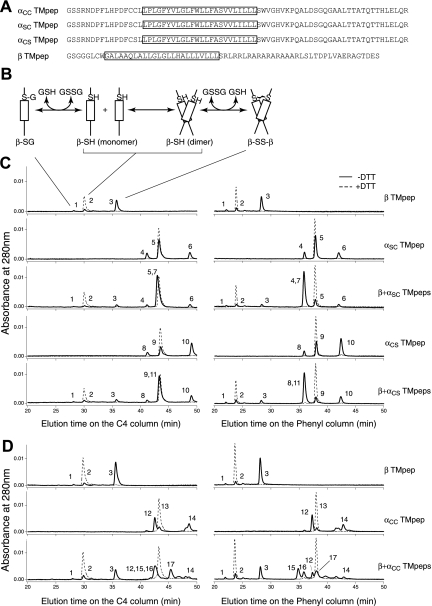
Figure 3.
Thiol-disulfide exchange assay of the TM peptides derived from GP Ibα and GP Ibβ. (A) Sequences of the Ibα and Ibβ TM peptides. The TM domain in each construct is boxed. Residue C148 in the Ibβ cytoplasmic domain was changed to Ser (the residue in the rat sequence) to avoid complication in data analysis. A GGG sequence was added between the thrombin cleavage site and the Ibβ sequence to facilitate thrombin digestion. (B) A reaction scheme for the Ibβ TM peptide. The TM-TM interaction mediates the dimerization step, which is coupled to formation of a disulfide bond between 2 Ibβ peptides. GSH indicates reduced glutathione; GSSG, oxidized glutathione; β-SG, Ibβ peptide linked to glutathione; β-SH, reduced monomeric Ibβ peptide; β-SS-β, disulfide-linked Ibβ homodimer. (C) The thiol-disulfide exchange reaction of the Ibβ TM peptide with the Ibα TM peptides containing one Cys residue. The TM peptides were mixed with DPC micelles and incubated at room temperature for the reaction to reach equilibrium. Acid was then added to quench the reaction, and the reaction products were analyzed by reverse-phase analytic HPLC using 2 different columns. DTT in the reaction prevented formation of any disulfides, and their chromatograms (dashed line) helped to assign the eluted peaks. Peak 1 indicates β-SG; peak 2, β-SH; peak 3, β-SS-β; peak 4, αSC-SG; peak 5, αSC-SH; peak 6, αSC-SS-αSC; peak 7, αSC-SS-β; peak 8, αCS-SG; peak 9, αCS-SH; peak 10, αCS-SS-αCS; peak 11, αCS-SS-β. (D) The thiol-disulfide exchange reaction of the Ibβ TM peptide with the αCC TM peptide, at the 2:1 molar ratio. Peak 12 indicates oxidized monomeric αCC with an intramolecular disulfide; peak 13, reduced monomeric αCC; peak 14, oxidized dimeric αCC with 2 intermolecular disulfides; peak 15, β-SS-αCC-SS-β; peak 16, αCC-SS-β; peak 17, β-SS-αCC-SS-αCC-SS-β.