Abstract
Mutations in ras and p53 are the most prevalent mutations found in human nonmelanoma skin cancers. Although some p53 mutations cause a loss of function, most result in expression of altered forms of p53, which may exhibit gain-of-function properties. Therefore, understanding the consequences of acquiring p53 gain-of-function versus loss-of-function mutations is critical for the generation of effective therapies for tumors harboring p53 mutations. Here we describe an inducible mouse model in which skin tumor formation is initiated by activation of an endogenous K-rasG12D allele. Using this model we compared the consequences of activating the p53 gain-of-function mutation p53R172H and of deleting the p53 gene. Activation of the p53R172H allele resulted in increased skin tumor formation, accelerated tumor progression, and induction of metastasis compared with deletion of p53. Consistent with these observations, the p53R172H tumors exhibited aneuploidy associated with centrosome amplification, which may underlie the mechanism by which p53R172H exerts its oncogenic properties. These results clearly demonstrate that p53 gain-of-function mutations confer poorer prognosis than loss of p53 during skin carcinogenesis and have important implications for the future design of therapies for tumors that exhibit p53 gain-of-function mutations.
Introduction
Nonmelanoma skin cancer (NMSC) is the most common form of human cancer, with incidence rates dramatically rising during the last decade, perhaps as a result of increased sun exposure and the continuous depletion of the ozone layer (1, 2). It is widely accepted that squamous cell carcinomas (SCCs), which are responsible for the majority of NMSC-related deaths, result from the accumulation of genetic alterations (3). Understanding the role of these genetic lesions in the etiology of skin cancer is essential for designing improved therapies for cancer treatment and prevention.
Mutations in ras genes are found in approximately 10%–30% of human skin SCCs (4, 5), and mice subjected to chemical carcinogenesis protocols by initiation with dimethylbenz[a]anthracene (DMBA) and tumor promotion with 12-O-tetradecanoylphorbol-13-acetate (TPA) develop skin tumors that exhibit H-ras mutations in more than 90% of the cases (6). In addition, mice treated with the carcinogen N-methyl-N′-nitro-N-nitrosoguanidine and H-ras–null mice subjected to DMBA/TPA protocols develop skin tumors that carry oncogenic mutations in the K-ras gene, suggesting that mutations in K-ras can substitute for H-ras mutations in skin cancer initiation (7, 8). Several mouse models have previously documented that overexpression of oncogenic H-ras or K-ras in the skin can induce tumor formation (9–12). However, activation of ras genes associated with skin cancer initiation may not necessarily result in overexpression of the mutant ras allele, suggesting that mutation of endogenous ras in the mouse may better reproduce the initial event in human skin carcinogenesis.
More than 50% of human cutaneous SCCs carry mutations in the p53 gene, most of which are missense mutations that result in the change of a single amino acid and expression of altered forms of p53 (13, 14). Some of these p53 mutations can promote tumorigenicity when introduced into p53-null cells, suggesting that they acquire gain-of-function properties (15, 16). Several lines of evidence, including clinical observations, suggest that tumors containing putative p53 gain-of-function mutations may have a poorer prognosis than those with loss of p53 (17). In addition, 2 recently generated knockin mouse models provided evidence to suggest that mutant p53 may also exhibit gain-of-function properties in vivo (18, 19). We previously generated a transgenic mouse model that overexpressed p53R172H (equivalent to human p53R175H) in the epidermis and found that these mice exhibited accelerated chemical carcinogenesis (20). Since the p53R172H transgene was expressed at levels 4- to 8-fold higher than WT p53, it was unclear whether p53R172H functioned as a true gain-of-function mutation in the genetic sense.
Here we describe the generation of an inducible mouse model for skin cancer based on the focal activation of an endogenous K-rasG12D allele and gain- or loss-of-function mutations in p53. This model allowed us to compare the consequences of activating endogenous p53 gain-of-function versus loss-of-function mutations during skin cancer development. To our knowledge, this is the first study in which these mutations have been compared in a tissue-specific manner with a common initiating event (K-rasG12D). In this context, the mutant p53R172H allele clearly exhibits gain-of-function properties that contribute to skin cancer initiation and malignant progression.
Results
Activation of an endogenous K-rasG12D mutation initiates skin tumor formation.
To activate an endogenous oncogenic ras mutation in mouse skin, as occurs in human sporadic skin cancer, we generated mice in which the K-rasG12D mutation could be focally induced in the skin by crossing LSL–K-rasG12D mice with K5.Cre*PR1 mice (K5Cre*-Ras mice) (21, 22). The LSL–K-rasG12D mice carry an endogenous K-ras allele that contains the G12D mutation, a mutation frequently found in human tumors, and a stop cassette flanked by loxP sites (LSL) that keeps the LSL–K-rasG12D allele silent until the stop cassette is deleted by Cre recombinase (Figure 1B). The K5.Cre*PR1 mice express an inducible Cre recombinase under the control of the keratin 5 (K5) promoter. In these mice the inducible Cre is expressed in stratified epithelia including the skin and remains inactive until activation with RU486. Topical application of RU486 to the skin of K5Cre*-Ras mice activates Cre in the epidermis, which results in deletion of the stop cassette from the LSL–K-rasG12D allele and expression of endogenous K-rasG12D (Figure 1B).
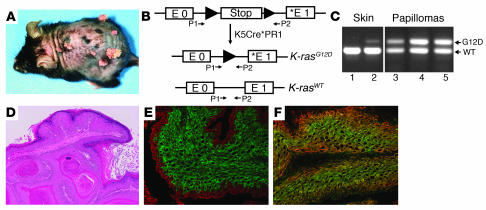
Figure 1. K-ras activation initiates skin tumor formation.
(A) Gross appearance of the epidermal papillomas that developed in the skin of RU486-treated K5Cre*-Ras mice 16 weeks after exposure to TPA. (B) Schematic representation of the LSL–K-rasG12D allele. The glycine–to–aspartic acid mutation (asterisk) in codon 12 is located in exon 1 (E1). Primers 1 and 2 (P1 and P2) were used for analysis of excision of the stop cassette. (C) Activation of the K-rasG12D allele in the skin (lanes 1 and 2) and papillomas (lanes 3, 4, and 5) of K5Cre*-Ras mice. Note that the K-rasG12D allele is only activated in RU486-treated skin (lane 2), but not in untreated mice (lane 1). (D) Hematoxylin and eosin staining of papillomas that developed in K5Cre*-Ras mice. (E and F) Keratin staining in papillomas: double immunofluorescence for K14 (red) and K13 (green) (E) and K14 (red) and K6 (green) (F) on frozen sections obtained from papillomas that developed in K5Cre*-Ras mice. Original magnification, ×40 (D), ×100 (E and F).
To determine whether endogenous activation of K-rasG12D can initiate skin tumor formation, we treated K5Cre*-Ras mice with a single topical application of RU486 (1 mg/ml) and weekly applications of the tumor promoter TPA. We found that K5Cre*-Ras mice developed skin tumors 7–10 weeks after tumor promotion (Figure 1A). Mice that were not treated with RU486 did not develop skin tumors, indicating that the inducible system was tightly regulated in vivo. PCR analysis with primers that flank the stop cassette in the LSL–K-rasG12D allele, using DNA purified from the skin of mice that were treated with RU486 and untreated mice, confirmed that the K-rasG12D allele was only activated in the RU486-treated skin, proving that activation of conditional alleles in this model only occurred after treatment with RU486 (Figure 1, B and C). The tumors that developed in K5Cre*-Ras mice exhibited similar levels of the activated mutant K-rasG12D and WT alleles, indicating that the tumors consisted primarily of epithelial cells that activated the mutant K-rasG12D allele (Figure 1C). The skin tumors that arose in the K5Cre*-Ras mice were benign papillomas histologically similar to those generated in classic 2-stage chemical carcinogenesis protocols using DMBA/TPA (Figure 1D). Analysis of keratin expression revealed that the K5Cre*-Ras papillomas expressed K14 in the entire epithelial component of the tumors, K13 in the most differentiated layers of the papillomas, and K6 in all the epithelial layers of the tumor (Figure 1, E and F), a keratin expression pattern similar to that observed in chemically induced papillomas (23, 24). Overall, these results demonstrate that activation of an endogenous oncogenic ras allele can initiate skin tumor formation. These results also document that the inducible Cre mice are ideal for generating mouse models for skin cancer and for testing the cooperativity of discrete genetic alterations detected in sporadic human cancers.
The p53R172H gain-of-function mutation contributes to skin cancer initiation and accelerates carcinoma formation.
To determine whether endogenous ras and p53 mutations cooperate during skin carcinogenesis, we used the inducible system to generate mice in which endogenous ras and p53 mutations could be activated in the skin. Since most p53 mutations that occur in human tumors, including NMSC, are missense mutations that may exhibit gain-of-function properties (25), it was important to determine whether endogenous p53 gain-of-function and loss-of-function mutations confer different skin cancer susceptibility. To this end, we generated mice in which the K-rasG12D and p53R172H alleles could be activated in the skin upon treatment with RU486 (K-ras–p53R172H/WT mice), by crossing K5Cre*-Ras mice with Neo-p53R172H mice, which carry a conditional p53R172H allele (18) (Figure 2A). To compare the effects of the p53R172H gain-of-function mutation with loss of p53, we also generated mice in which the K-rasG12D mutation and deletion of p53 could be induced in the skin (K-ras–p53f/WT mice) by crossing the K5Cre*-Ras mice with floxed p53 mice (p53f) (26) (Figure 2B). The breeding strategy followed to generate these mice allowed us to compare K-ras–p53R172H/WT and K-ras–p53f/WT littermates, thereby minimizing genetic background effects. We found that after treatment with TPA, the K-ras–p53R172H/WT mice developed 3-fold more tumors than K-ras–p53f/WT mice and mice carrying 2 WT p53 alleles (K-ras–p53WT/WT) (Figure 2C). These results indicate that the gain-of-function p53R172H mutation, but not a p53 loss-of-function mutation, cooperates with K-rasG12D in skin cancer initiation. In addition, we found that carcinoma development was dramatically accelerated in K-ras–p53R172H/WT mice compared with K-ras–p53f/WT and K-ras–p53WT/WT mice (Figure 2D). Thus, by 32 weeks, 90% of the K-ras–p53R172H/WT mice had developed skin carcinomas compared to 5% of K-ras–p53WT/WT littermates and 30% of the K-ras–p53f/WT mice. These data suggest that expression of an endogenous p53R172H allele dramatically accelerates malignant progression of skin tumors compared with loss of p53. Of note, carcinoma formation in K-ras–p53f/WT mice was accelerated compared with K-ras–p53WT/WT littermates, in agreement with the accelerated malignant progression observed in homozygous and heterozygous p53 knockout mice subjected to 2-stage chemical carcinogenesis protocols (27), thereby confirming that loss of p53 plays a role in skin cancer progression.
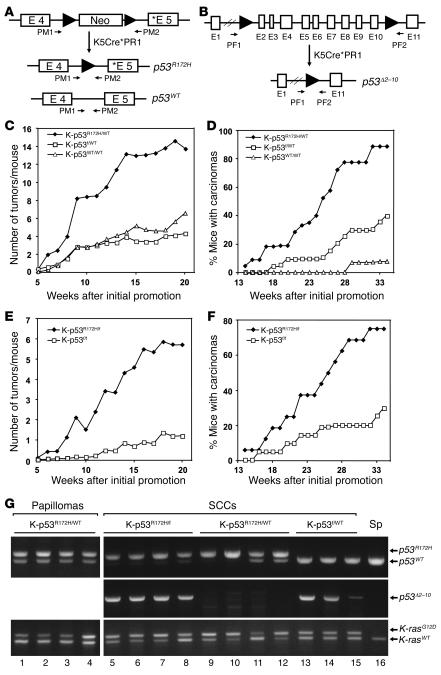
Figure 2. Activation of the p53R172H allele contributes to tumor initiation and malignant progression of skin tumors.
(A) Schematic representation of the neo-p53R172H allele showing exons 4 and 5, the presence of the neo cassette in intron 4, and primers R172H reverse and R172H forward (PM1 and PM2, respectively; see Methods), which were used for PCR analysis of the p53 allele. The neo cassette prevents expression of the p53R172H allele (C. Caulin, D.R. Roop, and G. Lozano, unpublished observations). Asterisks indicate mutation in exon 5 for p53R172H. (B) Schematic representation of the p53f allele showing loxP sites in introns 1 and 10 and primers PF1 and PF2 (26) that were used to analyze deletion of exons 2–10. (C) Kinetics of tumor formation in K-ras–p53R172H/WT mice (K-p53R172H/WT; n = 22) and K-ras–p53f/WT mice (K-p53f/WT; n = 21). K-ras–p53WT/WT mice (K-p53WT/WT; n = 17) were used as controls. Tumor formation was evaluated by the average number of tumors developed per mouse after the initial treatment with TPA. (D) Kinetics of carcinoma formation. Each time point represents percentage of mice bearing carcinomas. (E) Kinetics of tumor formation in K-ras–p53R172H/f mice (K-p53R172H/f; n = 16) and K-ras–p53f/f mice (K-p53f/f; n = 22). (F) Kinetics of carcinoma formation in K-ras–p53R172H/f and K-ras–p53f/f mice. (G) Activation of the p53R172H, p53f, and K-rasG12D alleles in papillomas and carcinomas. Primers PM1 and PM2 were used to analyze activation of the p53R172H allele. Primers PF1 and PF2 were used to analyze deletion of the loxP-flanked sequences in the p53f allele. Primers P1 and P2 were used to analyze activation of the K-rasG12D allele. DNA purified from spleen (Sp; lane 16) of a K-ras–p53R172H/WT mouse was used as a control and only generated the band corresponding to p53WT or K-rasWT alleles.
Since mutant p53 can bind and inactivate WT p53 (28), our results could be interpreted either as a consequence of the acquisition of gain-of-function properties of mutant p53R172H or due to dominant-negative effects of p53R172H toward WT p53. To address this question we generated mice that, in addition to carrying the K-rasG12D allele, also carry either 1 p53R172H allele and 1 p53f allele (K-ras–p53R172H/f mice) or 2 p53f alleles (K-ras–p53f/f mice). After activation of Cre in the skin by treatment with RU486, the K-ras–p53R172H/f mice express mutant p53R172H in the absence of WT p53. Therefore, a dominant-negative effect for p53R172H is ruled out in this model. Tumor formation in K-ras–p53R172H/f mice was compared with K-ras–p53f/f mice, which do not express p53 in Cre-activated cells. We observed that after treatment with TPA, the K-ras–p53R172H/f mice developed more tumors than K-ras–p53f/f mice (Figure 2E), and conversion to malignant carcinomas was also significantly accelerated in K-ras–p53R172H/f mice compared with K-ras–p53f/f mice (Figure 2F). Activation of the conditional alleles in the skin tumors that developed in these mice was confirmed by PCR using specific primers for the activated alleles (Figure 2G). These findings were supported by the detection of mutant p53 in the nuclei of skin tumors that developed in K-ras–p53R172H/f mice, primarily in the basal cells of the epithelial component of the tumors (Supplemental Figure 1; supplemental material available online with this article; doi:10.1172/JCI31721DS1), confirming that these tumors originated from epithelial cells that had activated the mutant p53R172H allele.
Expression of endogenous p53R172H results in formation of malignant skin carcinomas and metastasis.
Histological analysis revealed that 60% of the carcinomas that developed in K-ras–p53R172H mice were spindle cell carcinomas that consisted primarily of epithelial cells that showed little differentiation and large areas of spindle cells, which are the most undifferentiated and malignant cells in this type of tumor (29) (Figure 3A). In contrast, carcinomas that arose in K-ras–p53f/f mice were differentiated SCCs with abundant keratin pearls and an absence of spindle cells (Figure 3B).
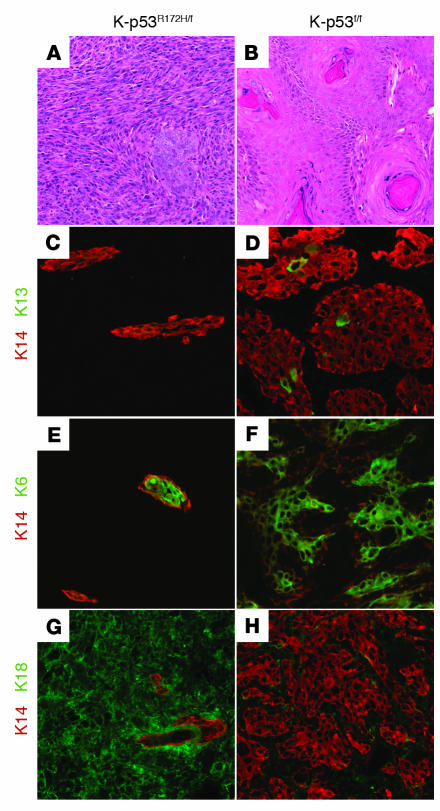
Figure 3. K-ras–p53R172H mice developed spindle cell carcinomas.
(A and B) Hematoxylin and eosin staining of skin carcinomas that developed in K-ras–p53R172H/f mice (A) and K-ras–p53f/f mice (B). (C–H) Keratin staining in carcinomas: double immunofluorescence for K14 (red) and K13 (green) (C and D), K14 (red) and K6 (green) (E and F), and K14 (red) and K18 (green) (G and H) on frozen sections obtained from carcinomas that developed in K-ras–p53R172H/f mice (C, E, and G) or K-ras–p53f/f mice (D, F, and H). Original magnification, ×100.
A more detailed characterization of tumor differentiation using antibodies specific for keratins revealed that K14 was only expressed in restricted areas of the K-ras–p53R172H/f carcinomas (Figure 3C). These K14-positive areas of the tumors failed to express K13, a molecular marker for early stages of skin carcinogenesis (30) (Figure 3C). In addition, these tumors induced expression of the simple epithelial K18, a keratin that is not expressed in normal epidermis but that has been associated with late stages of skin carcinogenesis (31). K18 was expressed in epithelial areas of the tumors that were negative for K14 (Figure 3G). These observations are in contrast with the widespread expression of K14 in the K-ras–p53f/f carcinomas, which also expressed K13 in the most differentiated areas of the tumor (Figure 3D). Moreover, the K-ras–p53f/f carcinomas showed complete absence of K18 (Figure 3H), again suggesting that these tumors represent earlier stages of progression compared with the K-ras–p53R172H/f carcinomas. The proliferation-associated K6 was expressed in both K-ras–p53R172H/f (Figure 3E) and K-ras–p53f/f carcinomas (Figure 3F). Overall, this analysis demonstrates that carcinomas that arise as a result of activation of an endogenous p53R172H gain-of-function mutation express keratin markers associated with malignant progression and present a more undifferentiated aggressive phenotype compared with tumors that arise as a result of loss of p53.
In support of these findings, we found that over 60% of the K-ras–p53R172H mice developed metastasis in lungs and/or lymph nodes (Table 1). This is a remarkable observation, as metastases are seldom observed in WT mice subjected to skin carcinogenesis protocols and were also absent in K-ras–p53f/f mice, which indicates that endogenous p53R172H promotes the metastatic potential of skin cancers. Histological examination demonstrated that the lungs and lymph nodes of mice with metastasis had been invaded by epithelial tumor cells (Figure 4, A–C). In addition, we confirmed that these metastases contained activated K-rasG12D and p53R172H alleles (Figure 4D). The presence of epithelial tumor cells in the metastases was further confirmed by staining with keratin antibodies, which revealed focal areas of cells that expressed K14, K6, and K18 (Figure 4, E and F). Although spindle cells, which originate by epithelial-to-mesenchymal transition, are considered to be the most malignant cells in skin cancers, our results suggest that cells that maintain expression of epithelial markers can also metastasize, as previously demonstrated in other mouse models (32). Overall, these results confirmed that the metastases originated from epithelial tumor cells that had activated the K-rasG12D and p53R172H alleles and documented that the p53R172H allele confers gain-of-function properties that accelerate malignant progression and metastasis of skin cancers.
Table 1 .
Mice that express p53R172H develop metastases in the lungs and/or lymph nodes
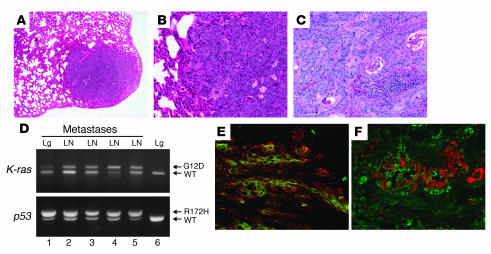
Figure 4. K-ras–p53R172H tumors metastasize to lungs and lymph nodes.
(A) Hematoxylin and eosin staining of a lung metastasis that developed in a K-ras–p53R172H/f mouse. (B) Higher magnification of the metastasis shown in A. (C) Hematoxylin and eosin staining of a lymph node metastasis that developed in a K-ras–p53R172H/f mouse. Note the massive presence of metastatic epithelial cells invading the organs. (D) Activation of the K-rasG12D and p53R172H alleles in lung (Lg) and lymph node (LN) metastases (lanes 1–5) using primers described in Figure 1B and Figure 2A. PCR analysis of DNA purified from lungs (lane 6) of a K-ras–p53R172H/WT mouse was used as control. (E and F) Double immunofluorescence for K14 (red) and K6 (green) (E) and K18 (green) and K14 (red) (F) on frozen sections obtained from lymph node metastases that developed in K-ras–p53R172H/f mice. Original magnification, ×40 (A), ×100 (B–C, E, and F).
The p53R172H tumors exhibit centrosome amplification and aneuploidy.
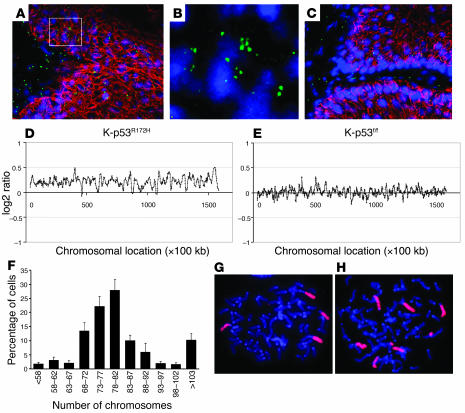
Previous mouse models have shown that skin and mammary tumors that overexpress p53R172H exhibit centrosome amplification (20, 33), a process that may result in genomic instability (34). To determine whether centrosome amplification also occurred in skin tumors that express the p53R172H allele, we stained frozen sections of skin papillomas with an antibody specific for centrosomes (35). This analysis detected supernumerary centrosomes in 3%–5% of the cells in K-ras–p53R172H/f tumors (Figure 5, A and B), but not in tumors that developed in K-ras–p53f/f mice (Figure 5C). Since centrosome amplification may promote tumor progression through mechanisms involving aneuploidy (34), we analyzed the genomic content of skin carcinomas that developed in these mice using comparative genomic hybridization on bacterial artificial chromosome (BAC-CGH) containing over 35,000 BAC clones, which cover more than 98% of the mouse genome. We found that 42% (8/19) of the K-ras–p53R172H carcinomas analyzed exhibited chromosome gains, compared with 23% (3/13) of the K-ras–p53f and 20% (1/5) of the K-ras–p53WT carcinomas, indicating that aneuploidy is more frequent in carcinomas that express the gain-of-function p53R172H than in tumors that lose p53 or that express WT p53. Interestingly, gains of chromosomes 15 (3/19) and 3 (3/19) were only observed in K-ras–p53R172H carcinomas (Figure 5, D and E), whereas gain of chromosome 6 was observed in tumors of all genotypes.
Figure 5. K-ras–p53R172H tumors exhibit centrosome amplification and aneuploidy.
(A–C) Double immunofluorescence for centrosomes (green) and K14 (red) in papillomas that developed in K-ras–p53R172H/f mice (A and B) and K-ras–p53f/f mice (C). Nuclei were stained with TO-PRO-3. Note the presence of cells with more than 2 centrosomes in K-ras–p53R172H/f tumors (A) but not in K-ras–p53f/f tumors (C). (B) Higher magnification of the area within the white square shown in A. The red staining for K14 was omitted to allow better visualization of multiple centrosomes in the cells. (D and E) BAC-CGH genomic profile for chromosome 3 in a K-ras–p53R172H carcinoma (D) and a K-ras–p53f/f carcinoma (E) obtained by plotting log2 ratios for each BAC clone (y axis) according to their chromosomal location (x axis). Note gains throughout the entire chromosome in the K-ras–p53R172H carcinoma compared with the K-ras–p53f/f carcinoma, which presents a mostly stable profile. (F) Chromosome counts in metaphase spreads obtained from 6 independent K-ras–p53R172H carcinomas. (G and H) FISH for chromosome 2 (G) and chromosome 6 (H) using whole-chromosome painting probes on metaphase spreads from K-ras–p53R172H carcinomas. Magnification, ×100 (A and C), ×1000 (G and H), ×2000 (B).
Because over 50% of the K-ras–p53R172H carcinomas analyzed did not show chromosome amplification in BAC-CGH arrays, we analyzed ploidy on metaphase spreads obtained from tumor cells cultured from 6 K-ras–p53R172H carcinomas. We found that most of the tumor cells contained 70–90 chromosomes, a near-tetraploid DNA content, which is difficult to detect by BAC-CGH arrays (Figure 5F). FISH analysis of the metaphase spreads using whole chromosome painting probes confirmed the presence of 4 copies of chromosome 2 (Figure 5G), a chromosome not found amplified in the BAC-CGH assays. In addition, multiple copies of chromosome 6 were detected in cells derived from tumors that showed chromosome 6 amplification by BAC-CGH (Figure 5H). These findings indicate that skin carcinomas that developed in these models displayed a near-tetraploid DNA content, with increased chromosome gains in tumors that expressed endogenous p53R172H. Overall, these results suggest that expression of the gain-of-function p53R172H allele in skin tumors predisposes to centrosome amplification, which may result in accelerated aneuploidy and malignant progression of skin carcinomas.
Discussion
Extensive evidence obtained from clinical studies suggests that mutations in ras and p53 genes are involved in human NMSC development. In addition, studies based on transgenic mouse models and chemical carcinogenesis experiments support a causal role for ras mutations in skin cancer initiation, whereas mutations in p53 may have a prominent role in later stages of malignant progression (36). Although these observations suggest that ras and p53 mutations may cooperate during skin carcinogenesis, prior to this study, in vivo models to assess cooperation between endogenous ras and p53 mutations had not, to our knowledge, been reported. Since most p53 mutations found in human cancers are gain-of-function rather than loss-of-function mutations, comparing the effects of gain-of-function and loss-of-function mutations was critical to understanding their contrasting roles during skin carcinogenesis. The inducible mouse model described here allowed us to demonstrate that endogenous ras and gain-of-function p53 mutations cooperate in skin cancer initiation, malignant progression, and metastasis.
Previously, several groups including our own have shown that overexpression of oncogenic ras in the skin can result in tumor formation (9–12). Since mutant ras was overexpressed in these mice, these models did not allow assessment of whether endogenous ras mutations, which occur in sporadic human cancers, are sufficient to initiate skin cancer formation. Skin papilloma formation was also reported in a subset of mice that expressed the K-rasG12D allele in the skin (37). However, the reason for the incomplete penetrance of the tumor phenotype in these mice was unclear. In this regard, we found that skin tumor formation in the K5Cre*-Ras mice only occurs after treatment with the tumor promoter TPA or in areas of the skin subjected to chronic injury, such as ear tags. Similar findings were also reported for mice that overexpressed oncogenic H-ras in the skin under the control of the K1 or K10 promoter (9, 11). Thus, it appears that a tumor-promoting stimulus is required for the development of skin tumors from epidermal cells initiated by oncogenic ras activation. These observations are in contrast with the spontaneous tumor formation observed upon activation of the K-rasG12D allele in other stratified epithelia such as the oral, anal, and vaginal epithelia (21, 38), which suggests the presence of intrinsic mechanisms that suppress tumor formation in the skin. Identifying such mechanisms may be useful for developing therapies for tumors that arise in tissues in which induction of K-ras mutations results in spontaneous tumor formation.
Ras mutations associated with skin cancers have been found primarily in H-ras and K-ras genes, although H-ras mutations seem to be more prevalent (4, 5). Similarly, DMBA preferentially induces H-ras mutations, whereas skin tumors that develop in mice treated with N-methyl-N′-nitro-N-nitrosoguanidine carry primarily K-ras mutations (8). These observations suggest that K-ras and H-ras mutations may play similar roles in skin cancer development, although these mutations may arise as a consequence of differential sensitivity to carcinogens. In this context, our model represents a genetically defined model that circumvents the use of carcinogens traditionally used in skin carcinogenesis, which induce ras mutations and perhaps other unidentified genetic alterations (7, 39–41).
The inducible system allowed us to analyze cooperation between ras and p53 mutations and to compare the consequences of activating endogenous p53 gain-of-function versus loss-of-function mutations in skin cancer development. This analysis demonstrated that activation of a mutant p53R172H allele, but not loss of p53, cooperates with K-rasG12D in skin cancer initiation. Interestingly, patches of cells that contain missense mutations in p53 are also found in sun-exposed normal human skin (42, 43). However, skin tumors very seldom arise from these mutant p53 cells, suggesting that additional mutations may be required for tumor development. Our observations indicate that the presence of p53 gain-of-function mutations in epidermal cells can facilitate skin tumor formation when additional genetic alterations such as ras mutations accumulate in these cells. Since tumors that have mutations in p53 do not always carry mutations in ras genes, it is tempting to speculate that p53 mutations may also cooperate with other genetic alterations to initiate skin cancer.
Mice subjected to chemical carcinogenesis protocols exhibit p53 mutations in SCCs, but not in benign papillomas, suggesting a role for p53 mutations in skin cancer progression (44, 45). Accordingly, p53-null mice treated with DMBA/TPA exhibit accelerated tumor progression, indicating that loss of p53 predisposes to malignant conversion of skin tumors (27, 46). In agreement with these findings, we also observed increased carcinoma formation after deletion of the p53 gene in the presence of the K-rasG12D allele. More striking, however, was our finding that mice that expressed the gain-of-function p53R172H mutation exhibited accelerated malignant progression and a much more aggressive tumor phenotype, including the ability to develop metastasis, compared with mice that exhibited loss of p53. These observations clearly indicate that the endogenous p53R172H mutation confers gain-of-function properties involved in malignant progression and metastasis of skin cancers and represent the strongest in vivo evidence to date for an oncogenic role for endogenous p53 missense mutations. Our findings are in agreement with previous reports showing that mice carrying knockin p53R172H or p53R270H alleles in the germline developed tumor spectra different from p53-null mice, with increased metastatic potential (18, 19). Unfortunately, an assessment of the role of gain-of-function p53 mutants in skin carcinogenesis was not possible in these initial reports, since mice homozygous for these mutant p53 alleles succumbed to a wide variety of internal tumors within 5 to 6 months. In the present study, we overcame this limitation of the constitutive knockin model by activating the conditional p53R172H allele in somatic epidermal cells. The use of this approach in other tissues may reveal the role of p53 gain-of-function mutations in other tumor types. In addition, since the oncogenic roles of different p53 mutations may differ (47), similar studies will be required to determine the impact of other potential p53 gain-of-function mutations in tumorigenesis.
The presence of supernumerary centrosomes in K-ras–p53R172H/f mice but not in K-ras–p53f/f mice may provide a mechanistic explanation for the oncogenic role of p53R172H during skin carcinogenesis. Abnormal centrosome amplification can lead to defective chromosome segregation, generally resulting in chromosome contents incompatible with cell viability. However, a small proportion of cells may acquire a growth advantage as a result of the gain of chromosomes that carry oncogenes or loss of chromosomes that carry tumor suppressor genes. In fact, we found a correlation between centrosome amplification and accelerated aneuploidy, which may contribute to the malignant properties of the tumors that developed in the K-ras–p53R172H mice. Similarly, genomic instability was also found in pancreatic tumors that developed in mice that express K-rasG12D and p53R172H (48), although it was unclear in that model whether genomic instability was the result of gain-of-function or loss of p53 activity. Despite substantial evidence to suggest that centrosome amplification may lead to genomic instability, we cannot exclude the possibility that centrosome abnormalities arise in tumors that express mutant p53 as a consequence of the more aggressive phenotype of these tumors.
There is a very interesting correlation between the results obtained with our mouse models and the types of p53 mutations found in human skin cancers. Notably, most of the p53 mutations found in human SCCs are point mutations that often coexist in the presence of the WT p53 allele, a mutation pattern expected for gain-of-function mutations (14). However, the more benign basal cell carcinomas primarily exhibit mutations that introduce stop codons, and both p53 alleles are frequently mutated or deleted (49). Furthermore, SCCs tend to be aneuploid, whereas basal cell carcinomas are largely diploid. Thus, our observations may provide a causal relation between p53 gain-of-function mutations and the overall more aggressive tumor phenotype found in human skin cancers that harbor this type of mutation.
Studies performed on osteosarcoma cells derived from the p53R172H germline mutant mice suggested that mutant p53 in these cells may exert its effects, at least in part, through binding and inactivation of the p53 family members p63 and p73 (18, 19). However, we did not detect binding of p53R172H to p63 or p73 in epidermal keratinocytes, nor did we find modulation of the p63 and p73 transactivation properties by p53R172H (Supplemental Figure 2). These results are not surprising considering that the predominant p63 and p73 isoforms expressed in the skin are the dominant-negative ΔN isoforms and considering the recent finding that TAp63 has tumor-promoting properties in the skin rather than a tumor suppressor role (50). In addition, Flores et al. have shown that mice heterozygous for either p53 and p63 or p53 and p73 develop broader tumor spectrum than p53+/– mice. However, skin tumors were not reported in this study (51), further suggesting that inactivation of p63 and p73 does not play a role in skin cancer development. This scenario suggests that mutant p53 could use different mechanisms to induce malignant properties in epidermal keratinocytes. In this regard, a recent report proposed that mutant p53 can bind and inactivate the NF-Y transcription factor, thereby expanding the spectrum of molecular mechanisms that may contribute to the oncogenic properties of mutant p53 (52). Although the role of the NF-Y complex in skin cancer is presently unknown, these findings suggest that mutant p53 may exert its oncogenic effects through a variety of molecular mechanisms that may function in a tissue-specific manner.
In summary, our study provides in vivo evidence documenting cooperation between endogenous ras and p53 mutations during skin carcinogenesis and clearly demonstrates an oncogenic role for the gain-of-function mutation p53R172H in skin cancer development. These observations have profound implications for the future design of antitumor therapies for tumors that carry putative gain-of-function mutations in p53, as these therapies should not only restore WT p53 function but also inactivate the gain-of-function properties of mutant p53.
Methods
Mouse models.
Generation of K5.Cre*PR1 and Neo-p53R172H (previously named Neo-p53515A) mice has been described (18, 21). LSL–K-rasG12D mice were provided by Tyler Jacks (Massachusetts Institute of Technology Center for Cancer Research, Cambridge, Massachusetts, USA) and David Tuveson (University of Pennsylvania School of Medicine, Philadelphia, Pennsylvania, USA) (22). Floxed p53 mice were provided by Anton Berns (Netherlands Cancer Institute, Amsterdam, The Netherlands) (26). The genetic backgrounds of the mouse lines were as follows: K5.Cre*PR1, C57BL/6; Neo-p53R172H, 129Sv/C57BL/6; LSL–K-rasG12D, 129Sv/C57BL/6; floxed p53, FVB/129Sv. K5.Cre*PR1 mice were crossed to LSL–K-rasG12D mice to generate K5.Cre*PR1/LSL–K-rasG12D (K5Cre*-Ras). Neo-p53R172H mice were crossed with floxed p53 to generate Neo-p53R172H/f mice. K5Cre*-Ras mice were crossed with Neo-p53R172H/f to generate K5.Cre*PR1/LSL–K-rasG12D/Neo-p53R172H/WT (K-ras–p53R172H/WT) and K5.Cre*PR1/LSL–K-rasG12D/p53f/WT (K-ras–p53f/WT) mice in a mixed genetic background (FVB/129Sv/C57BL/6). Additional matings between Neo-p53R172H/f and K-ras–p53f/WT mice were established to generate K-ras–p53R172H/f and K-ras–p53f/f mice. Excision of the stop cassette from the Neo-p53R172H allele was analyzed using the following primers: R172H reverse, GGAGCCAGGCCTAAGAGCAAGAATAAGTCA, and R172H forward, GACCCCTTCTCACCAAAAACAAAAACAGC. Analysis of excision of the stop cassette from the LSL–K-rasG12D allele and excision of the loxP-flanked sequences from the floxed p53 allele was performed as described in refs. 22 and 26. All experiments involving mice were approved by the Institutional Animal Care and Use Committee of Baylor College of Medicine.
Histological analysis and immunofluorescence.
For histology, biopsied tumors were fixed overnight in 10% neutral-buffered formalin at room temperature and then transferred to 75% ethanol, embedded in paraffin, sectioned, and stained with hematoxylin and eosin. For immunofluorescence, tumor biopsies were embedded in OCT compound and frozen at –70°C, and 5-μm sections were obtained and subjected to double-label immunofluorescence using the p53 antibody CM5p (Novacastra) and polyclonal antibodies for K14, K13, and K6 (53). OCT sections were fixed in methanol for 10 minutes for p53 detection and in 75% acetone/25% ethanol for 15 minutes for keratins, then blocked in 5% BSA and incubated overnight at room temperature with the primary antibody mixes indicated in Figure 1, C and F, Figure 3, C–H, and Figure 4, E and F. The sections were then washed with PBS and incubated with secondary antibodies conjugated to Alexa Fluor 488 or 594 dyes (Invitrogen) for 45 minutes at room temperature. Images from stained sections were acquired using a Nikon Eclipse E600 microscope and MetaVue Meta Imaging Series 6.2 software (Molecular Devices) and processed with Adobe Photoshop 6.0.
Confocal imaging and scoring of centrosomes.
OCT-embedded tumors were sectioned at 50 μm using a minitome cryostat. The thick frozen sections were mounted onto positively charged slides, fixed, and stained for centrosomes as described previously (35). Sections were costained with the anti-K14 antibody and TO-PRO-3 iodide (Invitrogen). Immunofluorescence microscopy was performed with a confocal microscope (Zeiss LSM 510). Scoring was accomplished by counting the number of stained nuclei and the corresponding number of centrosomes in tissue sections (35). A minimum of 300 nuclei/sample were counted.
BAC-CGH.
Genome-wide analysis of skin carcinomas was performed as previously described (54) using BAC microarrays containing 35,000 BAC clones.
Metaphase spreads and FISH.
Tumor cells growing in 100-mm plates were incubated with 2.5 μg/ml colchicine for 2 hours. After washing with PBS, the cells were detached with trypsin, resuspended in 5 ml medium, and centrifuged at 200 g for 5 minutes. The pellet was washed with PBS, resuspended in 0.056 M KCl, and left at room temperature for 30 minutes. After spinning at 200 g for 5 minutes, the pellet was resuspended in 75% methanol/25% acetic acid, a process that was repeated 2 more times. The cell suspension was spread in drops onto a cold and wet slide, allowed to dry at room temperature, and mounted using VECTASHIELD Mounting Media with DAPI (Vector Laboratories). For FISH analysis, metaphase spreads prepared as described above were hybridized with biotinylated whole-chromosome painting probes (Open Biosystems) for mouse chromosomes 6 and 2 according to the manufacturer’s instructions. Chromosomes were visualized by incubation with Texas Red Streptavidin (Vector Laboratories). Images were acquired using a Nikon Eclipse E600 microscope and MetaVue Meta Imaging Series 6.2 software (Molecular Devices).
Supplementary Material
Acknowledgments
C. Caulin was supported by a Research Career Development Award from the Dermatology Foundation. This work was supported by NIH grants CA52607 to D.R. Roop, DE015344 to C. Caulin, CA105491 to D.R. Roop and C. Caulin, CA41424 to B.R. Brinkley, and CA64255 to B.R. Brinkley and T.M. Goepfert.
Footnotes
Nonstandard abbreviations used: BAC, bacterial artificial chromosome; BAC-CGH, comparative genomic hybridization on BAC; DMBA, dimethylbenz[a]anthracene; K5, keratin 5; NMSC, nonmelanoma skin cancer; SCC, squamous cell carcinoma; TPA, 12-O-tetradecanoylphorbol-13-acetate.
Conflict of interest: The authors have declared that no conflict of interest exists.
Citation for this article: J. Clin. Invest. 117:1893–1901 (2007). doi:10.1172/JCI31721
See the related Commentary beginning on page 1752.
References
- 1.Alam M., Ratner D. Cutaneous squamous-cell carcinoma. N. Engl. J. Med. 2001;344:975–983. doi: 10.1056/NEJM200103293441306. [DOI] [PubMed] [Google Scholar]
- 2.Albert M.R., Weinstock M.A. Keratinocyte carcinoma. CA Cancer J. Clin. 2003;53:292–302. doi: 10.3322/canjclin.53.5.292. [DOI] [PubMed] [Google Scholar]
- 3.Boukamp P. Non-melanoma skin cancer: what drives tumor development and progression? Carcinogenesis. 2005;26:1657–1667. doi: 10.1093/carcin/bgi123. [DOI] [PubMed] [Google Scholar]
- 4.Pierceall W.E., Goldberg L.H., Tainsky M.A., Mukhopadhyay T., Ananthaswamy H.N. Ras gene mutation and amplification in human nonmelanoma skin cancers. Mol. Carcinog. 1991;4:196–202. doi: 10.1002/mc.2940040306. [DOI] [PubMed] [Google Scholar]
- 5.Spencer J.M., Kahn S.M., Jiang W., DeLeo V.A., Weinstein I.B. Activated ras genes occur in human actinic keratoses, premalignant precursors to squamous cell carcinomas. Arch. Dermatol. 1995;131:796–800. [PubMed] [Google Scholar]
- 6.Balmain A., Brown K. Oncogene activation in chemical carcinogenesis. Adv. Cancer Res. 1988;51:147–182. doi: 10.1016/s0065-230x(08)60222-5. [DOI] [PubMed] [Google Scholar]
- 7.Ise K., et al. Targeted deletion of the H-ras gene decreases tumor formation in mouse skin carcinogenesis. Oncogene. 2000;19:2951–2956. doi: 10.1038/sj.onc.1203600. [DOI] [PubMed] [Google Scholar]
- 8.Rehman I., et al. Frequent codon 12 Ki-ras mutations in mouse skin tumors initiated by N-methyl-N’-nitro-N-nitrosoguanidine and promoted by mezerein. Mol. Carcinog. 2000;27:298–307. [PubMed] [Google Scholar]
- 9.Bailleul B., et al. Skin hyperkeratosis and papilloma formation in transgenic mice expressing a ras oncogene from a suprabasal keratin promoter. Cell. 1990;62:697–708. doi: 10.1016/0092-8674(90)90115-u. [DOI] [PubMed] [Google Scholar]
- 10.Brown K., Strathdee D., Bryson S., Lambie W., Balmain A. The malignant capacity of skin tumours induced by expression of a mutant H-ras transgene depends on the cell type targeted. Curr. Biol. 1998;8:516–524. doi: 10.1016/s0960-9822(98)70203-9. [DOI] [PubMed] [Google Scholar]
- 11.Greenhalgh D.A., et al. Induction of epidermal hyperplasia, hyperkeratosis, and papillomas in transgenic mice by a targeted v-Ha-ras oncogene. Mol. Carcinog. 1993;7:99–110. doi: 10.1002/mc.2940070208. [DOI] [PubMed] [Google Scholar]
- 12.Vitale-Cross L., Amornphimoltham P., Fisher G., Molinolo A.A., Gutkind J.S. Conditional expression of K-ras in an epithelial compartment that includes the stem cells is sufficient to promote squamous cell carcinogenesis. Cancer Res. 2004;64:8804–8807. doi: 10.1158/0008-5472.CAN-04-2623. [DOI] [PubMed] [Google Scholar]
- 13.Bolshakov S., et al. p53 mutations in human aggressive and nonaggressive basal and squamous cell carcinomas. Clin. Cancer Res. 2003;9:228–234. [PubMed] [Google Scholar]
- 14.Brash D.E., et al. A role for sunlight in skin cancer: UV-induced p53 mutations in squamous cell carcinoma. Proc. Natl. Acad. Sci. U. S. A. 1991;88:10124–10128. doi: 10.1073/pnas.88.22.10124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dittmer D., et al. Gain of function mutations in p53. Nat. Genet. 1993;4:42–46. doi: 10.1038/ng0593-42. [DOI] [PubMed] [Google Scholar]
- 16.Sun Y., Nakamura K., Wendel E., Colburn N. Progression toward tumor cell phenotype is enhanced by overexpression of a mutant p53 tumor-suppressor gene isolated from nasopharyngeal carcinoma. Proc. Natl. Acad. Sci. U. S. A. 1993;90:2827–2831. doi: 10.1073/pnas.90.7.2827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cadwell C., Zambetti G.P. The effects of wild-type p53 tumor suppressor activity and mutant p53 gain-of-function on cell growth. Gene. 2001;277:15–30. doi: 10.1016/s0378-1119(01)00696-5. [DOI] [PubMed] [Google Scholar]
- 18.Lang G.A., et al. Gain of function of a p53 hot spot mutation in a mouse model of Li-Fraumeni syndrome. Cell. 2004;119:861–872. doi: 10.1016/j.cell.2004.11.006. [DOI] [PubMed] [Google Scholar]
- 19.Olive K.P., et al. Mutant p53 gain of function in two mouse models of Li-Fraumeni syndrome. Cell. 2004;119:847–860. doi: 10.1016/j.cell.2004.11.004. [DOI] [PubMed] [Google Scholar]
- 20.Wang X.J., et al. Expression of a p53 mutant in the epidermis of transgenic mice accelerates chemical carcinogenesis. Oncogene. 1998;17:35–45. doi: 10.1038/sj.onc.1201890. [DOI] [PubMed] [Google Scholar]
- 21.Caulin C., et al. Inducible activation of oncogenic K-ras results in tumor formation in the oral cavity. Cancer Res. 2004;64:5054–5058. doi: 10.1158/0008-5472.CAN-04-1488. [DOI] [PubMed] [Google Scholar]
- 22.Jackson E.L., et al. Analysis of lung tumor initiation and progression using conditional expression of oncogenic K-ras. Genes Dev. 2001;15:3243–3248. doi: 10.1101/gad.943001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Roop D.R., et al. Keratin expression in mouse epidermal tumors. Carcinog. Compr. Surv. 1989;11:257–271. [PubMed] [Google Scholar]
- 24.Gimenez-Conti I., et al. Early expression of type I K13 keratin in the progression of mouse skin papillomas. Carcinogenesis. 1990;11:1995–1999. doi: 10.1093/carcin/11.11.1995. [DOI] [PubMed] [Google Scholar]
- 25.Ko L.J., Prives C. p53: puzzle and paradigm. Genes Dev. 1996;10:1054–1072. doi: 10.1101/gad.10.9.1054. [DOI] [PubMed] [Google Scholar]
- 26.Jonkers J., et al. Synergistic tumor suppressor activity of BRCA2 and p53 in a conditional mouse model for breast cancer. Nat. Genet. 2001;29:418–425. doi: 10.1038/ng747. [DOI] [PubMed] [Google Scholar]
- 27.Kemp C.J., Donehower L.A., Bradley A., Balmain A. Reduction of p53 gene dosage does not increase initiation or promotion but enhances malignant progression of chemically induced skin tumors. Cell. 1993;74:813–822. doi: 10.1016/0092-8674(93)90461-x. [DOI] [PubMed] [Google Scholar]
- 28.de Vries A., et al. Targeted point mutations of p53 lead to dominant-negative inhibition of wild-type p53 function. Proc. Natl. Acad. Sci. U. S. A. 2002;99:2948–2953. doi: 10.1073/pnas.052713099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Klein-Szanto A.J. Pathology of human and experimental skin tumors. Carcinog. Compr. Surv. 1989;11:19–53. [PubMed] [Google Scholar]
- 30.Nischt R., et al. Aberrant expression during two-stage mouse skin carcinogenesis of a type I 47-kDa keratin, K13, normally associated with terminal differentiation of internal stratified epithelia. Mol.Carcinog. 1988;1:96–108. doi: 10.1002/mc.2940010205. [DOI] [PubMed] [Google Scholar]
- 31.Caulin C., Bauluz C., Gandarillas A., Cano A., Quintanilla M. Changes in keratin expression during malignant progression of transformed mouse epidermal keratinocytes. Exp. Cell Res. 1993;204:11–21. doi: 10.1006/excr.1993.1003. [DOI] [PubMed] [Google Scholar]
- 32.Han G., et al. Distinct mechanisms of TGF-beta1–mediated epithelial-to-mesenchymal transition and metastasis during skin carcinogenesis. J. Clin. Invest. 2005;115:1714–1723. doi: 10.1172/JCI24399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Murphy K.L., Dennis A.P., Rosen J.M. A gain of function p53 mutant promotes both genomic instability and cell survival in a novel p53-null mammary epithelial cell model. FASEB J. 2000;14:2291–2302. doi: 10.1096/fj.00-0128com. [DOI] [PubMed] [Google Scholar]
- 34.Brinkley B.R. Managing the centrosome numbers game: from chaos to stability in cancer cell division. Trends Cell Biol. 2001;11:18–21. doi: 10.1016/s0962-8924(00)01872-9. [DOI] [PubMed] [Google Scholar]
- 35.Goepfert T.M., et al. Centrosome amplification and overexpression of aurora A are early events in rat mammary carcinogenesis. Cancer Res. 2002;62:4115–4122. [PubMed] [Google Scholar]
- 36.Dlugosz A., Merlino G., Yuspa S.H. Progress in cutaneous cancer research. J. Investig. Dermatol. Symp. Proc. 2002;7:17–26. doi: 10.1046/j.1523-1747.2002.19631.x. [DOI] [PubMed] [Google Scholar]
- 37.Tuveson D.A., et al. Endogenous oncogenic K-ras(G12D) stimulates proliferation and widespread neoplastic and developmental defects. Cancer Cell. 2004;5:375–387. doi: 10.1016/s1535-6108(04)00085-6. [DOI] [PubMed] [Google Scholar]
- 38.Chan I.T., et al. Conditional expression of oncogenic K-ras from its endogenous promoter induces a myeloproliferative disease. J. Clin. Invest. 2004;113:528–538. doi: 10.1172/JCI200420476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Mao J.H., et al. Mutually exclusive mutations of the Pten and ras pathways in skin tumor progression. Genes Dev. 2004;18:1800–1805. doi: 10.1101/gad.1213804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Owens D.M., Spalding J.W., Tennant R.W., Smart R.C. Genetic alterations cooperate with v-Ha-ras to accelerate multistage carcinogenesis in TG.AC transgenic mouse skin. Cancer Res. 1995;55:3171–3178. [PubMed] [Google Scholar]
- 41.Quintanilla M., Brown K., Ramsden M., Balmain A. Carcinogen-specific mutation and amplification of Ha-ras during mouse skin carcinogenesis. Nature. 1986;322:78–80. doi: 10.1038/322078a0. [DOI] [PubMed] [Google Scholar]
- 42.Nakazawa H., et al. UV and skin cancer: specific p53 gene mutation in normal skin as a biologically relevant exposure measurement. Proc. Natl. Acad. Sci. U. S. A. 1994;91:360–364. doi: 10.1073/pnas.91.1.360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Jonason A.S., et al. Frequent clones of p53-mutated keratinocytes in normal human skin. Proc. Natl. Acad. Sci. U. S. A. 1996;93:14025–14029. doi: 10.1073/pnas.93.24.14025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Burns P.A., et al. Loss of heterozygosity and mutational alterations of the p53 gene in skin tumours of interspecific hybrid mice. Oncogene. 1991;6:2363–2369. [PubMed] [Google Scholar]
- 45.Ruggeri B., et al. Alterations of the p53 tumor suppressor gene during mouse skin tumor progression. Cancer Res. 1991;51:6615–6621. [PubMed] [Google Scholar]
- 46.Kelly-Spratt K.S., Gurley K.E., Yasui Y., Kemp C.J. p19Arf suppresses growth, progression, and metastasis of Hras-driven carcinomas through p53-dependent and -independent pathways. PLoS Biol. 2004;2:E242. doi: 10.1371/journal.pbio.0020242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Soussi T., Lozano G. p53 mutation heterogeneity in cancer. Biochem. Biophys. Res. Commun. 2005;331:834–842. doi: 10.1016/j.bbrc.2005.03.190. [DOI] [PubMed] [Google Scholar]
- 48.Hingorani S.R., et al. Trp53R172H and KrasG12D cooperate to promote chromosomal instability and widely metastatic pancreatic ductal adenocarcinoma in mice. Cancer Cell. 2005;7:469–483. doi: 10.1016/j.ccr.2005.04.023. [DOI] [PubMed] [Google Scholar]
- 49.Ziegler A., et al. Mutation hotspots due to sunlight in the p53 gene of nonmelanoma skin cancers. Proc. Natl. Acad. Sci. U. S. A. 1993;90:4216–4220. doi: 10.1073/pnas.90.9.4216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Koster M.I., Lu S.L., White L.D., Wang X.J., Roop D.R. Reactivation of developmentally expressed p63 isoforms predisposes to tumor development and progression. Cancer Res. 2006;66:3981–3986. doi: 10.1158/0008-5472.CAN-06-0027. [DOI] [PubMed] [Google Scholar]
- 51.Flores E.R., et al. Tumor predisposition in mice mutant for p63 and p73: evidence for broader tumor suppressor functions for the p53 family. Cancer Cell. 2005;7:363–373. doi: 10.1016/j.ccr.2005.02.019. [DOI] [PubMed] [Google Scholar]
- 52.Di Agostino S., et al. Gain of function of mutant p53: the mutant p53/NF-Y protein complex reveals an aberrant transcriptional mechanism of cell cycle regulation. Cancer Cell. 2006;10:191–202. doi: 10.1016/j.ccr.2006.08.013. [DOI] [PubMed] [Google Scholar]
- 53.Roop D.R., et al. Synthetic peptides corresponding to keratin subunits elicit highly specific antibodies. J. Biol. Chem. 1984;259:8037–8040. [PubMed] [Google Scholar]
- 54.Mao J.H., et al. Genomic instability in radiation-induced mouse lymphoma from p53 heterozygous mice. Oncogene. 2005;24:7924–7934.. doi: 10.1038/sj.onc.1208926. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.