Abstract
Rats transgenic (TG) for the human major histocompatibility complex (MHC) class I HLA-B27 and β2-microglobulin genes develop chronic colitis under specific pathogen-free (SPF) but not sterile (germ-free, GF) conditions. We investigated the role of antigen-presenting molecules involved in generating immune responses by CD4+ mesenteric lymph node (MLN) cells from colitic HLA-B27 TG rats to commensal enteric micro-organisms. All TG MLN cells expressed HLA-B27. A higher level of MHC class II was expressed on cells from TG rats, both SPF and GF, compared to non-TG littermates. In contrast, rat MHC class I expression was lower on TG than non-TG cells. Both TG and non-TG antigen presenting cells (APC) pulsed with caecal bacterial antigens induced a marked interferon-γ (IFN-γ) response in TG CD4+ T lymphocytes but failed to stimulate non-TG cells. Blocking MHC class II on both TG and non-TG APC dramatically inhibited their ability to induce TG CD4+ T cells to produce IFN-γ. Blocking HLA-B27 on TG APC similarly inhibited IFN-γ responses. When the antibodies against MHC class II and HLA-B27 were combined, no APC-dependent IFN-γ response was detected. These data implicate both native rat MHC class II and TG HLA-B27 in CD4+ MLN T-cell IFN-γ responses to commensal enteric microflora in this colitis model.
Keywords: CD4+ T-cell responses, HLA-B27, MHC class II, enteric bacterial antigens, transgenic rats
Introduction
Ulcerative colitis and Crohn's disease are two chronic diseases characterized by relapsing inflammation of the distal intestinal tract. Their aetiologies remain unclear but an unrestrained cell-mediated immune response to non-pathogenic commensal luminal bacteria in a genetically susceptible host has been proposed.1 Substantial data from clinical observations and animal experiments implicate activated CD4+ T lymphocytes, which accumulate in inflamed tissues.1,2 Evidence incriminates a polarized T helper 1 immune response with exaggerated interferon (IFN)-γ, interleukin-12 (IL-12), and tumour necrosis factor (TNF) production. In several murine models, intestinal inflammation can be prevented by neutralization of endogenous IL-123,4 and IFN-γ5 and dramatically reduced by administration of anti-TNF antibody.5,6 Likewise, anti-TNF and anti-IL-12 antibodies induce rapid clinical responses in Crohn's disease patients.7,8
Transgenic (TG) rats expressing the human major histocompatibility complex (MHC) class I gene, HLA-B27 and its associated light chain, human β2-microglobulin spontaneously develop a progressive, multisystemic disease including gastritis, colitis, and arthritis when maintained in specific pathogen-free (SPF) conditions.9 The essential role of T cells in disease pathogenesis has been demonstrated in cell transfer experiments using athymic HLA-B27 TG nude rats which, unlike euthymic TG rats, fail to develop intestinal inflammation unless they are reconstituted with T cells.10
Antigen-presenting cells (APC) are critical for induction of inflammatory disease. Inflammation can be transferred by engraftment of bone marrow from HLA-B27 TG nu/nu donors into non-TG recipients.10,11 Another study12 showed that freshly isolated colonic lamina propria dendritic cells from CD4+ T-cell reconstituted immunodeficient mice with severe colitis but not from healthy, syngeneic controls could induce a high IFN-γ response in CD4+ cells. It is likely therefore, that chronic intestinal inflammation is triggered by APC interacting with T cells.
Mucosal microflora are also crucial to the initiation and perpetuation of spontaneous colitis in animal models.1,2 Genetically susceptible rodents raised in germ-free (GF) conditions do not develop colitis 13–16 or have markedly attenuated experimental intestinal inflammation.17 In contrast, colonization of these susceptible GF animals with commensal SPF bacteria rapidly induces gut inflammation. Moreover, broad-spectrum antibiotics can prevent as well as reverse experimental colitis.18–21
Although these observations suggest that a genetic predisposition, commensal intestinal micro-organisms, activated APC and CD4+ T lymphocytes are key factors for the development of colitis, neither the triggering antigens nor the mechanisms by which bacterial products activate immune cells have been identified. The aim of the present study therefore was to evaluate activation of HLA-B27 TG CD4+ T cells by physiologically relevant components of intestinal micro-organisms. Our results show that IFN-γ is induced during in vitro coculture of caecal bacterial lysate-pulsed APC with CD4+ T cells from HLA-B27 TG rats with colitis. CD4+ cells from non-TG littermates do not produce IFN-γ in parallel cocultures. Moreover, we demonstrate a role for both HLA-B27 and endogenous MHC class II molecules on APC in the induction of the IFN-γ response.
Materials and methods
Animals
HLA-B27 TG rats of the high transgene copy number line designated 33-3 and non-TG littermates on an inbred F344 background, originally obtained from Dr Joel D. Taurog (South-western Medical School, Dallas, TX), were maintained in a SPF environment at the University of North Carolina at Chapel Hill or under GF conditions in the Gnotobiotic Animal Core of the Center for Gastrointestinal Biology and Disease at the College of Veterinary Medicine, North Carolina State University. Offspring were genotyped for the HLA-B27 transgene by polymerase chain reaction analysis of DNA from tail clippings; the presence of HLA-B27 on TG MLN cells was further confirmed by flow cytometry.
Rats of both sexes, 4–6 months old, when the SPF TG rats uniformly developed clinically evident colitis, were used throughout the studies. The investigation was approved by the Institutional Animal Care and Use Committees of the University of North Carolina at Chapel Hill, and North Carolina State University.
Preparation of caecal bacterial lysate
Caecal bacterial lysate was prepared according to the method described by Cong et al.22 Briefly, caecal contents from three to four SPF non-TG rats were vortexed, treated with 20 µg/ml DNA-ase and 3·7 mg/ml MgCl2, and disrupted using 0·1 mm glass beads in a mini-bead beater for 3 min (Biospec Products, Bartlesville, OK). The supernatant was collected after centrifugation and filter-sterilized through a 0·45 µm filter. The protein concentration was determined using a standard assay (Biorad Laboratories, Hercules, CA).
Preparation of single-cell suspensions and enriched lymphoid cell subpopulations
MLNs were collected from HLA-B27 TG and non-TG rats, and single cell suspensions were prepared by gentle mechanical teasing. Cell were washed, counted and resuspended in RPMI for fractionation or flow cytometry.
APC preparation
APC were prepared from MLNs by rabbit complement-mediated lysis of T cells bound by IgM anti-rat CD3 monoclonal antibody (Clone 1F4, PharMingen, San Diego, CA). The resulting population from non-TG rats contained over 96% surface immunoglobulin-expressing cells, a characteristic of B cells, and less than 4% CD4+ or CD8+ cells. In the case of TG rats, more than 92% of the remaining cells expressed surface immunoglobulin and less than 7–8% positive for CD4 or CD8.
CD4+ T lymphocyte purification
CD4+ T cells were enriched from MLN cells by negative selection using a magnetic-activated cell sorting system (MACS, Miltenyi, Auburn, CA) and magnetic beads conjugated with antibodies directed against rat CD45RA (Clone OX-33) and CD8α (Clone G38) to deplete B cells and CD8+ cells, respectively. The purity of the selected CD4+ T lymphocyte population on average was 96%.
Flow cytometry
Unseparated MLN cells and APC or CD4+ T lymphocytes (2–5 × 105/50 µl) were analysed on a flow cytometer (FACScan, Becton Dickinson, San Jose, CA) using the CELLQuest™ software program.
The following fluorochrome labelled or unlabelled reagents were used. We used cell culture supernatants from the murine hybridomas, designated ME-1 (ATCC, Rockville, MD) or OX-18 (Hybridoma Laboratory, North Carolina State University), followed by fluoroscein isothiocyanate (FITC)-labelled goat anti-mouse immunoglobulin G (IgG)(γ) antibody (Southern Biotechnology, Birmingham, AL) to identify HLA-B27 and rat MHC class I expressing cells, respectively. MHC class II expressing cells were detected by FITC-conjugated anti-MHC class II monoclonal antibody (Clone OX-6, PharMingen). Surface immunoglobulin expressing B cells were identified by FITC labelled goat anti-rat IgG (H + L) antibody (Kirkegaard & Perry Laboratories, Gaithersburg, MD). CD4+ and CD8+ cells were identified using phycoerythrin (PE)–anti-CD4 (Clone W3/25) and FITC–anti-CD8 (Clone OX-8) monoclonal antibodies (Caltag, Burlingame, CA), respectively. For analyses of MHC class I expression on T and B cells, FITC-labelled anti-MHC class I monoclonal antibody (Clone OX-18, PharMingen) was used together with PE–anti-CD3 (Clone G4.18) or PE–anti-CD45RA (Clone OX-33) monoclonal antibodies (PharMingen). To compare CD86 expression on non-TG and TG MLN cells, monoclonal antibody against CD86 (B7-2; Clone 24F, PharMingen) was used.
Co-cultures of APC and CD4+ T lymphocytes
APC (3 × 107 cells/8 ml) were pulsed with caecal bacterial lysate or with keyhole limpet haemocyanin (KLH; Pierce, Rockford, IL), an unrelated antigen, at 50 or 100 µg/ml (amount equivalent to optimal concentration determined for each batch of bacterial lysate) in complete culture medium (RPMI-1640 supplemented with 5% heat-inactivated fetal calf serum, 2 m l-glutamine, 1 m sodium pyruvate, 50 µm 2-mecaptoethanol, and 50 µg/ml gentamicin).
The APC were collected after overnight culture and washed twice to remove soluble bacterial antigens and other components. They were then either analysed by flow cytometry or cultured at 3 × 105 cells per well in a final volume of 0·2 ml in flat-bottom 96-well plates, together with 2 × 105 CD4+ cells, in a humidified incubator at 37°, 7% CO2. The chosen cell concentrations were determined by previous studies to be optimal for IFN-γ responses. Triplicate supernatants were harvested after 3 or 6 days. Supernatants collected on day 6 contained equivalent or only slightly higher amounts of IFN-γ compared to supernatants collected on day 3. Therefore, analyses of IFN-γ detected on day 3 after culture initiation are shown.
In selected experiments, APC were treated with one or a combination of the following monoclonal antibodies to functionally block transgenic HLA-B27, rat MHC class I, rat MHC class II RT1. B or MHC class II RT1. D (ME-1, OX-18, OX-6, and OX-17, respectively). Supernatants of the murine hybridoma cell cultures were concentrated using an Amicon YM30 membrane to obtain the following stock concentrations, ME-1, 0·7 mg/ml; OX-18, 4·1 mg/ml; OX-6, 2·8 mg/ml; and OX-17, 60·5 mg/ml. The concentration of isotype control, mouse IgG1 (Clone 107.3, PharMingen) was adjusted to be equivalent to the highest concentration of each monoclonal antibody used in blocking experiments. Antibodies were added to APC at different concentrations as indicated for 30 min. Thereafter, APC were either washed to remove the unbound antibodies or directly added to cocultures with CD4+ T lymphocytes. No differences were observed in the amounts of IFN-γ produced whether or not the APC were washed to remove excess antibodies. Results are shown for experiments in which antibodies remained during the entire 3-day cocultures.
Enzyme linked immunosorbent assay (ELISA)
IFN-γ was measured in triplicate cell culture supernatants by ELISA using unlabelled capture antibody (rabbit polyclonal anti-IFN-γ) and biotin-labelled detection antibody (monoclonal Clone DB-1; BioSource International, Camarillo, CA), followed by horseradish peroxidase-labelled streptavidin (PharMingen). The concentration was determined by comparison to a standard curve generated using recombinant rat IFN-γ (PharMingen).
Statistical analysis
Results from flow cytometric analyses and ELISA are expressed as mean ± SEM. The probability of zero difference between the groups was judged by two-tailed Student's t-test. A P-value less than 0·05 was considered to be statistically significant.
Results
Compromised function of SPF TG APC
TG rats kept in SPF conditions showed severe and consistent colitis when killed at 4–6 months of age (histology scores >3 on a scale of 0–4), consistent with previous observations.13 In contrast, neither non-TG littermates exposed to the same microflora nor GF rats of either genotype developed disease (histologic scores <0·5). The MLNs of SPF TG rats were markedly increased in size and contained 3–10-fold more cells with a significantly higher percentage and total number of CD4+ T cells compared to MLNs from non-TG littermates (data not shown), consistent with our previous observations.23
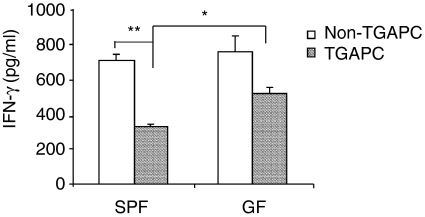
To address the relative roles of HLA-B27 expression on APC and T cells in mucosal immune responses as well as the influence of in vivo activation of APC, we studied CD4+ T lymphocytes, the main source of IFN-γ23 from TG and non-TG rat MLN cocultured with caecal bacterial lysate-pulsed APC. Caecal bacterial lysate-pulsed APC from four separate sources (SPF non-TG MLN, SPF TG MLN, GF non-TG MLN, GF TG MLN) induced a significant IFN-γ response in CD4+ lymphocytes from SPF TG rats with colitis (Fig. 1). The IFN-γ level was low or undetectable in cultures of CD4+ cells stimulated with KLH-pulsed APC (<76 pg/ml of IFN-γ was measured), indicating that secretion of this cytokine depends on in vitro activation of APC by bacterial components and/or presentation of bacterial antigens by APC. CD4+ cells isolated from MLN of non-TG rats did not produce IFN-γ above baseline levels in cocultures with each of the caecal bacterial lysate-pulsed APC populations studied (<103 pg/ml of IFN-γ was measured). This and our previous observation that MLN cells from GF TG rats did not produce IFN-γ after in vitro stimulation with caecal bacterial lysate23 indicate that prior in vivo activation of CD4+ cells is necessary in order to induce in vitro responses.
Figure 1.
IFN-γ production by SPF HLA-B27 TG MLN CD4+ T lymphocytes cocultured with caecal bacterial lysate-pulsed SPF or GF APC preparations. Co-culture supernatants were harvested on day 3 and assayed by ELISA. Values represent mean ± SEM of pg/ml of IFN-γ in triplicate culture supernatants. Results shown are representative of three independent experiments. Statistically significant differences between SPF TG and non-TG APC or between SPF and GF TG APC in inducing IFN-γ production by SPF TG CD4+ cells are denoted by asterisks (*P < 0·05, **P < 0·01).
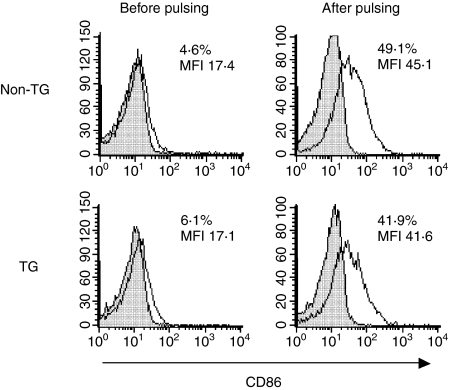
We consistently observed that caecal bacterial lysate-pulsed SPF TG APC stimulated TG CD4+ T cells to produce significantly less IFN-γ than did non-TG APC (Fig. 1). In addition, lysate-pulsed SPF TG APC induced less IFN-γ than did GF TG APC. To determine whether or not costimulatory molecules are normally expressed on SPF TG APC, we evaluated cell surface CD86 by flow cytometry. As shown in Fig. 2 and Table 1, while a slightly higher proportion of SPF TG than non-TG APC expressed CD86 prior to pulsing or after overnight pulsing with KLH (used as an unrelated antigen control), we observed the opposite effect after pulsing APC with caecal bacterial lysate. The bacterial lysate induced increased expression of CD86 on both non-TG and TG APC. However, non-TG APC contained more CD86-expressing cells with higher mean fluorescence intensity (MFI) compared to cells from TG littermates. Thus, up-regulation of CD86 by bacterial components appeared to be compromised in SPF TG APC.
Figure 2.
Surface expression of CD86 on T-cell depleted MLN cells from SPF non-TG (upper panels) and TG (lower panels) rats before (left column) and after (right column) overnight pulsing with caecal bacterial lysate. Isotype control staining with FITC labelled mouse IgG1 is shown as filled histograms. Values indicate percentage of positive cells and MFI. Results are representative of three independent experiments.
Table 1.
CD86 and MHC class II expression on T cell depleted mesenteric lymph node cells
| Non-TG | TG | |||
|---|---|---|---|---|
| (%) | MFI | (%) | MFI | |
| CD86 | ||||
| Before pulsing | 6·2 ± 0·3 | 20·8 ± 2·2 | 7·3 ± 0·7 | 24·8 ± 3·4 |
| KLH pulsing | 11·9 ± 0·3 | 20·5 ± 1·9 | 14·7 ± 0·8** | 25·0 ± 2·0 |
| CBL pulsing | 51·3 ± 0·9 | 48·0 ± 2·5 | 41·7 ± 1·3*** | 41·3 ± 2·3 |
| MHC class II | ||||
| Before pulsing | 79·0 ± 1·2 | 1183·9 ± 38·9 | 84·0 ± 1·0** | 1698·7 ± 38·2*** |
| KLH pulsing | 91·6 ± 0·7 | 1156·4 ± 55·0 | 93·0 ± 0·8 | 1968·5 ± 33·4*** |
| CBL pulsing | 94·3 ± 0·6 | 3183·8 ± 39·2 | 94·2 ± 0·8 | 2963·3 ± 42·7*** |
Surface expression of CD86 and MHC class II (RT1.B) on T-cell depleted mesenteric lymph node cells from SPF non-transgenic (non-TG) and TG rats before and after overnight pulsing with caecal bacterial lysate (CBL) or unrelated antigen KLH was analyzed by flow cytometry. Results are summarized from four individual experiments including eight non-TG and seven TG rats. The percentage (%) of positive cells and mean fluorescence intensity (MFI) are shown as mean ± SEM.
P < 0·01;
P < 0·001: versus non-TG rats.
When caecal bacterial lysate-pulsed non-TG APC from GF and SPF rats were compared for their ability to stimulate SPF TG CD4+ cells, a similar amount of IFN-γ was detected (Fig. 1). Therefore, prior in vivo activation of APC by exposure to enteric micro-organisms or secondary to chronic inflammation was not required for optimal in vitro stimulation of CD4+ T cells.
It should be noted that IFN-γ was detected at very low levels in supernatants of pulsed APC cultured alone (<124 pg/ml of IFN-γ was measured), even though the cultures contained a small number of residual T cells (less than 4% and 8% for non-TG and TG APC, respectively).
Evaluation of MLN cells from SPF and GF TG and non-TG rats for MHC molecule expression
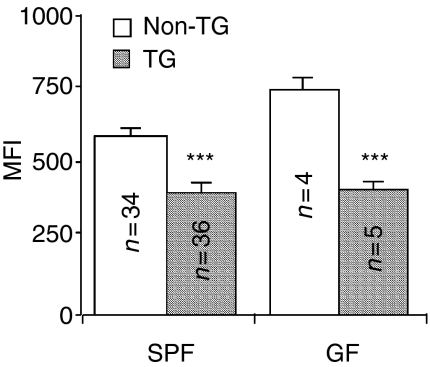
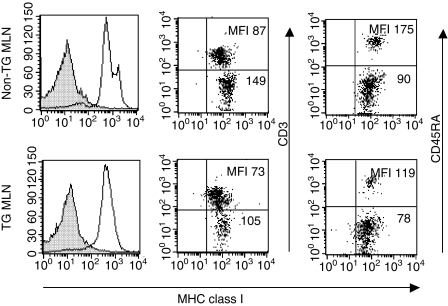
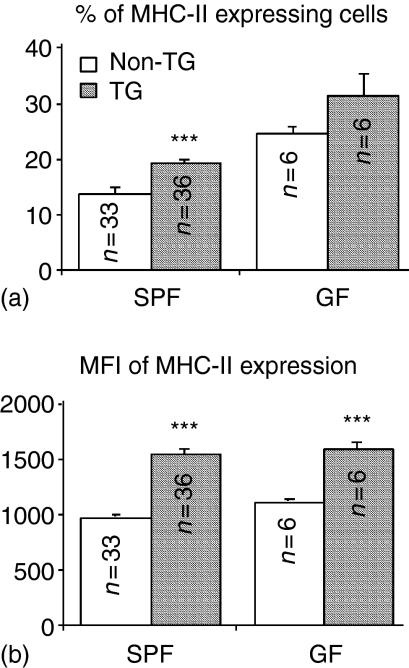
We next evaluated the expression of rat MHC class I, MHC class II and transgenic HLA-B27 on TG and non-TG MLN cells from SPF and GF rats. MLN cells from TG rats expressed lower levels of endogenous rat MHC class I compared to cells from non-TG rats (Fig. 3), as had been previously reported.9 We extended these findings by demonstrating that non-activated MLN cells of TG rats housed under GF conditions also displayed lower constitutive levels of rat MHC class I. In addition, we found that rat MHC class I expression was lower on both T and B cells of TG compared to non-TG rats, from both SPF and GF colonies (Fig. 4 and data not shown). T cells expressed less rat MHC class I than did B cells and the difference between T- and B-cell rat MHC class I expression was greater in non-TG compared to TG rats (Fig. 4). Therefore two separate peaks, one representing B cells with higher levels of MHC class I expression, another showing T cells with lower amounts of MHC class I, were consistently noted in the flow cytometry histograms for non-TG but not for TG MLN cells (Fig. 4). T-cell depleted non-TG and TG MLN cells both showed single peak of MHC class I expression (data not shown). In contrast, freshly isolated MLN from TG rats, especially SPF TG rats, contained a higher proportion of cells that expressed MHC class II than did MLN from non-TG rats (Fig. 5 and Table 1). In addition, the expression of MHC class II per cell (mean fluorescence intensity, MFI) was greater on TG compared to non-TG MLN cells from both SPF and GF rats. Interestingly, as shown in Table 1, we observed a greater increase in MHC class II expression on non-TG APC following caecal bacterial lysate pulsing (2·8-fold higher MFI compared to KLH-pulsed non-TG APC) than after caecal bacterial lysate pulsing of TG APC (1·5-fold higher MFI compared to KLH-pulsed TG APC), indicating a relative deficiency of TG APC responses to in vitro restimulation despite their higher baseline MHC class II expression. All MLN cells from TG rats expressed HLA-B27 while the non-TG cells did not (data not shown).
Figure 3.
Analyses of surface expression of rat MHC class I on freshly isolated SPF or GF MLN cells. Unseparated MLN cells were incubated with unlabelled hybridoma cell culture supernatant OX-18. Binding of OX-18 was detected by FITC-labelled goat anti-mouse IgG(γ) antibody. MFI is shown as mean ± SEM. Statistically significant differences between TG and non-TG cells are denoted by asterisks (***P < 0·001). n = number of rats/group.
Figure 4.
Cell surface expression of rat MHC class I. Histograms showing MHC class I expression on unseparated MLN cells from SPF non-TG and TG rats, detected by indirect fluorescence using unlabelled OX-18 followed by FITC labelled goat anti-mouse IgG(γ) (black line) compared to control staining with the FITC-labelled secondary antibody alone (filled) are shown in the left column. Dot plots showing MHC class I expression on CD3-positive and CD3-negative MLN cells are in the middle column. Dot plots showing MHC class I expression on CD45RA-positive and CD45RA-negative MLN cells are in the right column. For identification of MHC class I expression on T and B cells, direct fluorescence of FITC-labelled anti-MHC class I monoclonal antibody (OX-18) and PE-anti-CD3 or PE-anti-CD45RA monoclonal antibody was evaluated. Histograms showing all MLN cells are representative of over 30 individual TG and non-TG rats. Dot plots showing two-colour analysis for MHC class I and CD3 or CD45RA are representative of three individual TG and non-TG rats.
Figure 5.
Analyses of surface expression of MHC class II (RT1.B detected by OX-6 monoclonal antibody) on freshly isolated MLN cells. The percentage of positive cells and MFI are presented as mean ± SEM. Statistically significant differences between TG and non-TG rats are denoted by asterisks (***P < 0·001). n = number of rats/group.
Blocking MHC class II but not rat MHC class I on APC significantly reduced IFN-γ production by TG CD4+ T lymphocytes
The role of the MHC class I glycoprotein HLA-B27 in the development of inflammatory disease in either humans or HLA-B27 TG rats is not well understood. In addition, while the observations that SPF but not GF HLA-B27 TG rats develop colitis13,15 and that SPF T-cell deficient TG rats fail to develop disease10 implicate intestinal micro-organisms and suggest that T cells from TG rats respond to enteric bacterial antigens, the role of bacterial antigens and of host antigen-presenting molecules in T-cell responses has not been evaluated. To determine if the pronounced IFN-γ response of activated TG CD4+ T lymphocytes to caecal bacterial lysate-pulsed APC depends on interaction between MHC and T-cell receptor (TCR) molecules and to examine the relative contribution of various MHC molecules to activate CD4+ T cells, we designed experiments to functionally block rat MHC class I, MHC class II and TG HLA-B27 on APC from SPF TG and non-TG rats.
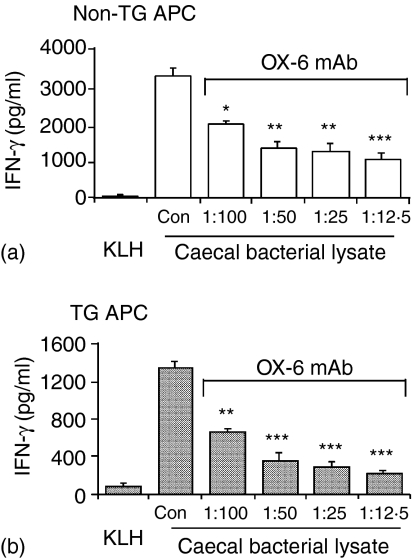
For these experiments, serial dilutions of antibodies directed against rat MHC class I (OX-18), MHC class II locus RT1.B (OX-6), MHC class II locus RT1.D (OX-17), and transgenic HLA-B27 (ME-1), were added to caecal bacterial lysate-pulsed APC prior to the addition of TG CD4+ T lymphocytes. Treatment of both non-TG and TG APC with the anti-MHC class II RT1.B antibody significantly inhibited, in a dose-dependent manner, cocultured TG CD4+ T-cell response (50–85% reduction in IFN-γ production, Fig. 6). Non-TG and TG APC-dependent IFN-γ responses were also inhibited, but less effectively, by the anti-MHC class II RT1.D antibody (39–59% suppression of IFN-γ responses, data not shown).
Figure 6.
Blockade of MHC class II significantly inhibited IFN-γ production induced by caecal bacterial lysate-pulsed APC in cocultures of TG CD4+ T lymphocytes plus non-TG APC (a) or TG APC (b). Anti-MHC class II RT1.B antibody (OX-6 monoclonal antibody) at the dilutions indicated, or the isotype control (Con), purified mouse IgG1 at the amount equivalent to the highest concentration of OX-6 mAb, was added to the pulsed SPF APC for 30 min before the start of cocultures with SPF TG CD4+ T lymphocytes. Supernatants were harvested at day 3 and assayed by ELISA. Values represent mean ± SEM of pg/ml of IFN-γ in triplicate culture supernatants. TG CD4+ cells cocultured with an unrelated antigen KLH-pulsed APC served as a control. Results shown are representative of six independent experiments. Statistically significant differences between the amounts of IFN-γ produced in cocultures containing APC treated with anti-MHC class II antibody and in cocultures containing APC treated with the isotype control are denoted by asterisks (*P < 0·05, **P < 0·01, ***P < 0·001).
Inhibition of IFN-γ production of TG CD4+ lymphocytes was equivalent when the anti-MHC class II antibodies remained in the cultures for 3 days or were added to APC for 30 min and then the excess antibodies were removed by washing prior to initiation of cocultures (data not shown).
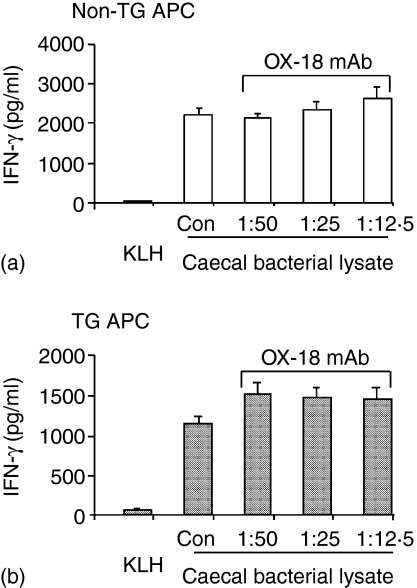
The influence of blockade of rat MHC class I on non-TG or TG APC was also analysed. Addition of anti-rat MHC class I antibody did not have any significant effect on IFN-γ responses in TG CD4+ cells cocultured with either TG or non-TG APC (Fig. 7).
Figure 7.
Blockade of rat MHC class I did not alter IFN-γ production induced by caecal bacterial lysate-pulsed APC in cocultures of TG CD4+ T lymphocytes plus non-TG APC (a) or TG APC (b). Anti-MHC class I antibody (OX-18 monoclonal antibody) at the dilutions indicated, or the isotype control (Con), purified mouse IgG1, at the amount equivalent to the highest concentration of OX-18 mAb, was added to the pulsed SPF APC for 30 min before the start of cocultures with SPF TG CD4+ T lymphocytes. Supernatants were harvested at day 3 and assayed by ELISA. Values represent mean ± SEM of pg/ml of IFN-γ in triplicate culture supernatants. TG CD4+ cells cocultured with an unrelated antigen KLH-pulsed APC served as a control. Results shown are representative of three independent experiments.
Blocking HLA-B27 dramatically suppressed IFN-γ production by TG CD4+ cells cocultured with TG APC
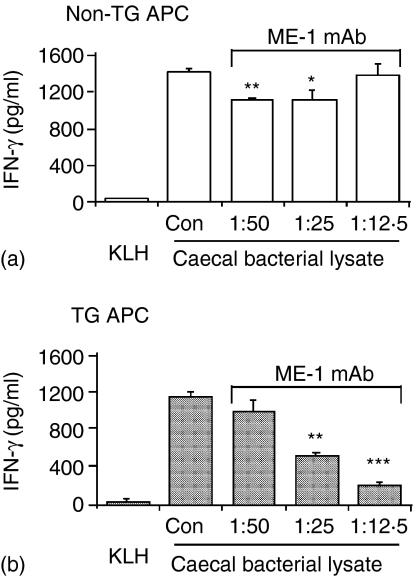
Experiments were carried out to investigate the role of transgenic HLA-B27 in the T-cell response. For these studies, anti-HLA-B27 antibody (ME-1) was incubated with APC as described above. In cocultures containing caecal bacterial lysate-pulsed TG APC, the presence of anti-HLA-B27 antibody caused a marked reduction in IFN-γ production by TG CD4+ T lymphocytes (Fig. 8b). However, the inhibition was not complete even at the highest concentration of antibody (79–87% blockade of IFN-γ responses). As we observed for blocking MHC class II, IFN-γ production was equivalent whether or not the excess anti-HLA-B27 antibody was removed by washing prior to coculture initiation. A small but significant suppression of IFN-γ production was also noted in cocultures containing non-TG APC treated with the anti-HLA-B27 antibody (Fig. 8a), most likely caused by interference with APC–T-cell interaction, by binding to HLA-B27 on TG T cells.
Figure 8.
Blockade of HLA-B27 markedly inhibited IFN-γ production induced by caecal bacterial lysate-pulsed APC in cocultures with TG CD4+ T lymphocytes. Anti-HLA-B27 antibody (ME-1 monoclonal antibody) at the dilutions indicated, or the isotype control (Con), purified mouse IgG1 at the amount equivalent to the highest concentration of ME-1 monoclonal antibody, was added to the pulsed SPF APC for 30 min before the start of cocultures with SPF TG CD4+ T lymphocytes. Supernatants were harvested at day 3 and assayed by ELISA. Values represent mean ± SEM of pg/ml of IFN-γ in triplicate culture supernatants of TG CD4+ T lymphocytes cocultured with non-TG APC (a) or TG APC (b). TG CD4+ cells cocultured with an unrelated antigen KLH-pulsed APC served as a control. Results shown are representative of six independent experiments. Statistically significant differences between the amounts of IFN-c produced in cocultures containing APC treated with anti-HLA-B27 antibody and in cocultures containing APC treated with isotype control are denoted by asterisks (*P < 0·05, **P < 0·01, ***P < 0·001).
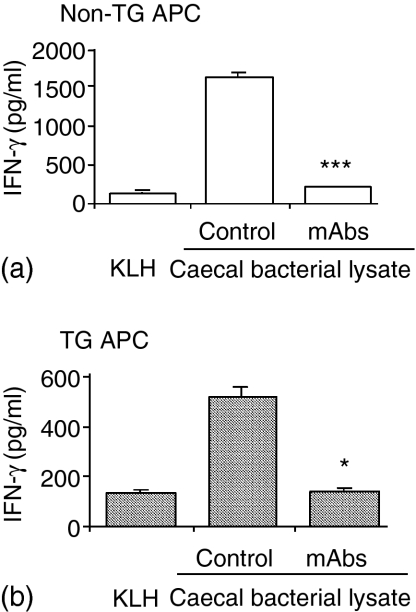
Blockade of both MHC class II and HLA-B27 eliminated the APC-induced IFN-γ response of TG CD4+ cells
When antibodies against products of the two loci of MHC class II (OX-6 and OX-17) and HLA-B27 (ME-1) were added concomitantly, the TG CD4+ T-cell response was abrogated, reaching levels observed with KLH stimulation (Fig. 9). These results indicate that MHC class II and HLA-B27 are the antigen-presenting molecules that activate IFN-γ producing CD4+ T cells.
Figure 9.
Combined administration of antibodies to MHC class II and HLA-B27 completely inhibited IFN-γ production induced by caecal bacterial lysate-pulsed APC in cocultures with TG CD4+ T lymphocytes. Antibodies were added to the pulsed SPF APC for 30 min before the start of cocultures with SPF TG CD4+ T lymphocytes. The isotype control, purified mouse IgG1, was added at the amount equivalent to the highest concentration of the antibodies used. Supernatants were harvested at day 3 and assayed by ELISA. Values represent mean ± SEM of pg/ml of IFN-γ in triplicate culture supernatants of TG CD4+ T lymphocytes cocultured with (a) non-TG APC in the presence of anti-MHC class II RT1. B (OX-6 monoclonal antibody, dilution 1 : 50) plus anti-MHC class II RT1. D (OX-17 monoclonal antibody, dilution 1 : 250) or (b) TG APC in the presence of anti-MHC class II RT1.B, anti-MHC class II RT1.D, and anti-HLA-B27 (ME-1 monoclonal antibody, dilution 1 : 25). TG CD4+ cells cocultured with an unrelated antigen KLH-pulsed APC served as a control. Results shown are representative of four independent experiments. Statistically significant differences between the APC treated with the specific antibodies and the isotype control in inducing IFN-γ production of TG CD4+ cells are denoted by asterisks (*P < 0·05, ***P < 0·001).
Discussion
The principal goal of this study was to evaluate bacterial antigen-activated APC-dependent CD4+ T-lymphocyte response in HLA-B27 TG rats with established colitis. The major findings that emerged are as follows. First, SPF TG CD4+ T lymphocytes produced high levels of IFN-γ in response to both TG and non-TG APC stimulated with physiologically relevant bacterial components, whereas non-TG CD4+ cells did not respond. We have previously shown that MLN cells from GF TG rats did not produce IFN-γ following in vitro stimulation with caecal bacterial lysates.23 Thus CD4+ T-cell reactivity that develops in HLA-B27 TG rats with colitis appears to be dependent on the genetic susceptibility of the host conferred by expression of HLA-B27 and in vivo stimulation of T cells by commensal enteric bacteria. Of interest, APC from both SPF and GF rats had the capacity to induce IFN-γ production by CD4+ T lymphocytes from SPF TG rats, indicating that previous activation of APC by in vivo encounters with enteric bacterial components is not necessary for antigen-presenting function. Somewhat surprisingly, under these in vitro conditions, SPF TG APC were less effective inducers of TG CD4+ T-cell responses than non-TG APC. The reasons for the apparent defect in SPF TG APC function in vitro are not clear, but one possible explanation may be that bacterial antigen-pulsed TG APC do not efficiently up-regulate expression of a key costimulatory molecule, CD86. Such an interpretation is consistent with the findings reported recently by Hacquard-Bouder et al.24 Their results demonstrate that HLA-B27 TG dendritic cells form fewer conjugates with T cells compared to non-TG dendritic cells. Furthermore, CD86 has been shown to be crucial for conjugate formation. Another contributing factor might be that constant activation and challenge in an inflammatory milieu in vivo could gradually compromise the ability of TG APC to be further activated in vitro by the same bacterial antigens and adjuvants. Indeed, it has been reported that systemic stimulation with a soluble tachyzoite extract can result in a ‘paralysis’ of murine dendritic cells as evidenced by their failure to make IL-12 in response to subsequent in vivo restimulation.25
Second, we consistently observed up-regulated membrane expression of MHC class II and lower levels of MHC class I on MLN cells of HLA-B27 TG rats, compared to age-matched non-TG controls. TG rats from both SPF and GF colonies had similar changes in the surface expression of MHC molecules as compared to non-TG rats from the same housing conditions.
Finally, we demonstrated that the IFN-γ response to caecal bacterial lysate-pulsed non-TG APC was dependent on molecules encoded by both of the two loci of MHC class II, while MHC class II and HLA-B27 were the molecules mediating the T-cell response to lysate-pulsed TG APC. Blocking rat MHC class I on either TG or non-TG APC had no effect on the T-cell response.
In humans, the MHC class I allele HLA-B27 is strongly associated with a group of inflammatory disorders that includes ankylosing spondylitis, reactive arthritis, and uveitis/iritis.26 However, the contribution of HLA-B27 to the pathogenesis of these diseases is still unresolved. Several hypotheses have been proposed: (1) HLA-B27 is a particularly efficient restriction element for antigenic bacterial or self peptide binding and presentation; (2) HLA-B27 shares homology in peptide sequences with some enterobacteria. Presenting such closely related peptides by host MHC molecules could potentially lead to cross-reactive immune responses;27–29 (3) peptides derived from HLA-B27 itself are presented by other MHC molecules;30,31 (4) binding of HLA-B27 heavy chain homodimers to populations of lymphoid and myeloid cells including APC via recognition of paired immunoglobulin-like receptors (PIRs) may be important for the initiation of chronic inflammation.32 (5) it has been reported recently that HLA-B27 misfolding is associated with up-regulation of the unfolded protein response (UPR) in the endoplasmic reticulum of macrophages from HLA-B27 TG rats with colitis and that IFN-γ further exacerbates activation of UPR-associated genes.33
In the present study we showed that blockade of HLA-B27 on TG APC resulted in a dose-dependent suppression of the IFN-γ response by TG CD4+ T lymphocytes cocultured with caecal bacterial lysate-pulsed TG APC. These results indicate that the role of HLA-B27 is at least partially to present enteric bacterial peptides. HLA-B27 is unlikely to be the source of peptide antigens that are presented by MHC class II because KLH-pulsed TG APC did not induce IFN-γ responses in TG CD4+ T cells.
In conclusion, caecal bacterial lysate, a complex mixture of physiologically relevant bacterial components including antigens and adjuvants, can elicit an evident in vitro IFN-γ response in APC-activated TG CD4+ T lymphocytes. This T-cell stimulation is triggered through two distinct MHC molecules, HLA-B27 and MHC class II on TG APC. However, HLA-B27 is not required to stimulate in vitro T cell recall responses to caecal bacterial lysate, since bacterial lysate-pulsed non-TG APC efficiently induce IFN-γ production by TG CD4+ T cells. Molecules encoded by both of the two rat MHC class II loci are involved in T-cell activation by APC. Results shown in the present study provide a more complete understanding of the pathogenesis of colitis in HLA-B27 TG rats and thus also clarify the role of the immune response in maintaining chronic intestinal inflammation in this model system.
Acknowledgments
The authors thank Desmond McDonnell for technical assistance at the College of Veterinary Medicine, North Carolina State University, Raleigh, Drs Bo Liu and Feng Ling Li at the University of North Carolina at Chapel Hill for breeding the SPF rats, and Donna Kronstadt, Gnotobiotic Animal Core, Center of Gastrointestinal Biology and Disease at North Carolina State University, for maintaining the GF rat colony.
These studies were supported by US Public Health Service Grants NIH RO1 DK 40249 and 5 P30 DK 34987. Dr Qian is supported by a Research Fellowship Award from the Crohn's and Colitis Foundation of America.
Abbreviations
- APC
antigen-presenting cells
- ELISA
enzyme-linked immunosorbent assay
- GF
germ-free
- IFN
interferon
- IL
interleukin
- KLH
keyhole limpet haemocyanin
- MFI
mean fluorescence intensity
- MHC
major histocompatibility complex
- MLN
mesenteric lymph node
- PIRs
paired immunoglobulin-like receptors
- SPF
specific pathogen free
- TCR
T-cell receptor
- TG
transgenic
- TNF
tumour necrosis factor
- UPR
unfolded protein response
References
- 1.Sartor RB. Microbial influences in inflammatory bowel disease: role in pathogenesis and clinical implications. In: Sartor RB, Sandborn WJ, editors. Kirsner's Inflammatory Bowel Diseases. Philadelphia: Elsevier; 2004. pp. 138–62. [Google Scholar]
- 2.Sartor RB. Animal models of intestinal inflammation. In: Sartor RB, Sandborn WJ, editors. Kirsner's Inflammatory Bowel Diseases. Philadelphia: Elsevier; 2004. pp. 120–37. [Google Scholar]
- 3.Davidson NJ, Hudak SA, Lesley RE, Menon S, Leach MW, Rennick DM. IL-12, but not IFN-gamma, plays a major role in sustaining the chronic phase of colitis in IL-10-deficient mice. J Immunol. 1998;161:3143–9. [PubMed] [Google Scholar]
- 4.Neurath MF, Fuss I, Kelsall BL, Stuber E, Strober W. Antibodies to interleukin 12 abrogate established experimental colitis in mice. J Exp Med. 1995;182:1281–90. doi: 10.1084/jem.182.5.1281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Powrie F, Leach MW, Mauze S, Menon S, Caddle LB, Coffman RL. Inhibition of Th1 responses prevents inflammatory bowel disease in scid mice reconstituted with CD45RBhi CD4+ T cells. Immunity. 1994;1:553–62. doi: 10.1016/1074-7613(94)90045-0. [DOI] [PubMed] [Google Scholar]
- 6.Mackay F, Browning JL, Lawton P, et al. Both the lymphotoxin and tumor necrosis factor pathways are involved in experimental murine models of colitis. Gastroenterology. 1998;115:1464–75. doi: 10.1016/s0016-5085(98)70025-3. [DOI] [PubMed] [Google Scholar]
- 7.Targan SR, Hanauer SB, van Deventer SJ, et al. A short-term study of chimeric monoclonal antibody cA2 to tumor necrosis factor alpha for Crohn's disease. Crohn's Disease cA2 Study Group. N Engl J Med. 1997;337:1029–35. doi: 10.1056/NEJM199710093371502. [DOI] [PubMed] [Google Scholar]
- 8.Mannon PJ, Fuss IJ, Mayer L, et al. Anti-interleukin-12 antibody for active Crohn's disease. N Engl J Med. 2004;351:2069–79. doi: 10.1056/NEJMoa033402. [DOI] [PubMed] [Google Scholar]
- 9.Hammer RE, Maika SD, Richardson JA, Tang JP, Taurog JD. Spontaneous inflammatory disease in transgenic rats expressing HLA-B27 and human beta 2m. an animal model of HLA-B27-associated human disorders. Cell. 1990;63:1099–112. doi: 10.1016/0092-8674(90)90512-d. [DOI] [PubMed] [Google Scholar]
- 10.Breban M, Fernandez-Sueiro JL, Richardson JA, Hadavand RR, Maika SD, Hammer RE, Taurog JD. T cells, but not thymic exposure to HLA-B27, are required for the inflammatory disease of HLA-B27 transgenic rats. J Immunol. 1996;156:794–803. [PubMed] [Google Scholar]
- 11.Breban M, Hammer RE, Richardson JA, Taurog JD. Transfer of the inflammatory disease of HLA-B27 transgenic rats by bone marrow engraftment. J Exp Med. 1993;178:1607–16. doi: 10.1084/jem.178.5.1607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Krajina T, Leithauser F, Moller P, Trobonjaca Z, Reimann J. Colonic lamina propria dendritic cells in mice with CD4+ T cell-induced colitis. Eur J Immunol. 2003;33:1073–83. doi: 10.1002/eji.200323518. [DOI] [PubMed] [Google Scholar]
- 13.Rath HC, Herfarth HH, Ikeda JS, et al. Normal luminal bacteria, especially Bacteroides species, mediate chronic colitis, gastritis, and arthritis in HLA-B27/human beta2 microglobulin transgenic rats. J Clin Invest. 1996;98:945–53. doi: 10.1172/JCI118878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sellon RK, Tonkonogy S, Schultz M, Dieleman LA, Grenther W, Balish E, Rennick DM, Sartor RB. Resident enteric bacteria are necessary for development of spontaneous colitis and immune system activation in interleukin-10-deficient mice. Infect Immun. 1998;66:5224–31. doi: 10.1128/iai.66.11.5224-5231.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Taurog JD, Richardson JA, Croft JT, Simmons WA, Zhou M, Fernandez-Sueiro JL, Balish E, Hammer RE. The germfree state prevents development of gut and joint inflammatory disease in HLA-B27 transgenic rats. J Exp Med. 1994;180:2359–64. doi: 10.1084/jem.180.6.2359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Veltkamp C, Tonkonogy SL, De Jong YP, Albright C, Grenther WB, Balish E, Terhorst C, Sartor RB. Continuous stimulation by normal luminal bacteria is essential for the development and perpetuation of colitis in Tg (epsilon26) mice. Gastroenterology. 2001;120:900–13. doi: 10.1053/gast.2001.22547. [DOI] [PubMed] [Google Scholar]
- 17.Schultz M, Tonkonogy SL, Sellon RK, et al. IL-2-deficient mice raised under germfree conditions develop delayed mild focal intestinal inflammation. Am J Physiol. 1999;276:G1461–72. doi: 10.1152/ajpgi.1999.276.6.G1461. [DOI] [PubMed] [Google Scholar]
- 18.Hoentjen F, Harmsen HJ, Braat H, Torrice CD, Mann BA, Sartor RB, Dieleman LA. Antibiotics with a selective aerobic or anaerobic spectrum have different therapeutic activities in various regions of the colon in interleukin 10 gene deficient mice. Gut. 2003;52:1721–7. doi: 10.1136/gut.52.12.1721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Linskens RK, Huijsdens XW, Savelkoul PH, Vandenbroucke-Grauls CM, Meuwissen SG. The bacterial flora in inflammatory bowel disease: current insights in pathogenesis and the influence of antibiotics and probiotics. Scand J Gastroenterol Suppl. 2001;234:29–40. doi: 10.1080/003655201753265082. [DOI] [PubMed] [Google Scholar]
- 20.Madsen KL, Doyle JS, Tavernini MM, Jewell LD, Rennie RP, Fedorak RN. Antibiotic therapy attenuates colitis in interleukin 10 gene-deficient mice. Gastroenterology. 2000;118:1094–105. doi: 10.1016/s0016-5085(00)70362-3. [DOI] [PubMed] [Google Scholar]
- 21.Rath HC, Schultz M, Freitag R, Dieleman LA, Li F, Linde HJ, Scholmerich J, Sartor RB. Different subsets of enteric bacteria induce and perpetuate experimental colitis in rats and mice. Infect Immun. 2001;69:2277–85. doi: 10.1128/IAI.69.4.2277-2285.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cong Y, Brandwein SL, McCabe RP, Lazenby A, Birkenmeier EH, Sundberg JP, Elson CO. CD4+ T cells reactive to enteric bacterial antigens in spontaneously colitic C3H/HeJBir mice: increased T helper cell type 1 response and ability to transfer disease. J Exp Med. 1998;187:855–64. doi: 10.1084/jem.187.6.855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Dieleman LA, Hoentjen F, Qian BF, et al. Reduced ratio of protective versus proinflammatory cytokine responses to commensal bacteria in HLA-B27 transgenic rats. Clin Exp Immunol. 2004;136:30–9. doi: 10.1111/j.1365-2249.2004.02410.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hacquard-Bouder C, Falgarone G, Bosquet A, Smaoui F, Monnet D, Ittah M, Breban M. Defective costimulatory function is a striking feature of antigen-presenting cells in an HLA-B27-transgenic rat model of spondylarthropathy. Arthritis Rheum. 2004;50:1624–35. doi: 10.1002/art.20211. [DOI] [PubMed] [Google Scholar]
- 25.Reis e Sousa C, Yap G, Schulz O, Rogers N, Schito M, Aliberti J, Hieny S, Sher A. Paralysis of dendritic cell IL-12 production by microbial products prevents infection-induced immunopathology. Immunity. 1999;11:637–47. doi: 10.1016/s1074-7613(00)80138-7. [DOI] [PubMed] [Google Scholar]
- 26.Said-Nahal R, Miceli-Richard C, Gautreau C, et al. The role of HLA genes in familial spondyloarthropathy: a comprehensive study of 70 multiplex families. Ann Rheum Dis. 2002;61:201–6. doi: 10.1136/ard.61.3.201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Frauendorf E, von Goessel H, May E, Marker-Hermann E. HLA-B27-restricted T cells from patients with ankylosing spondylitis recognize peptides from B*2705 that are similar to bacteria-derived peptides. Clin Exp Immunol. 2003;134:351–9. doi: 10.1046/j.1365-2249.2003.02289.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Scofield RH, Warren WL, Koelsch G, Harley JB. A hypothesis for the HLA-B27 immune dysregulation in spondyloarthropathy: contributions from enteric organisms, B27 structure, peptides bound by B27, and convergent evolution. Proc Natl Acad Sci USA. 1993;90:9330–4. doi: 10.1073/pnas.90.20.9330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Scofield RH, Kurien B, Gross T, Warren WL, Harley JB. HLA-B27 binding of peptide from its own sequence and similar peptides from bacteria: implications for spondyloarthropathies. Lancet. 1995;345:1542–4. doi: 10.1016/s0140-6736(95)91089-1. [DOI] [PubMed] [Google Scholar]
- 30.Parham P. Presentation of HLA class I-derived peptides: potential involvement in allorecognition and HLA-B27-associated arthritis. Immunol Rev. 1996;154:137–54. doi: 10.1111/j.1600-065x.1996.tb00932.x. [DOI] [PubMed] [Google Scholar]
- 31.Sieper J, Kingsley G. Recent advances in the pathogenesis of reactive arthritis. Immunol Today. 1996;17:160–3. doi: 10.1016/0167-5699(96)80612-8. [DOI] [PubMed] [Google Scholar]
- 32.Kollnberger S, Bird LA, Roddis M, et al. HLA-B27 heavy chain homodimers are expressed in HLA-B27 transgenic rodent models of spondyloarthritis and are ligands for paired Ig-like receptors. J Immunol. 2004;173:1699–710. doi: 10.4049/jimmunol.173.3.1699. [DOI] [PubMed] [Google Scholar]
- 33.Turner MJ, Sowders DP, DeLay ML, et al. HLA-B27 misfolding in transgenic rats is associated with activation of the unfolded protein response. J Immunol. 2005;175:2438–48. doi: 10.4049/jimmunol.175.4.2438. [DOI] [PubMed] [Google Scholar]