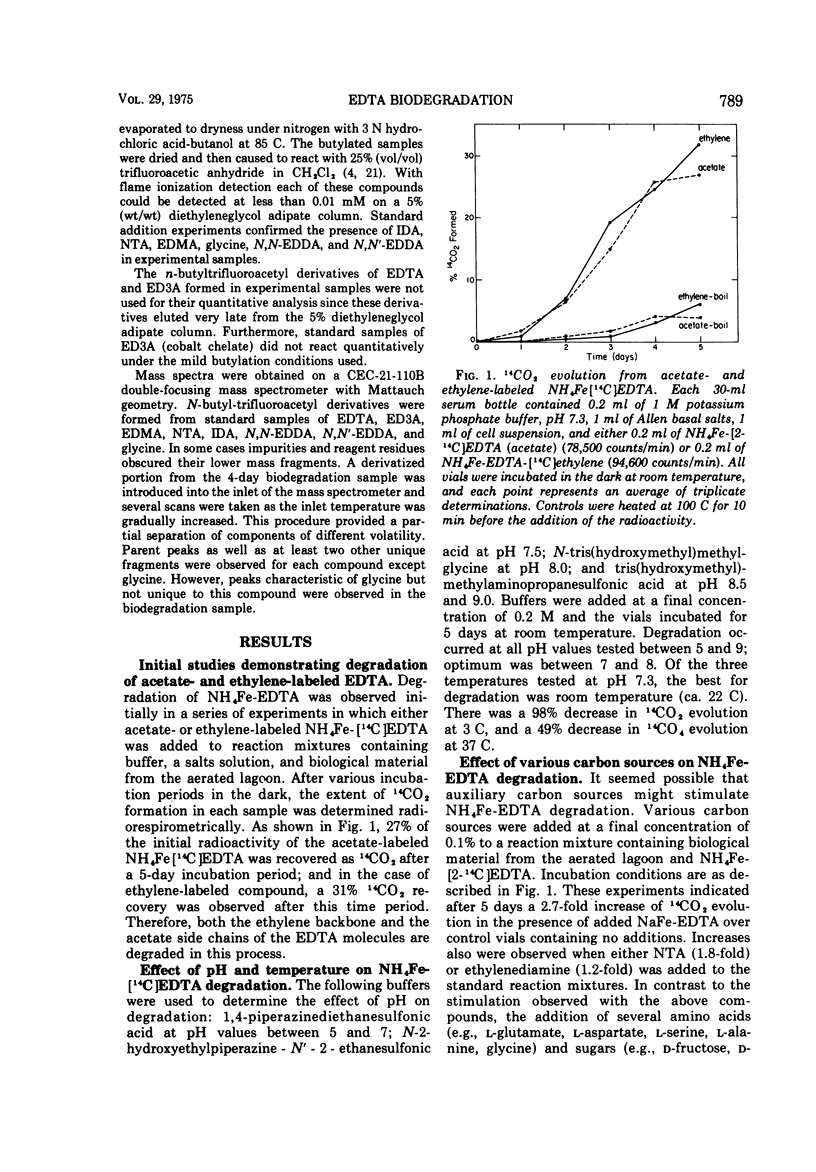
Abstract
The ferric chelate of ethylenediaminetetraacetic acid (EDTA) was biologically degraded by a mixed population of microorganisms present in an aerated lagoon receiving this chemical in its feed. As determined radiorespirometrically, 28% of the acetate-2-C and 30% of the ethylene position of the ammonium ferric chelate of [14C]EDTA was recovered as 14CO2 after 5 days. In a separate experiment using gas liquid chromatography and the sodium ferric chelate, as much as 89% disappearance of EDTA (0.1% wt/vol) was observed during a similar time period. Optimum 14CO2 evolution was observed at a pH value between 7 and 8 and at room temperature. Degradation of NH4Fe-[2-14C]EDTA was stimulated by the addition of either unlabeled NaFe-EDTA, nitrilotriacetic acid or ethylenediamine, and inhibited by the addition of a variety of different sugars and amino acids. Consistent with the biological nature of this degradation, little or no 14CO2 evolution was observed after heat treatment of the microorganisms at 100 C for 10 min, or after the addition of formalin or antibiotics to the incubation mixtures. Gas-liquid chromatography and mass spectral analyses were performed to demonstrate EDTA disappearance and to identify various possible intermediates of EDTA degradation.
Full text
PDF






Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- ALLEN M. B. Studies with Cyanidium caldarium, an anomalously pigmented chlorophyte. Arch Mikrobiol. 1959;32(3):270–277. doi: 10.1007/BF00409348. [DOI] [PubMed] [Google Scholar]
- Atherton E., Law H. D., Moore S., Elliott D. F., Wade R. Synthesis of peptides containing N-2-aminoethylglycine--'reduction analogues'. J Chem Soc Perkin 1. 1971;20:3393–3398. [PubMed] [Google Scholar]
- Aue W. A., Hastings C. R., Gerhardt K., Pierce J. O., 2nd, Hill H. H., Moseman R. F. The determination of part-per-billion levels of citric and nitrilotriacetic acids in tap water and sewage effluents. J Chromatogr. 1972 Oct 18;72(2):259–267. doi: 10.1016/s0021-9673(00)89509-6. [DOI] [PubMed] [Google Scholar]
- Cripps R. E., Noble A. S. The metabolism of nitrilotriacetate by a pseudomonad. Biochem J. 1973 Dec;136(4):1059–1068. doi: 10.1042/bj1361059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eady R. R., Jarman T. R., Large P. J. Microbial oxidation of amines. Partial purification of a mixed-function secondary-amine oxidase system from Pseudomonas aminovorans that contains an enzymically active cytochrome-P-420-type haemoprotein. Biochem J. 1971 Nov;125(2):449–459. doi: 10.1042/bj1250449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Focht D. D., Joseph H. A. Bacterial degradation of nitrilotriacetic acid (NTA). Can J Microbiol. 1971 Dec;17(12):1553–1556. doi: 10.1139/m71-247. [DOI] [PubMed] [Google Scholar]
- Smith D. W., Fliermans C. B., Brock T. D. Technique for measuring 14 CO 2 uptake by soil microorganisms in situ. Appl Microbiol. 1972 Mar;23(3):595–600. doi: 10.1128/am.23.3.595-600.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tiedje J. M., Mason B. B., Warren C. B., Malec E. J. Metabolism of nitrilotriacetate by cells of Pseudomonas species. Appl Microbiol. 1973 May;25(5):811–818. doi: 10.1128/am.25.5.811-818.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warren C. B., Malec E. J. Quantitative determination of nitrilotriacetic acid and related aminopolycarboxylic acids in inland waters. Analysis by gas chromatography. J Chromatogr. 1972 Feb 2;64(2):219–237. doi: 10.1016/s0021-9673(00)85400-x. [DOI] [PubMed] [Google Scholar]


