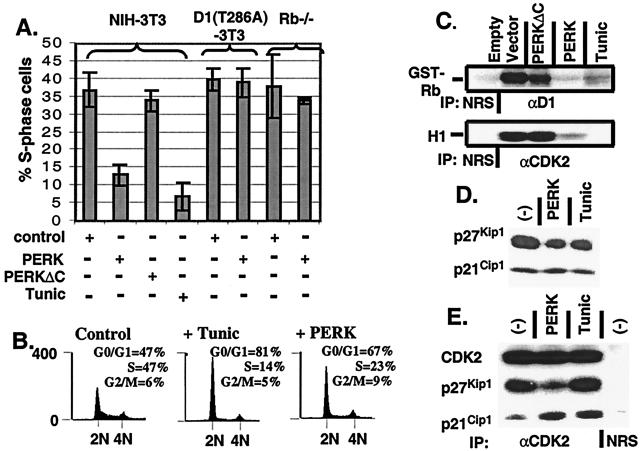
Figure 3.
Enforced overexpression of cyclin D1 prevents PERK-dependent cell-cycle arrest. (A) NIH 3T3 cells, cyclin D1-T286A derivatives, and Rb−/− MEFs were infected with the viruses indicated at the bottom of the graph. Empty virus-infected cells were left untreated or treated with tunicamycin for 20 h. During the last 2 h of tunicamycin treatment, all cell populations were pulsed with BrdUrd and processed for immunofluorescence with a BrdUrd-specific monoclonal antibody except for Rb−/− MEFs in which the S-phase fraction was determined by flow cytometry, as in B. The number of BrdUrd-positive cells from a minimum of three independent experiments were quantitated and are expressed relative to the total population of cells. (B) NIH 3T3 cells were infected with virus encoding PERK or empty virus (control) and left uninfected or treated with 0.5 μg/ml tunicamycin for 20 h. Forty-eight hours after infection (or 20 h after addition of tunicamycin), cells were stained with propidium iodide and assayed for DNA content by flow cytometry (14). (C) NIH 3T3 cells were infected the indicated retroviruses, lysed, and immune complexes were recovered with either a cyclin D1 antibody or a CDK2 antibody and analyzed for protein kinase activity by using retinoblastoma protein or histone H1 as the substrate respectively. (D) Whole-cell lysates prepared from NIH 3T3 cells left untreated, treated with 0.5 μg/ml tunicamycin for 20 h, or infected with virus encoding PERK were subjected to a direct immunoblot analysis by using either p21Cip1- or p27Kip1-specific antibodies. (E) Whole-cell lysates prepared from cells treated as in D were precipitated with antibodies to CDK2. Denatured immune complexes separated on gels were blotted with antibodies to CDK2, p21Cip1, or p27Kip1.