPV type 16 (HPV-16) is most frequently found in cervical squamous cell carcinoma, with more than 50% of these tumours harbouring this type.4 However, even though the prevalence of HPV infection can be as high as 60%, only 10% of infected women will develop cervical dysplasia.5,6
The infection of cervical cells by HPV manifests itself by changes in the function or expression of host genes, and the detection of these alterations can play a role in screening and diagnosis. The different levels of association with cancer of different HPV types presumably reflects the potency of their respective oncoproteins encoded by the E6 and E7 genes, which, by binding to host regulatory proteins, lead to degradation of the p53 tumour suppressor gene product and the inactivation of the retinoblastoma gene protein.7,8 These interactions cause deregulation of the cell cycle, manifested by abnormal expression of cell cycle associated proteins, such as Ki-67.8 The detection of abnormal expression can identify clinically important cases of HPV infection with risk of progression towards dysplasia and carcinoma.
Ki-67 expression is normally confined to the basal and parabasal layers of the normal cervical squamous epithelium.9 In dysplasia and carcinoma, expression extends above the basal one third of the epithelium and the number of positive cells increases.8
Over the past decade, many large prospective studies have investigated the prognostic value of Ki-67 in the outcome of cervical carcinoma.10–21 Ki-67 has been used in routine cervical pathology to assess adequate excision of cervical intraepithelial neoplasia (CIN) and to differentiate between postmenopausal atrophy and CIN.22
“In dysplasia and carcinoma, Ki-67 expression extends above the basal one third of the epithelium and the number of positive cells increases”
However, few studies have investigated the application of Ki-67 in cervical cytology using classic Papanicolaou (Pap) smears,23,24 although the results have been encouraging. With the introduction of new technology, screening for cervical cancer precursors has evolved. By using a combination of methods, both screening and cost effectiveness could be improved.25 Within the setting of these new developments, we investigated the possibility of applying Ki-67 to liquid based samples. The potential of using Ki-67 to identify cytological abnormalities and its possible use as a surrogate marker for HPV infection were investigated.
METHODS AND MATERIALS
Study population
The Laboratory of Clinical Pathology, Antwerp, derives its routine screening samples from general practitioners and gynaecologists in Flanders (Belgium).26,27 From routine screening, a series of 150 samples showing a spectrum of cytological abnormalities and tested for HPV DNA by the polymerase chain reaction (PCR) was selected at random. This series was then stained for Ki-67.
All investigations were performed in accordance with the guidelines of the local ethical committee. Informed consent was not obtained from the patients because no risks were attributable to our study and patients had the opportunity to decline counselling. Study specific patient identification codes were assigned and transmitted in such a way that patient confidentiality was preserved.
Laboratory assays
Cytology
Cervical cells were collected using the Cervex-Brush™ (Rovers, Oss, the Netherlands) and placed immediately in alcohol based preservative (Tripath Imaging, Burlington, North Carolina, USA). Thin layer liquid based cytology (LBC) preparations (Tripath Imaging) were made with the fully robotic AutoCyte™ PREP system.28 From a starting volume of 1000 μl for each sample, a 200 μl aliquot was used to prepare each LBC.
All slides were manually screened by cytotechnologists after intensive training in the evaluation of thin layer slides. Five percent of all LBCs, in addition to all abnormal and suspicious cases, were reviewed by senior cytotechnologists. Abnormal or dubious cases were forwarded to one of the pathologists for final diagnosis.
The cytological results were classified according to the Bethesda 1992 system,29 using the following classes: within normal limits (WNL), atypical squamous cells of undetermined significance (ASCUS), low grade squamous intraepithelial lesions (LSIL), high grade squamous intraepithelial lesions (HSIL), and atypical glandular cells of undetermined significance.
HPV DNA testing by PCR
As reported previously, from the remaining cell suspension of 800 μl, 400 μl was transferred to an Eppendorf tube and the cells were pelleted by centrifugation.27 The supernatant was discarded and the pellet resuspended in 50 μl of digestion solution (10mM Tris, 1mM EDTA, and 200 μg/ml proteinase K) and digested for three hours at 56°C. The digestion was followed by a 10 minute incubation at 95°C to inactivate the proteinase K. The DNA extracts were stored at −20°C until PCR was performed.
All samples were tested with MY09/MY11 consensus primers.30 Typing was performed with type specific PCRs for oncogenic types (HPV-16, HPV-18, HPV-31, HPV-33, HPV-35, HPV-39, HPV-45, HPV-51, HPV-52, HPV-56, HPV-58, HPV-59, HPV-66, and HPV-68).2 Samples that were positive for the consensus PCR, but negative for the type specific primers, were classified as unidentified HPV genotypes of unknown malignant potential.
Immunocytochemistry
From the remaining 400 μl cell suspension after HPV DNA detection, 50 μl was used to make thin layer preparations with the Cytospin3™ cytocentrifuge (ThermoShandon, Astmoor Runcorn, Cheshire, UK) on poly-L-lysine-coated glass slides. This volume was determined in a pilot study as the lowest volume with which it was possible to prepare an adequate cytospin. Preparations were fixed in methanol.
Immunocytochemistry was performed with the prediluted primary anti-Ki-67 antibody (Dako, Glostrup, Denmark), and the slides were incubated for 30 minutes. The secondary goat antirabbit antibody was applied for 25 minutes (1/500 dilution; Dako). Slides were treated with the streptavidin–biotin complex reagent (Dako) for 25 minutes and the reaction product was developed with 0.4% diaminobenzidine/H2O2. All steps were performed at room temperature and slides were washed in phosphate buffered saline. Slides were counterstained with haematoxylin.
Evaluation of the immunocytochemistry was done by light microscopy. All cells in one high power field (×400), which was considered to be representative of the whole cell area, were counted, and the total amount of cells on the slide was assessed. This method was tested in a pilot study and was based on the recommendations of the Bethesda system 2001 concerning the assessment of LBC cellularity.31 Because there was no significant difference in results when counting one, two, or five high power fields, we decided to count one field only. All positively stained cells in the whole cell area were counted and the fraction of positive cells on the slide was calculated. This fraction was expressed as the number of positive cells/1000 cells to compare results of all samples.
Cells were considered immunopositive if the nucleus showed homogeneous or punctate staining. The staining intensity was not graded to avoid subjective interpretation. Cytoplasmic staining without nuclear staining was not considered to be positive.
Statistical analysis
Using the software package Graphpad Prism 3™ (Graphpad Software, San Diego, California, USA) a Kruskal-Wallis test, followed by Dunn’s multiple comparison post-test, was performed. A p value < 0.05 was considered to be significant. All analysed data are expressed as mean number and standard error of the mean (SEM) of immunopositive cells/1000 cells.
Receiver operating characteristic (ROC) plots were generated using SPSS 10™ (SPSS, Chicago, Illinois, USA) to determine the diagnostic accuracy of the Ki-67 immunocytochemistry test.32 Sensitivity and specificity were calculated on the premise that true negative cases were those cases of WNL without infection with a high risk HPV type. When determining the diagnostic accuracy of Ki-67 immunocytochemistry for all abnormal cytology, true positive cases were defined as those cases with abnormal cytology (ASCUS or worse) and infection with a high risk HPV type. When determining the accuracy of the test for LSIL or worse, true positive cases were defined as those cases with a diagnosis of LSIL or worse and infection with a high risk HPV type. When determining the accuracy of the test for HSIL, true positive cases were defined as those cases with a diagnosis of HSIL and infection with a high risk HPV type (all HSIL cases).
RESULTS
Ki-67 immunostaining
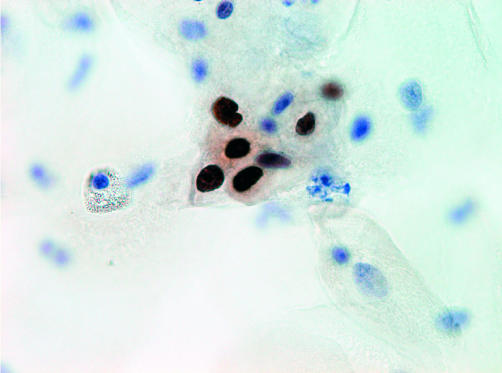
Immunoreactive cells showed dark brown, homogeneous or punctate staining, limited exclusively to the nucleus (fig 1). Enlarged naked nuclei that stained positive were included in the count. In some cases of severe inflammation, neutrophilic granulocytes were stained, which were not included in the count. Sometimes, immunopositive cells could be identified as (para-)basal cells, but there was also staining of intermediate or superficial cells. In several cases, clearly dysplastic cells were stained. Three smears were excluded because the cell preparation was not thin layered and cells were obscured by overlying layers.
Figure 1.
Ki-67 immunocytochemistry. A group of immunopositive cells in a high grade squamous intraepithelial lesion case, detail of the nuclear staining pattern.
Cytological diagnosis
Table 1 shows the distribution of the study group into cytological categories. For each category, the numbers of high risk HPV infections, HPV-16 infections, infections with HPV types of unknown malignant potential, and HPV negative samples are given.
Table 1.
Study population divided by cytological diagnosis according to Bethesda classification and the number of different types of HPV infection
| Cytology | All HR HPVs | HPV-16 | HPV unknown | HPV negative | Total (%) |
| WNL | 7 | 1 | 5 | 18 | 30 (20.4) |
| ASCUS | 28 | 9 | 9 | 12 | 49 (33.3) |
| LSIL | 42 | 8 | 2 | 3 | 47 (32.0) |
| HSIL | 20 | 12 | 0 | 0 | 20 (13.6) |
| AGUS | 0 | 0 | 0 | 1 | 1 (0.68) |
| Total | 97 | 30 | 16 | 33 | 147 (100) |
AGUS, atypical glandular cells of undetermined significance; ASCUS, atypical squamous cells of undetermined significance; HPV, human papillomavirus; HPV unknown, infections with HPV types of unknown malignant potential; HR, high risk; HSIL, high grade squamous intraepithelial lesion; LSIL, low grade squamous intraepithelial lesion; WNL, within normal limits.
The mean number of cells on a cytospin slide was 26 800 (95% confidence interval, 23 500 to 30 150).
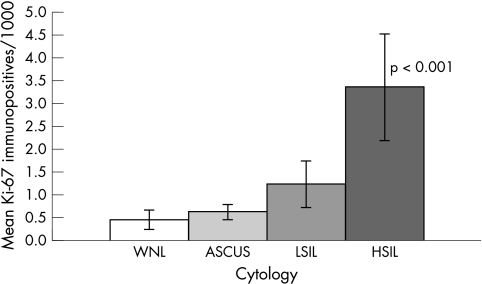
When comparing the number of Ki-67 immunoreactive cells/1000 cells between the different cytological groups, the HSIL group showed a significantly higher count (mean, 3.36; SEM, 0.86; p < 0.001) than did the other groups (fig 2). No significant differences in immunopositive counts were seen between the WNL (mean, 0.45; SEM, 0.19), ASCUS (mean, 0.62; SEM, 0.13), and LSIL (mean, 1.23; SEM, 0.48) groups.
Figure 2.
Ki-67 immunopositivity in cytology. Mean and SEM depicted for each cytological category, given as the number of immunopositive cells/1000 cells. The HSIL group differs significantly from the other groups. ASCUS, atypical squamous cells of undetermined significance; HSIL, high grade squamous intraepithelial lesion; LSIL, low grade squamous intraepithelial lesion; WNL, within normal limits.
When those abnormal samples containing HPV-16 were considered separately, the immunopositive count for HSIL (mean, 3.58; SEM, 1.08) was still double that for LSIL (mean, 1.69; SEM, 0.78), which in turn was double that of ASCUS (mean, 0.78; SEM, 0.32). There were no significant differences, possibly because of the low number of samples.
HPV typing
Table 2 shows the distribution of HPV types in our study group, in addition to the number of abnormal samples for each type. In cases with a single infection, types 16, 18, 33, 35, 39, 51, 52, 56, and 66 were identified. In cases with multiple infections, the pairs 16/33, 16/35, 31/35, 33/35, 35/66, 39/56 and 56/66 were found, in addition to two cases of triple infection, namely: 16/31/35 and 16/33/35.
Table 2.
Study population presented by human papillomavirus (HPV) type, with the number of abnormal samples for each type
| HPV type | n (%) | Total | Cytological abnormalities | |
| Single | 16 | 24 (16.3) | 23 | |
| 51 | 12 (8.2) | 11 | ||
| 56 | 8 (5.4) | 7 | ||
| 66 | 7 (4.8) | 7 | ||
| Other* | 17 (11.6) | 68 | 13 | |
| Multiple | Containing HPV-16 | 6 (4.1) | 6 | |
| 31/35 | 11 (7.5) | 11 | ||
| 56/66 | 7 (4.8) | 7 | ||
| Other† | 5 (3.4) | 29 | 5 | |
| Unknown | 16 | 11 | ||
| Negative | 34 | 16 | ||
| Total | 147 | 117 |
*Types 18, 33, 35, 39, 52, and 58; †double/triple combinations of oncogenic types, other than mentioned in this table.
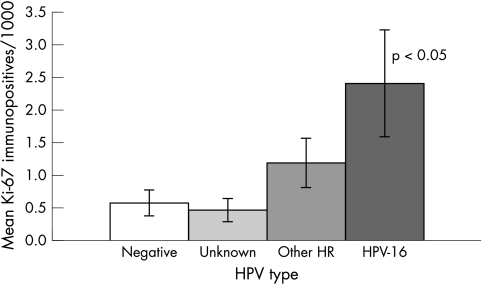
When comparing the number of Ki-67 immunoreactive cells/1000 cells between the different HPV types, HPV-16 showed a significantly higher count (mean, 2.40; SEM, 0.65) than did the other high risk HPV types (mean, 1.18; SD, 0.35) (fig 3). Within the group of other high risk types, no significant differences could be found (data not shown). For samples containing HPV types of unknown malignant potential, the Ki-67 count was lower and comparable to the count in HPV negative cases (mean, 0.46; SEM, 0.13; mean, 0.56; SEM, 0.17, respectively; p > 0.05).
Figure 3.
Ki-67 immunopositivity in human papillomavirus (HPV) typing. Mean and SEM depicted for HPV-16, other high risk (HR) HPV types, HPV types of unknown malign potential (unknown), and HPV negatives, given as the number of immunopositive cells/1000 cells. The HPV-16 group differs significantly from the other groups.
Diagnostic accuracy
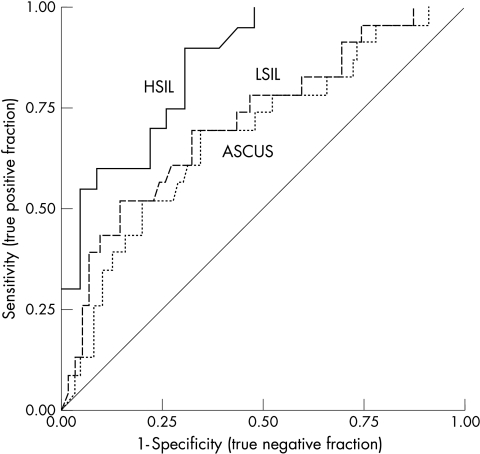
The overlap of the number of Ki-67 immunoreactive cells was relatively broad, both between the cytological groups and the groups of HPV types. To determine the ability of Ki-67 immunocytochemistry to discriminate between the various cytological categories, ROC plots were generated to illustrate the diagnostic accuracy of the test (fig 4). These plots show the specificity (true negative fraction) and sensitivity (true positive fraction) of the test for all possible thresholds. The accuracy of the test is given by the area under the curve (AUC). A value of 0.50 indicates that the test works randomly, indiscriminately identifying a case as either positive or negative, as shown by the diagonal straight line in the plot. A value of 1.00 indicates a perfect test, which always identifies all true positive cases correctly (100% sensitive), in addition to all true negative cases (100% specificity). Table 3 gives the AUCs and thresholds for 95% sensitivity and 95% specificity.
Figure 4.
Receiver operating characteristic curves for Ki-67 immunocytochemistry in three different diagnostic circumstances. ASCUS, a diagnosis of atypical squamous cells of undetermined significance or worse; LSIL, a diagnosis of low grade squamous intraepithelial lesion or worse; HSIL, a diagnosis of high grade squamous intraepithelial lesion.
Table 3.
Ki-67 immunocytochemistry test performance as indicated by ROC plot
| ASCUS or worse | LSIL or worse | HSIL | |
| 95% sensitivity | |||
| Specificity | 11% | 26% | 48% |
| Threshold | 0.07 | 0.08 | 0.15 |
| 95% specificity | |||
| Sensitivity | 19% | 22% | 42% |
| Threshold | 1.9 | 1.9 | 1.9 |
| AUC (95% CI) | 0.68 (0.56 to 0.81) | 0.72 (0.60 to 0.85) | 0.86 (0.75 to 0.97) |
At 95% sensitivity the corresponding specificity is given and vice versa. AUC (area under the curve) indicates diagnostic accuracy. Threshold values are the number of Ki-67 immunopositive cells/1000 cells.
ASCUS, atypical squamous cells of undetermined significance; CI, confidence interval; HSIL, high grade squamous intraepithelial lesion; LSIL, low grade squamous intraepithelial lesion; ROC, receiver operating characteristic.
DISCUSSION
Several large studies have investigated the use of Ki-67 as a prognostic factor in cervical carcinoma.10 Many found Ki-67 to have prognostic and diagnostic value in the classification of CIN or as a surrogate marker for HPV related neoplasia.8–22
There are only a few studies that have used cytology to test whether Ki-67 could function as an adjunctive or complementary tool. Similar to the results of our present study, they report Ki-67 immunocytochemistry to be of value as a complementary tool in cervical cytology, not only in the initial triage of minor abnormalities, but also in the follow up of abnormal Pap smears.23,24
The evolution of screening for cervical cancer has been considerable. Important efforts have been made to improve screening with the introduction and combination of new methods and techniques, such as liquid based cytology and HPV testing. In our present study, Ki-67 immunocytochemistry was applied to liquid based cytology, not only bypassing the disadvantages of the classic Pap smear, but also preventing bias caused by split samples because cytology, HPV DNA detection, HPV typing, and immunocytochemistry were all performed on the same sample.
In our series of 147 samples, a significantly higher number of Ki-67 immunopositive cells/1000 cells was found in the HSIL group compared with the other groups. This difference could not be explained by the fact that there were more HPV-16 infections in the HSIL group. Even if only those abnormal samples containing HPV-16 were compared, the immunopositive count for HSIL was still twice that of LSIL. This might indicate that there are other factors apart from HPV infection involved in the progression towards carcinoma in situ. These could be integration of the viral genome, viral load, factors that are not HPV related, or any combination of these.
Ki-67 immunocytochemistry might help to reduce the number of patients categorised as ASCUS, and help differentiate dysplasia from mimics such as atypical metaplasia or atrophy in diagnostically problematic cases. The diagnostic accuracy of the test for HSIL is good enough for Ki-67 immunocytochemistry to be used as an adjunct to LBC.
Hypothetically, the cytological diagnosis of ASCUS combined with a high count of Ki-67 immunopositive cells could be useful to identify those women who need follow up, because these would be cases where the cell cycle is disrupted. This hypothesis needs to be studied in a prospective situation.
The mean number of Ki-67 immunopositive cells/1000 cells for the HPV-16 group was significantly higher than that for the group of other high risk HPV types, highlighting the greater oncogenic potential of HPV-16. The count for HPV types of unknown malignant potential was lower than that for the group of high risk HPV types, presumably because many HPV types in the first group might be low risk types.
Immunocytochemistry could be used here as a surrogate marker for HPV-16 infection. Because the detection of deregulation of the cell cycle identifies those infections at risk of progression towards dysplasia and carcinoma, this test would be more specific than HPV testing.
It would be interesting to investigate whether, in cases where false negativity of the HPV DNA test is suspected, a high Ki-67 count could help identify women at risk. In our study, there were 21 cases of ASCUS where no high risk HPV type could be identified. Ki-67 immunocytochemistry would have identified 18 cases as positive at a threshold of 0.1 Ki-67 immunopositive cells/1000 cells and four at a threshold of 1.0 Ki-67 immunopositive cells/1000 cells. Similarly, there were five cases of LSIL without an infection with a high risk HPV type. Immunocytochemistry would have identified three cases as positive at a threshold of 0.1 Ki-67 immunopositive cells/1000 cells and one at a threshold of 1.0 Ki-67 immunopositive cells/1000 cells.
“It would be interesting to investigate whether, in cases where false negativity of the human papillomavirus DNA test is suspected, a high Ki-67 count could help identify women at risk”
The overlap in immunopositive counts in the different groups could partly be explained by the fact that normal proliferating cells from the basal layer of the epithelium will regularly be seen as positive cells.9 Such (para-)basal cells can be recognised on the slides used for immunocytochemistry. One option is not to count those cells, but this may turn out to be time consuming and subjective. In our present study, the diagnostic accuracy of Ki-67 immunocytochemistry was determined using ROC plots.32 This method has the advantage that it shows the full spectrum of sensitivities and specificities of a test for all possible thresholds. The accuracy is indicated by the AUC and is preferred to be > 0.70.33 In this study the AUCs of LSIL and HSIL were 0.72 and 0.86, respectively.
The identification of useful thresholds or cut off values is simplified by the ROC curves. The more difficult part is determining the relative cost and acceptability of errors made by the test. Another influencing factor is the prevalence of the disease, which determines the relative proportions of the two states of health that the test has to discriminate between.32 To illustrate the possibilities of Ki-67 immunocytochemistry, diagnostic indices are given for two possible thresholds (table 4).
Table 4.
Sensitivity, specificity, positive (PPV) and negative (NPV) predictive value for different screening techniques
| Conventional cytology | LBC | HPV testing by PCR | HPV testing by HCII | 0.2 ASCUS | 0.2 HSIL | 0.4 ASCUS | 0.4 HSIL | |
| Sensitivity | 52.038 to 68.035 | 61.337 to 87.035 | 88.237 to 91.036 | 73.338 to 10035 | 68.0 | 90.0 | 51.0 | 75.0 |
| Specificity | 52.838 to 95.035 | 82.437 to 93.035 | 78.837 | 62.338 to 87.335 | 61.0 | 61.0 | 73.0 | 73.0 |
| PPV | 23.535 to 60.38 | 15.735 | ND | 14.235 to 88.339 | 87.1 | 66.7 | 88.5 | 71.4 |
| NPV | 43.838 to 99.335 | 99.835 | ND | 62.338 to 10035 | 32.6 | 87.5 | 27.9 | 77.3 |
Data from the literature35,39 are given in percentages. Complementary diagnostic indices are given for 2 thresholds of Ki-67 immunocytochemistry in 2 different diagnostic circumstances. 0.2/0.4 ASCUS/HSIL represents a threshold of 0.2/0.4 Ki-67 immunopositive cells/1000 cells for the diagnosis of ASCUS/HSIL.
ASCUS, atypical squamous cells of undetermined significance; HCII, Hybrid Capture II (Digene); HPV, human papillomavirus; HSIL, high grade squamous intraepithelial lesion; LBC, liquid based cytology; ND, no data; PCR, polymerase chain reaction.
The efficiency of the conventional cervical smear has never been tested in a prospective blinded study.34 With the introduction of new techniques in screening, several studies have focused upon the sensitivity and specificity of screening techniques (table 4).35–39 Although the methods of comparison and definition of endpoints may diverge in these studies, they give a comprehensive insight into what is known about the efficiency of these screening techniques at this time. When the data from our study are compared with those in the literature, Ki-67 immunostaining yields good results, especially with regard to the diagnosis of HSIL.
To conclude, our present study shows that Ki-67 immunostaining can be performed on LBC samples for cervical cytology and yields significantly higher numbers of immunopositive cells in HSIL and in HPV-16 positive samples. Because of the overlap in numbers between groups, the use of an appropriate cut off value is advisable. Ki-67 immunocytochemistry is an easy and fast technique, which could perform well as part of a panel of screening procedures. Particularly in smaller or less specialised laboratories, Ki-67 immunocytochemistry could be an attractive alternative to PCR or Hybrid Capture II (Digene, Gaithersburg, Maryland, USA), which require more expensive and specialised equipment. It would be relatively simple to automate the staining procedure and in time it might be possible to automate the counting of immunopositive cells. It could be used as an adjunct to LBC to identify HSIL and as a surrogate marker of HPV infection in situations where HPV detection is not available or appropriate.
Take home messages.
Ki-67 immunocytochemistry can be applied to liquid based cytology, and yields significantly higher numbers of immunopositive cells in high grade squamous intraepithelial lesions (HSIL) and in human papillomavirus type 16 (HPV-16) positive samples
Because of the overlap in numbers between groups, we recommend the use of an appropriate cut off value
The accuracy and diagnostic indices of the test are good when compared with those of other techniques
Ki-67 immunocytochemistry could be used as an adjunct to liquid based cytology to identify high grade HSIL, and as a surrogate marker of HPV-16 infection
Abbreviations
ASCUS, atypical squamous cells of undetermined significance
AUC, area under the curve
CIN, cervical intraepithelial neoplasia
HPV, human papillomavirus
HSIL, high grade squamous intraepithelial lesion
LBC, liquid based cytology
LSIL, low grade squamous intraepithelial lesion
Pap, Papanicolaou
PCR, polymerase chain reaction
ROC, receiver operating characteristic
SEM, standard error of the mean
WNL, within normal limits
REFERENCES
- 1.Ho GY, Burk RD, Klein S, et al. Persistent genital human papillomavirus infection as a risk factor for persistent cervical dysplasia. J Natl Cancer Inst 1995;87:1365–71. [DOI] [PubMed] [Google Scholar]
- 2.Walboomers JM, Jacobs MV, Manos MM, et al. Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. J Pathol 1999;189:12–19. [DOI] [PubMed] [Google Scholar]
- 3.Bosch FX, Lorincz A, Munoz N, et al. The causal relation between human papillomavirus and cervical cancer. J Clin Pathol 2002;55:244–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bosch FX, Manos MM, Munoz N, et al. Prevalence of human papillomavirus in cervical cancer: a worldwide perspective. International biological study on cervical cancer (IBSCC) study group. J Natl Cancer Inst 1995;87:796–802. [DOI] [PubMed] [Google Scholar]
- 5.Ho GY, Bierman R, Beardsley L, et al. Natural history of cervicovaginal papillomavirus infection in young women. N Engl J Med 1998;338:423–8. [DOI] [PubMed] [Google Scholar]
- 6.Schiffman MH, Bauer HM, Hoover RN, et al. Epidemiologic evidence showing that human papillomavirus infection causes most cervical intraepithelial neoplasia. J Natl Cancer Inst 1993;85:958–64. [DOI] [PubMed] [Google Scholar]
- 7.Munoz N, Bosch FX, de Sanjose S, et al. Epidemiologic classification of human papillomavirus types associated with cervical cancer. N Engl J Med 2003;348:518–27. [DOI] [PubMed] [Google Scholar]
- 8.Keating JT, Cviko A, Riethdorf S, et al. Ki-67, cyclin E, and p16INK4 are complementary surrogate biomarkers for human papillomavirus-related cervical neoplasia. Am J Surg Pathol 2001;25:884–91. [DOI] [PubMed] [Google Scholar]
- 9.Bulten J, van der Laak JA, Gemmink JH, et al. MIB1, a promising marker for the classification of cervical intraepithelial neoplasia. J Pathol 1996;178:268–73. [DOI] [PubMed] [Google Scholar]
- 10.Brown DC, Gatter KC. Ki-67 protein: the immaculate deception? Histopathology 2002;40:2–11. [DOI] [PubMed] [Google Scholar]
- 11.Garzetti GG, Ciavatinni A, Lucarni G, et al. MIB1 immunostaining in stage I squamous cervical carcinoma: relationship with natural killer cell activity. Gynecol Oncol 1995;58:28–33. [DOI] [PubMed] [Google Scholar]
- 12.Nakano T, Oka K. Differential values of Ki-67 index and mitotic index of proliferating cell population. An assessment of cell cycle and prognosis in radiation therapy for cervical cancer. Cancer 1993;72:2401–08. [DOI] [PubMed] [Google Scholar]
- 13.Nanjyo T, Watanabe Y, Ikeda M. [The correlation between the therapeutic effects and tumor growth fraction in cervical cancer and ovarian cancer]. Nippon Sanka Fujinka Gakkai Zasshi 1994;46:27–34. [PubMed] [Google Scholar]
- 14.Nakano T, Oka K, Ishakawa A, et al. Correlation of cervical carcinoma c-erbB-2 oncogene with cell proliferation parameters in patients treated with radiation therapy for cervical carcinoma. Cancer 1997;79:513–20. [PubMed] [Google Scholar]
- 15.Ho DM, Hsu CY, Chiang H. MIB-1 labeling index as a prognostic indicator for survival in patients with FIGO stage IB squamous cell carcinoma of the cervix. Gynecol Oncol 2000;76:97–102. [DOI] [PubMed] [Google Scholar]
- 16.Avall-Lundqvist EH, Silfversward C, Aspenblad U, et al. The impact of tumour angiogenesis, p53 overexpression and proliferative activity (MIB-1) on survival in squamous cervical carcinoma. Eur J Cancer 1997;33:1799–1804. [DOI] [PubMed] [Google Scholar]
- 17.Dellas A, Moch H, Schultheiss E, et al. Angiogenesis in cervical neoplasia: microvessel quantitation in precancerous lesions and invasive carcinomas with clinicopathological correlation. Gynecol Oncol 1997;67:27–33. [DOI] [PubMed] [Google Scholar]
- 18.Dellas A, Schultheiss E, Almendral AC, et al. Altered expression of mdm-2 and its association with p53 protein status, tumor-cell-proliferation rate and prognosis in cervical neoplasia. Int J Cancer 1997;74:421–5. [DOI] [PubMed] [Google Scholar]
- 19.Oka K, Arai T. MIB1 growth fraction is not related to prognosis in cervical squamous cell carcinoma treated with radiotherapy. Int J Gynecol Pathol 1996;15:23–7. [DOI] [PubMed] [Google Scholar]
- 20.Levine EL, Renehan A, Gossiel R, et al. Apoptosis, intrinsic radiosensitivity and prediction of radiotherapy response in cervical carcinoma. Radiother Oncol 1995;37:1–9. [DOI] [PubMed] [Google Scholar]
- 21.Cole DJ, Brown DC, Crossley E, et al. Carcinoma of the cervix uteri: an assessment of the relationship of tumour proliferation to prognosis. Br J Cancer 1992;65:783–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bulten J, de Wilde PC, Schijf C, et al. Decreased expression of Ki-67 in atrophic cervical epithelium of post-menopausal women. J Pathol 2000;190:545–53. [DOI] [PubMed] [Google Scholar]
- 23.Zeng Z, Del PG, Cohen JM, et al. MIB-1 expression in cervical Papanicolaou tests correlates with dysplasia in subsequent cervical biopsies. Appl Immunohistochem Mol Morphol 2002;10:15–9. [DOI] [PubMed] [Google Scholar]
- 24.Dunton CJ, Van Hoeven KH, Kovatich AJ, et al. Ki-67 antigen staining as an adjunct to identifying cervical intraepithelial neoplasia. Gynecol Oncol 1997;64:451–5. [DOI] [PubMed] [Google Scholar]
- 25.Vassilakos P, Petignat P, Boulvain M, et al. Primary screening for cervical cancer precursors by the combined use of liquid-based cytology, computer-assisted cytology and HPV DNA testing. Br J Cancer 2002;86:382–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sahebali S, Depuydt CE, Segers K, et al. Cervical cytological screening and HPV DNA testing in Flanders. Acta Clin Belg [In press.] [DOI] [PubMed]
- 27.Depuydt CE, Vereecken AJ, Salembier GM, et al. Thin-layer liquid-based cervical cytology and PCR for detecting and typing human papillomavirus DNA in Flemish women. Br J Cancer 2003;88:560–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Vassilakos P, Saurel J, Rondez R. Direct-to-vial use of the AutoCyte PREP liquid-based preparation for cervical-vaginal specimens in three European laboratories. Acta Cytol 1999;43:65–8. [DOI] [PubMed] [Google Scholar]
- 29.Kurman RJ, Solomon D. The Bethesda system for reporting cervical/vaginal cytologic diagnoses. New York: Springer Verlag, 1994.
- 30.Bauer HM, Ting Y, Greer CE, et al. Genital human papillomavirus infection in female university students as determined by a PCR-based method. JAMA 1991;265:472–7. [PubMed] [Google Scholar]
- 31.Solomon D, Davey D, Kurman R, et al. The 2001 Bethesda system: terminology for reporting results of cervical cytology. JAMA 2002;287:2114–19. [DOI] [PubMed] [Google Scholar]
- 32.Zweig MH, Campbell G. Receiver-operating characteristic (ROC) plots: a fundamental evaluation tool in clinical medicine. Clin Chem 1993;39:561–77. [PubMed] [Google Scholar]
- 33.Hanley JA, McNeill BJ. The meaning and use of the area under a receiver operating characteristic (ROC) curve. Radiology 1982;143:29–36. [DOI] [PubMed] [Google Scholar]
- 34.Koss LG. The Papanicolaou test for cervical cancer detection. A triumph and a tragedy. JAMA 1989;261:737–43. [PubMed] [Google Scholar]
- 35.Clavel C, Masure M, Bory JP, et al. Human papillomavirus testing in primary screening for the detection of high-grade cervical lesions: a study of 7932 women. Br J Cancer 2001;84:1616–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Karlsen F, Kalantari M, Jenkins A, et al. Use of multiple PCR primer sets for optimal detection of human papillomavirus. J Clin Microbiol 1996;34:2095–100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kulasingam SL, Hughes JP, Kiviat NB, et al. Evaluation of human papillomavirus testing in primary screening for cervical abnormalities: comparison of sensitivity, specificity and frequency of referral. JAMA 2002;288:1749–57. [DOI] [PubMed] [Google Scholar]
- 38.Ratnam S, Franco EL, Ferenczy A. Human papillomavirus testing for primary screening of cervical cancer precursors. Cancer Epidemiol Biomarkers Prev 2000;9:945–51. [PubMed] [Google Scholar]
- 39.Hong IS, Marshalleck J, Williams RH, et al. Comparative analysis of a liquid-based Pap test and concurrent HPV DNA assay of residual samples. A study of 608 cases. Acta Cytol 2002;46:828–34. [DOI] [PubMed] [Google Scholar]