Abstract
Background:
The purpose of this study was to identify factors that may be associated with the development of a sterile inflammatory reaction to OsteoSet (Wright Medical Technology) bone graft substitute. We report on a series of 58 consecutive cases of patients with benign bone lesions who developed an adverse reaction to Osteoset. All procedures were performed by a single orthopaedic oncology surgeon.
Methods:
Patients who exhibited an adverse reaction to OsteoSet and those who did not were analyzed for statistical differences with respect to age, sex, volume of graft material used, size of pellet, use of alcohol washes, mixing of OsteoSet with allograft, medications, drug allergies, and smoking history. Comparisons were made between the reaction group and the non-reaction group with respect to the these variables.
Results:
Eight definite (13.8%) and 3 probable (5.2%) reactions were identified. Symptoms developed an average of 38.9 days after the operation. There was no statistically significant difference (p>0.05) in the volume of Osteoset used, size of pellet, use of alcohol washes, mixing of OsteoSet with allograft, patient age, sex, drug allergies, or smoking history. Reported use of at least one medication was statistically significant between groups when probable reactions were included with the non-reaction group (p < 0.05), but not when probables were analyzed with the definite reaction group ( p < 0.10). A review of our patients' medication lists did not reveal any single or combination of medications likely to be responsible for this difference. Type and location of tumor also did not appear to differ significantly between the two groups.
Conclusions:
Although we report a 13.8%-19.0% incidence of adverse reaction to OsteoSet, we are unable to identify any statistically significant factors that might predict the development of a reaction. Fortunately, the reaction is essentially self limited and fairly benign requiring only anti-inflammatories and observation.
INTRODUCTION
The search for a satisfactory substitute for autogenous bone graft has been at the forefront of research in orthopaedics for many decades. The goal is to match the known success rate and dependability of autograft while avoiding the morbidity of harvest. There have been countless products on the market in recent years, and the quest for the perfect substitute continues.1
In 1999, OsteoSet pellets where introduced by Wright Medical Technology (Arlington, TN). OsteoSet has since gained wide spread use.2 OsteoSet is a surgical grade calcium sulfate in a pure, uniform crystalline form. It is available in pellets of two different sizes and with various devices to aid in its implantation. It is marketed for use in a variety of clinical situations including trauma, tumors and reconstruction. At our institution, it is used primarily after excision of benign tumors and as filler after autograft harvest from metaphyseal bone in various locations.
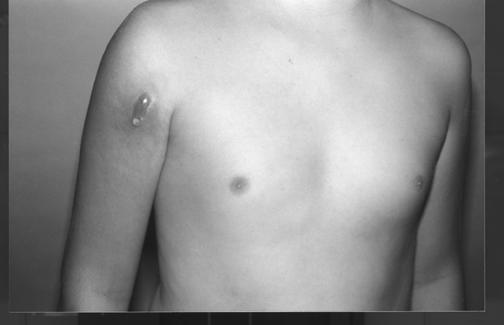
Near the beginning of our experience with OsteoSet, a 27-year-old male presented to clinic with a recurrent giant cell tumor in the proximal tibia. The tumor was curretted, the cavity washed three times with 95% alcohol then packed tightly with OsteoSet. He returned 12 days later with an unusual reaction. His skin was grossly intact but thin and shiny. The entire area immediately surrounding the wound had erupted with edema and erythema. The peri-incisional area appeared to be heaped up and copious amounts of serous drainage could be expressed from the wound. We had no previous experience with this and assumed it to be an infection. The patient was taken immediately to the operating room and the wound explored. The OsteoSet was seeping out of the wound and surrounded with a white, foamy fluid. The area was cultured then vigorously debrided. Gram stain showed inflammatory cells but no organisms; cultures were negative. His laboratory work was entirely negative. He did well and went on to heal his wound uneventfully. Several months later we encountered another similar case with the same clinical and laboratory findings, but no obvious evidence of infection. Similar reactions began to occur elsewhere and a few case reports appeared in the literature.3,4 It became apparent that the use of OsteoSet was associated with some unknown rate of sterile, inflammatory reaction. Following these two cases, we have had six other reactions that were observed and treated with anti-inflammatories. All have done well, and none required surgery. The graft incorporated uneventfully (Figures 1 and 2).
Figure 1.

Figure 2.
The purpose of this study was to identify factors that may be associated with the development of an adverse reaction to OsteoSet. Since 1999, we have used OsteoSet in 58 consecutive cases performed by a single orthopaedic oncology surgeon at our academic medical center. In most cases, it was used after curettage of benign lesions of bone.
MATERIAL AND METHODS
We performed a retrospective review of all cases in which OsteoSet was used to determine if the following factors were associated with the development of a reaction:
Age
Sex
Volume of OsteoSet used
Size of pellet
Use of alcohol washes
Mixing of OsteoSet with allograft
Medications
Drug allergies
Smoking history
Type of tumor
-
Location of tumor
Which bone
Epiphyseal v. metaphyseal v. diaphyseal location
Superficial v. deep
No cases were excluded. All data was complete. A probable adverse reaction was noted when the post-operative incision site was edematous, erythematous, and appeared to have collected fluid in the absence of other signs of infection. Active drainage or expression of serous fluid was an additional criterion for inclusion in the definite adverse reaction group. The data was entered into a spreadsheet and analyzed for any differences in the above factors with reference to the appearance of a reaction (Table 1). We reported on 58 total cases, 29 males and 29 females, with an average age of 26.5 years at the time of Osteoset placement (range, 4 to 66 years). On average, 22.8 mL of OsteoSet was used per case (range, 2.5 to 60 mL). There were 27 cases (46.6%) performed using only the 3.3 mm size pellets, 22 cases (37.9%) with the 4.8 mm pellets, and 9 instances (15.5%) where pellets of both size were used. Out of 58 total procedures, allograft was mixed with OsteoSet on 18 occasions (31.0%) and alcohol washes were used 26 times (44.8%). In addition, our study population consisted of 8 reported smokers (13.8%), 35 patients (60.3%) taking at least one medication, and 20 patients (34.5%) with known drug allergy. Unicameral bone cyst was the most common surgically excised tumor with 17 occurrences (29.3%). There were 8 diagnoses of fibrous dysplasias (13.8%), 7 of giant cell tumor (12.1%), 6 of aneurysmal bone cyst (10.3%), 4 non-ossifying fibromas (6.9%), 2 chondroblastomas (3.4%), 2 chondrosarcomas (3.4%), and 12 other diagnoses (20.7%).
TABLE 1. STUDY POPULATION.
| N | SEX | AGE (years) |
VOL (ml) |
PELLET SIZE |
ALLOGRAFT MIX |
EtOH WASH |
SMOKER | MEDS | ALLERGIES |
|---|---|---|---|---|---|---|---|---|---|
| 58 | 29 M 29 F |
26.5 ±17.3 |
22.6 ±19.0 |
27 (3.3 mm) 22 (4.8 mm) 9 (mixed) |
18 Yes 40 No |
26 Yes 32 No |
8 Yes 50 No |
35 Yes 23 No |
20 Yes 38 No |
STATISTICAL METHODS
The statistical analysis was performed with use of a Student two-sample t test for parametric variables and chi-square analysis for non-parametric variables. P values of less than 0.05 were considered significant. Comparisons were made between the reaction group and the non-reaction group both inclusive and exclusive of probable reactions. These groups were analyzed for statistical differences with respect to age, sex, volume of graft material used, size of pellet, use of alcohol washes, mixing of OsteoSet with allograft, medications, drug allergies, and smoking history.
RESULTS
Eight definite (13.8%) and 3 probable (5.2%) reactions were identified by chart review. These 8 patients averaged 38.9 postoperative days before reporting symptoms consistent with an adverse reaction. When probable reactions were included, these 11 patients averaged 34.4 postoperative days before presenting with symptoms. In addition, the definite reaction group was analyzed both with and without the probable group (Tables 2–5).
TABLE 2. REACTION GROUP + PROBABLE REACTORS.
| N | SEX | AGE (years) |
VOL (ml) |
PELLET SIZE |
ALLOGRAFT MIX |
EtOH WASH |
SMOKER | MEDS | ALLERGIES |
|---|---|---|---|---|---|---|---|---|---|
| 11 | 8 M 3 F |
26.0 ±18.0 |
21.0 ±16.1 |
8 (3.3 mm) 2 (4.8 mm) 1 (mixed) |
2 Yes 9 No |
5 Yes 6 No |
2 Yes 9 No |
4 Yes 7 No |
3 Yes 8 No |
TABLE 5. NON-REACTION GROUP + PROBABLE REACTORS.
| N | SEX | AGE (years) |
VOL (ml) |
PELLET SIZE |
ALLOGRAFT MIX |
EtOH WASH |
SMOKER | MEDS | ALLERGIES |
|---|---|---|---|---|---|---|---|---|---|
| 50 | 24 M 26 F |
27.4 ±17.8 |
22.5 ±19.9 |
21 (3.3 mm) 21 (4.8 mm) 8 (mixed) |
17 Yes 33 No |
22 Yes 28 No |
7 Yes 43 No |
33 Yes 17 No |
18 Yes 32 No |
There was no statistically significant difference (p < 0.75) in the volume of Osteoset used between the reaction and non-reaction groups regardless of whether probable reactions were included with either group. Age was also statistically insignificant whether probable reactions were included with the reaction group (p < 0.91) or the non-reaction group (p < 0.33). Additionally, no statistically significant differences were found when comparing the two groups with respect to sex (p < 0.10 and p < 1.0 for reaction group + probable reactions and reaction group alone, respectively), pellet size (p < 0.20), use of alcohol washes (p < 1.0), mixing of Osteoset with allograft (p < 1.0), smoking history (p < 1.0), or reported drug allergy (p < 1.0). Reported use of at least one medication was statistically significant between groups when probable reactions were included with the nonreaction group (p < 0.05), but not when probables were analyzed with the reaction group ( p < 0.10). Furthermore, a review of our patients' medication lists did not reveal any single or combination of medications likely to be responsible for this difference. Type and location of tumor also did not appear to differ significantly between the two groups.
DISCUSSION
Calcium sulfate has been used as a bone graft substitute for over 100 years5 and has never been associated with unusual inflammatory or foreign body reactions6. However, its use in a purified, surgical-grade pellet is a relatively recent innovation. The process by which OsteoSet is manufactured produces a a-hemihydrate form with regular, prism-shaped crystals different from the irregularly shaped crystals found in traditional b-hemihydrate plaster of Paris7. This uniformity in crystalline shape and size affords predictable resorption rates4, but may lead to accelerated graft resorption and accumulation of calcium-rich fluid which we believe is responsible for the inflammatory response3. An alternative explanation proposed for the occasional serous drainage seen in response to OsteoSet pellets implicates an osmotic effect4. More studies are needed to further elucidate the exact mechanism involved in these adverse reactions.
Although we report a 13.8-19.0% incidence of adverse reaction to OsteoSet in our series, we are unable to identify any statistically significant factors that might predict the development of a reaction. Despite this incidence of reaction, we continue to use OsteoSet for the filling of defects after curettage of benign tumors. OsteoSet is beginning to see increased use in other subspecialties of Orthopaedic surgery within our department and has met with great success. The reaction is well known to us and we can now confidently report this to our patients before surgery along with the other risks of surgery. The reaction is essentially self limited and fairly benign requiring only anti-inflammatories and observation.
TABLE 3. NON-REACTION GROUP.
| N | SEX | AGE (years) |
VOL (ml) |
PELLET SIZE |
ALLOGRAFT MIX |
EtOH WASH |
SMOKER | MEDS | ALLERGIES |
|---|---|---|---|---|---|---|---|---|---|
| 47 | 21 M 26 F |
26.6 ±17.3 |
23.3 ±20.3 |
19 (3.3 mm) 20 (4.8 mm) 8 (mixed) |
16 Yes 31 No |
21 Yes 26 No |
6 Yes 41 No |
31 Yes 16 No |
17 Yes 30 No |
TABLE 4. REACTION GROUP.
| N | SEX | AGE (years) |
VOL (ml) |
PELLET SIZE |
ALLOGRAFT MIX |
EtOH WASH |
SMOKER | MEDS | ALLERGIES |
|---|---|---|---|---|---|---|---|---|---|
| 8 | 5 M 3 F |
21.0 ±12.8 |
25.0 ±17.6 |
6 (3.3 mm) 1 (4.8 mm) 1 (mixed) |
1 Yes 7 No |
4 Yes 4 No |
1 Yes 7 No |
2 Yes 6 No |
2 Yes 6 No |
Footnotes
Investigation performed at the Department of Orthopaedic Surgery University of Iowa Health Care, Iowa City, IA.
References
- 1.Damien CJ, Parsons JR. Bone graft and bone graft substitutes: a review of current technology and applications. Journal of Applied Biomaterials. 1991;2(3):187–203. doi: 10.1002/jab.770020307. [DOI] [PubMed] [Google Scholar]
- 2.Mirzayan R, Panossian V, Avedian R, Forrester DM, Menendez LR. The use of calcium sulfate in the treatment of benign bone lesions: a preliminary report. Journal of Bone and Joint Surgery. 2001;83-A(3):355–358. doi: 10.2106/00004623-200103000-00006. [DOI] [PubMed] [Google Scholar]
- 3.Robinson D, Alk D, Sandbank J, Farber R, Halperin N. Inflammatory reactions associated with a calcium sulfate bone substitute. Annals of Transplantation. 1999;4(3-4):91–97. [PubMed] [Google Scholar]
- 4.Kelly CM, Wilkins RM, Gitelis S, Hartjen C, Watson JT, Kim PT. The use of surgical grade calcium sulfate as a bone graft substitute: results of a multicenter trial. Clinical Orthopaedics and Related Research. 2001;382:42–50. doi: 10.1097/00003086-200101000-00008. [DOI] [PubMed] [Google Scholar]
- 5.Gitelis S, Piasecki P, Turner T, Haggard W, Charters J, Urban R. Use of calcium sulfate-based bone graft substitute for benign bone lesions. Orthopedics. 2001;24(2):162–166. doi: 10.3928/0147-7447-20010201-19. [DOI] [PubMed] [Google Scholar]
- 6.Peltier LF, Bickel EY, Lillo R, et al. The use of plaster of paris to fill defects in bone. Annals of Surgery. 1957;146:61–69. doi: 10.1097/00000658-195707000-00007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Pietrzak WS, Ronk R. Calcium sulfate bone void filler: a review and a look ahead. Journal of Craniofacial Surgery. 2000;11(4):327–333. doi: 10.1097/00001665-200011040-00009. [DOI] [PubMed] [Google Scholar]


