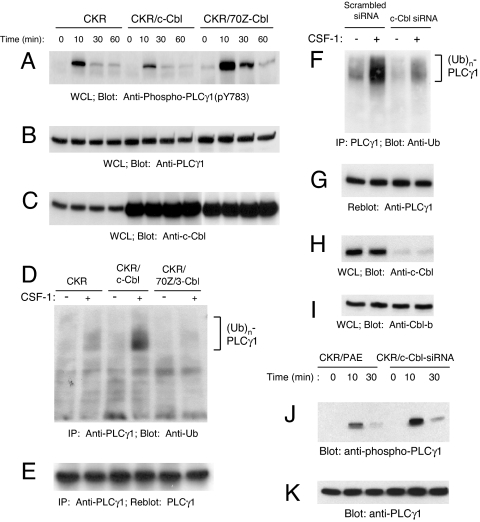
Fig. 1.
c-Cbl inhibits PLCγ1 tyrosine phosphorylation and promotes its ubiquitylation in endothelial cells. (A) PAE cells expressing either CKR (chimeric VEGFR-2) alone, coexpressing the wild-type Cbl or 70Z/3-Cbl were either unstimulated or stimulated with CSF-1 for various times as indicated. PLCγ1 activation in normalized whole cell lysates (WCL) was analyzed by immunoblotting with a phospho-specific (pY783) PLCγ1 antibody. (B) PLCγ1 expression in whole cell lysates was determined in a parallel immunoblot using an anti-PLCγ1 antibody. (C) overexpression of c-Cbl and 70Z/3-Cbl proteins was determined by immunoblotting with an anti-c-Cbl antibody. (D) PLCγ1 was immunoprecipitated (IP) from RIPA whole cell lysates and was assessed by immunoblotting with an anti-ubiquitin (Ub) antibody. (E) The blot shown in D was stripped and reprobed with an anti-PLCγ1 antibody to demonstrate equal amounts of immunoprecipitated PLCγ1. (F) CKR/PAE cells were retrovirally transduced with either constructs expressing a control siRNA (Scrambled siRNA) or a siRNA targeting c-Cbl (c-Cbl siRNA). The aforementioned cell lines were left either unstimulated or stimulated for 10 min, immunoprecipitated with anti-PLCγ1 antibody, and immunoblotted with anti-ubiquitin antibody. (G) The same membrane was immunoblotted with anti-PLCγ1 antibody. (H and I) Whole cell lysates were immunoblotted with anti-c-Cbl (H) or anti-Cbl-b (I) antibodies. (J and K) CKR/PAE cells and CKR/PAE cells expressing c-Cbl-siRNA were stimulated with CSF-1 for various times as indicated, and whole cell lysates was analyzed by immunoblotting with a phospho-specific (pY783) PLCγ1 antibody (J) or with an anti-PLCγ1 antibody (K).