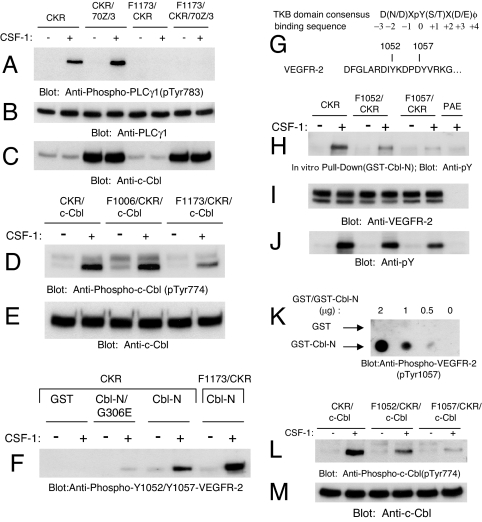
Fig. 4.
Role of tyrosines 1173, 1052, and 1057 of VEGFR-2 in the recruitment and tyrosine phosphorylation of c-Cbl. (A) PAE cells expressing CKR, F1173/CKR alone or coexpressing F1173/CKR with 70Z/3-Cbl were either unstimulated (−) or stimulated (+) for 10 min with CSF-1, and whole cell lysates were immunoblotted with an anti-phospho-PLCγ1 antibody (pTyr783). (B and C) Parallel blots of whole cell lysates were probed with an anti-PLCγ1 (B) and an anti-c-Cbl (C) antibodies to show their expression. (D) PAE cells coexpressing CKR with c-Cbl, F1006/CKR with c-Cbl, and F1173/CKR with c-Cbl were either unstimulated (−) or stimulated (+) for 10 min with CSF-1, and whole cell lysates were immunoblotted with an anti-phospho-c-Cbl antibody (pTyr774). (E) Parallel blot of whole cell lysates was probed with an anti-c-Cbl antibody. (F) CKR/PAE and F1173/CKR/PAE cells were treated as in A. Whole cell lysates were incubated with equal amounts of GST, GST-Cbl-N/G306E, and GST-Cbl-N fusion proteins as indicated in an in vitro GST pull-down assay. Precipitated CKR was analyzed by immunoblotting with an anti-phospho-VEGFR-2 antibody that detects VEGFR-2 autophosphorylated at Y1052 and Y1057 (pTyr1052/pTyr1057). (G) The c-Cbl TKB domain consensus binding sequence and a partial alignment of the activation loop primary amino acid sequence of VEGFR-2. (H) PAE cells expressing either wild-type CKR or the indicated activation loop mutants were unstimulated (−) or stimulated (+) for 10 min with CSF-1. Whole cell lysates were incubated with equal amounts of a GST-Cbl-N fusion protein in an in vitro GST pull-down assay. Cell lysates from CSF-1-stimulated PAE cells were used as a negative control. CKR·GST-Cbl-N complexes were analyzed by immunoblotting with an anti-phosphotyrosine antibody (pY). (I) A parallel blot of whole cell lysates was probed with an anti-VEGFR-2 antibody (1412) as a control for receptor levels. (J) A parallel blot of whole cell lysates was probed with an anti-phosphotyrosine antibody (pY). (K) To detect a direct interaction between the Cbl TKB domain and VEGFR-2 activation loop tyrosines, the indicated quantities of purified recombinant GST control (Upper) and GST-Cbl-N fusion proteins (Lower) were dot blotted as described in Materials and Methods. (L and M) PAE cells coexpressing wild-type c-Cbl and either wild-type CKR or the indicated mutant receptors were treated as described in H. Whole cell lysates were immunoblotted in parallel with anti-phospho-c-Cbl and anti-c-Cbl antibodies.