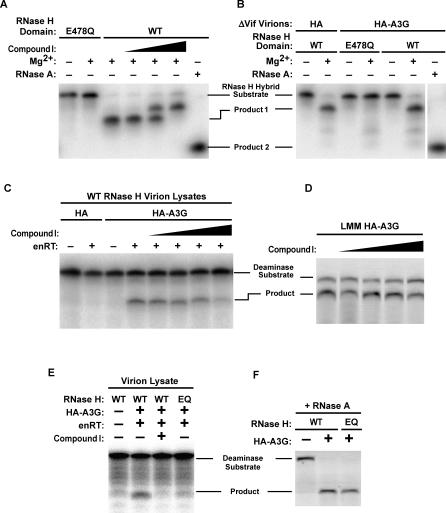
Figure 6. Enzymatically Inactive Virion-Incorporated HA-A3G Is Activated by Viral RNase H.
(A) Recombinant RTs containing either a WT or mutant (E478Q) RNase H catalytic domain were assessed for RNase H activity in vitro in the absence or presence of the RNase H inhibitor Compound I (final concentration of 1, 10, or 100 μM). The RNA of an RNA–DNA hybrid remains intact unless RNase H digests the RNA into a smaller cleavage product that is distinguishable from the more complete cleavage product generated by RNase A. WT RNase H cannot digest ssDNA or DNA of an RNA–DNA hybrid, or RNA–RNA hybrids (data not shown). RNase H assays were performed in RNase H buffer (50 mM Tris [pH 8.0], 60 mM KCl) with (+) or without (−) 5 mM MgCl2 or RNase A, as indicated.
(B) Viruses bearing the RNase H E478Q mutation are compromised for in vitro RNase H activity. RNase H assays were performed in RNase H buffer with (+) or without (−) 5 mM MgCl2 or RNase A, as indicated.
(C) Virion lysates were subjected to endogenous reverse transcription (enRT) conditions with or without Compound I (final concentration of 0.1, 1, 10, or 100 μM), and A3G activity in these samples assessed in the in vitro deoxycytidine deaminase assay. Deaminase assays were performed in RNase H buffer either supplemented (enRT:+) or not (enRT:−) with 4 mM MgCl2 and 1 mM dNTPs.
(D) Compound I does not inhibit the intrinsic deoxycytidine deaminase activity of A3G. HA-A3G from RNase A–treated virion lysates was assessed for in vitro deaminase activity in the presence of increasing doses of Compound I (0.1, 1, 10, and 100 μM). Deaminase assay was performed in RNase H buffer supplemented with RNase A only.
(E) Virions containing WT RNase H or the E478Q mutation in the RNase H catalytic domain were subjected to the enRT reaction followed by assessment of A3G enzymatic activity. Deaminase assays were performed in RNase H buffer either supplemented (enRT:+) or not (enRT:−) with 4 mM MgCl2 and 1 mM dNTPs.
(F) WT and RNase H–compromised ΔVif virions containing WT or mutant RNase H displayed equivalent A3G activity when RNase A was added to the virion lysate. Deaminase assay was performed in RNase H buffer with (+) or without (−) RNase A, as indicated.
All data are representative of multiple experiments.