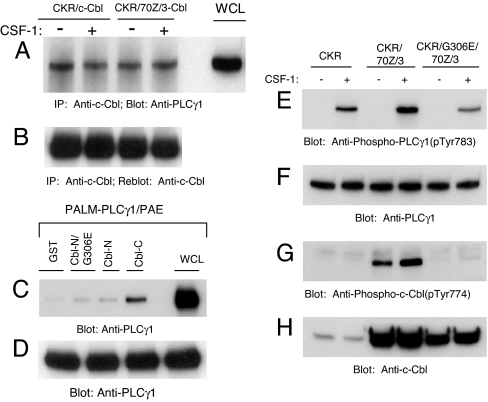
Fig. 3.
c-Cbl constitutively associates with PLCγ1 via its carboxyl domain. (A) CKR/c-Cbl/PAE and CKR/70Z/3-Cbl/PAE cells were either unstimulated (−) or stimulated (+) for 10 min with CSF-1. Normalized whole cell lysates were immunoprecipitated (IP) with an anti-c-Cbl antibody and immunoprecipitates were immunoblotted with an anti-PLCγ1 antibody. Positive controls consist of whole cell lysate (WCL) aliquots and are indicated as such. (B) The same membrane was stripped and reprobed with an anti-c-Cbl antibody. (C) PAE cells ectopically expressing constitutively active PLCγ1 (Palm-PLCγ1) were serum-starved overnight. Normalized whole cell lysates from each of the four dishes were incubated separately with equal amounts of GST, GST-Cbl-N/G306E, GST-Cbl-N, and GST-Cbl-C fusion proteins as indicated in an in vitro GST pull-down assay. Precipitated PLCγ1 was analyzed by immunoblotting with an anti-PLCγ1 antibody. Positive controls consist of whole cell lysate aliquots and are indicated as such. (D) A parallel blot of whole cell lysates was probed with an anti-PLCγ1 antibody as a loading control. (E) PAE cells either expressing wild-type CKR alone or with the indicated Cbl constructs were either unstimulated (−) or stimulated (+) for 10 min with CSF-1 and normalized whole cell lysates were immunoblotted with an anti-phospho-PLCγ1 antibody (pTyr783). (F) Parallel blot of whole cell lysates were probed with an anti-PLCγ1 antibody. (G and H) Phosphorylation of 70Z/3-Cbl and G306E-70Z/3-Cbl and their expression.