Abstract
Objective
To study the phenomenology, clinical correlates, and response to treatment of delirium in critically ill children in the pediatric intensive care unit (PICU).
Design, setting and patients
Descriptive study of a cohort of child psychiatric consultations from a tertiary PICU between January 2002 and December 2005. Demographic data, clinical presentation, and response to treatment of children subsequently diagnosed with delirium were analyzed.
Results
Out of 877 admissions (age distribution 0–18 years) arose 61 requests for psychiatric assessment. Of the 61 children, 40 (15 girls and 25 boys) were diagnosed with delirium (cumulative incidence 5%; mean age 7.6 years). Age-specific incidence rates varied from 3% (0–3 years) to 19% (16–18 years). In addition to the classical hypoactive and hyperactive presentations, a third presentation was apparent, characterized mainly by anxiety, with a higher prevalence in boys. All but 2 of the 40 children received antipsychotic medication: 27 (68%) haloperidol, 10 (25%) risperidone, and 1 both in succession. Two children treated with haloperidol experienced an acute torticollis as side effect. All children made a complete recovery from the delirium; five, however, died of their underlying disease.
Conclusion
The rate of delirium in critically ill children on a PICU is not negligible, yet prospective studies of the phenomenology, risk factors and treatment of childhood delirium are very rare. Once pediatric delirium has been recognized, it generally responds well to treatment.
Keywords: Delirium, Critically ill children, Pediatric intensive care unit (PICU), Consensus meetings, Haloperidol, Risperidone
Introduction
Delirium is a neuropsychiatric disorder secondary to a general medical condition, and must be considered a serious complication of the underlying disease or its treatment. In the revised fourth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-IV-TR®), delirium is defined by four concurrent diagnostic criteria: (1) acute onset and fluctuation of (2) a disturbance of consciousness with reduced ability to focus, shift or maintain attention and (3) a change of cognition with memory deficit, disorientation, language disturbance, perceptual disturbances or hallucinations, (4) caused by the direct physiological consequences of a general medical condition [1]. It is frequently seen in critically ill adult and geriatric patients [2–5] and is associated with a poor prognosis, reflected by longer hospital stay, worse functional and cognitive outcome, and a higher mortality rate after discharge from hospital [3]. In mechanically ventilated critically ill adults, delirium is an independent predictor of elevated 6-month mortality and a longer hospital stay [6]. If appropriate diagnostic tools validated for bedside use by non-psychiatrists [e.g. Delirium Rating Scale (DRS), Confusion Assessment Instrument for the Intensive Care Unit (CAM-ICU)] are used, delirium is diagnosed in over 80% of critically ill adult patients [7]. Thus, systematic monitoring for delirium and appropriate treatment with haloperidol in critically ill adult patients were included in the recently published clinical practice guidelines for sedatives and analgesia of the Society of Critical Care Medicine [8]. However, the optimal management of patients with delirium and the effects of the pharmacological treatment on the outcome are still key concerns for today [9]. Given lack of age-appropriate diagnostic criteria and assessment tools in children, even less is known about the incidence, clinical presentation, response to treatment and consequences of childhood delirium in general, and in critically ill children in particular [10–12]. The few available published data on childhood delirium suggest that morbidity and mortality are higher in children with than in children without delirium [13]. Therefore, delirium in children should be considered a serious complication and be treated accordingly. Unfortunately, while there are comprehensive guidelines on the diagnosis and treatment of delirium in adults, clinical guidelines for delirium in children are nonexistent.
The aim of this study was to investigate the incidence, patient and population characteristics, clinical presentation and response to treatment of delirium in a cohort of critically ill children admitted to a tertiary pediatric intensive care unit (PICU). Given the necessarily multidisciplinary approach to assessment and treatment of these children, the input of four disciplines – child psychiatry, pediatric intensive care medicine, child neurology and adult neuropsychiatry – was used.
Methods
Design, setting and patients
A descriptive study was carried out over a 4-year period (January 2002 to December 2005) in an eight-bed tertiary PICU. This PICU is a tertiary referral center for both general and surgically critically ill children in the southeastern region of the Netherlands (population 1.4 million, 350 annual admissions). Critically ill children, acutely, non-electively and consecutively admitted, were prospectively sampled. Both mechanically ventilated and non-ventilated patients were included.
Diagnostic approach
All children with (1) confusion, agitation, anxiety, moaning, discomfort, or behavioral disturbances with no acceptable medical explanation or (2) failure of standard analgosedative treatment were systematically assessed for the presence of delirium in a two-step diagnostic approach. The standard analgosedative treatment can be summarized as follows: children who required analgosedation because of obvious or expected pain or because of stress related to their underlying disease or treatment received adequate doses of opioids and/or benzodiazepines according to internationally published guidelines for analgesia and sedation in critically ill children [14]. Drug doses were individually tailored to achieve optimal patient comfort and were slowly reduced in order to avoid a withdrawal syndrome. In the event that a withdrawal syndrome was suspected, based on clinical observation or the revised Finnegan score, specific treatment with long-acting benzodiazepines (e.g. lorazepam) or opioids (e.g. methadone) was started according to internationally published guidelines [15, 16].
At the time the assessment for delirium was initiated, none of the patients had signs of imminent life-threatening respiratory, circulatory or neurological failure, while ongoing asphyxia, respiratory acidosis, metabolic disturbances, fighting the ventilator due to inappropriate ventilator settings and withdrawal syndrome all had been excluded systematically as an explanation for the observed behavior.
The first step of the diagnostic approach was a systematic assessment by a child neuropsychiatrist (J.S.) using DSM-IV criteria for delirium. Criteria were evaluated on the basis of (1) hetero-anamnestic information from parents, nurses, intensivists, and child neurologists about behavior and behavioral changes and (2) child psychiatric examination. Based on the findings, patients were categorized as having a (probable) delirium or not. In a second step, the provisional diagnosis of delirium was further tested in a daily multidisciplinary consensus meeting. The team consisted of the child neuropsychiatrist, the attending pediatric intensivist, and occasionally a geriatric neuropsychiatrist specialized in delirium in geriatric patients and/or a child neurologist. If this team agreed that alternative explanations for a child's behavior were unlikely, the consensus diagnosis was delirium.
Based on the dominant clinical presentation, cases of delirium were classified as “hyperactive” when psychomotor agitation was present, and as “hypoactive” when retardation and/or inhibition was present. A number of children presented with cognitive and/or attentional disturbances in the context of severe anxiety states, often accompanied with moaning and restlessness, but without clear agitation or retardation. This latter group was classified as “emerging” or “veiled” delirium [17], in reference to the way delirious syndromes have been described as “partial delirium” in adult ICU patients or as “subsyndromal delirium” in elderly medical patients [7, 18, 19]. The different presentation forms were not always clear-cut, and some fluctuated dramatically over time. The severity of illness was scored according to the Pediatric Index of Mortality (PIM) and Pediatric Risk of Mortality (PRISM) [20].
Therapeutic approach
Whenever delirium was identified or suspected, a two-track treatment approach consisting of both psychosocial and pharmacological interventions was implemented. Psychosocial interventions – the parents' presence and comforting throughout the day (and night), familiar music, favorite toys, pictures of home and pets, friends, school, lighting schedules, sometimes even fragrances – are standard in the PICU. The parents also received an information leaflet on childhood delirium [21]. All patients were also treated with antipsychotic medication after the referring pediatric intensivist had agreed and the parents, because of the off-label use, had given informed consent, which was never refused. In children with psychomotor agitation that was acutely threatening to their health status, haloperidol at a loading dosage of 0.15–0.25 mg i.v. was used, given slowly over a period of 30–45 min, followed by a maintenance dose of 0.05–0.5 mg/kg/24 h i.v. [11, 22, 23]. In less acute situations, and when oral medication was possible, risperidone at a loading dose of 0.1–0.2 mg p.o. was used, followed by a maintenance dose of 0.2–2.0 mg/24 h p.o. as the treatment of choice. Clinical response and side effects were recorded by the child neuropsychiatrist and the pediatric intensivists. In order to tailor the treatment for delirium, daily discussions were held with the multidisciplinary team. Adjustment of treatment was based on the clinical observations and judgements of the parents, nurses, intensivists and child psychiatrist.
Children were followed up for 6 weeks after discharge from the hospital either at the outpatient clinic or by contacting the parents by telephone.
As the study solely involved the structured recording of routine clinical practice, under Dutch law no institutional review board approval was required.
Results
From January 2002 to December 2005, there were 877 acute, non-elective admissions to the PICU. Distribution of age and gender are shown in Table 1. In 61 cases (7%), a systematic assessment by a child neuropsychiatrist was requested, usually for agitation, anxiety, moaning, discomfort, behavioral disturbance or problematic analgosedation.
Table 1.
Number and incidence of delirium in the total sample* by age and gender
| Age | Total sample* | Patients with delirium | Incidence (%) |
|---|---|---|---|
| 0–2.99 years | 513 | 14 | 2.7 |
| Male | 310 | 9 | 2.9 |
| Female | 203 | 5 | 2.5 |
| 3–5.99 years | 106 | 4 | 3.8 |
| Male | 56 | 3 | 5.4 |
| Female | 50 | 1 | 2.0 |
| 6–8.99 years | 80 | 6 | 7.5 |
| Male | 46 | 1 | 2.2 |
| Female | 34 | 5 | 14.7 |
| 9–11.99 years | 77 | 3 | 3.9 |
| Male | 61 | 3 | 4.9 |
| Female | 16 | 0 | 0 |
| 12–14.99 years | 70 | 7 | 10 |
| Male | 35 | 5 | 14.3 |
| Female | 35 | 2 | 5.7 |
| 15–18 years | 31 | 6 | 19.4 |
| Male | 13 | 4 | 30.8 |
| Female | 18 | 2 | 11.1 |
| Total | 877 | 40 | 4.6 |
| Male | 521 | 25 | 4.8 |
| Female | 356 | 15 | 4.2 |
*Critically ill children, acutely, non-electively and consecutively admitted during a 4-year period
Of these 61 patients, 40 (61%) were diagnosed with delirium, yielding a cumulative incidence of 5% (boys 5%; girls 4%). Age-specific incidences increased from 3% in the 0–3 years age group (boys 3%; girls 3%), to 19% in the 16–18 years age group (boys 31%; girls 11%) (Table 1, Fig. 1).
Fig. 1.
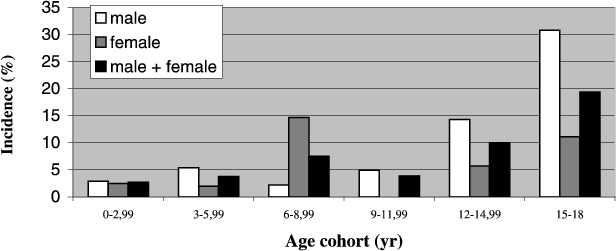
Incidence of delirium in the sub-groups by age and gender
The child psychiatric disorders diagnosed in the 61 referrals are summarized in Table 2.
Table 2.
Child psychiatric diagnosis at the first consultation (n = 61)
| n | Diagnosis |
|---|---|
| 40 | delirium |
| 5 | adjustment disorders with anxiety and depressed mood, post operative |
| 4 | psychological–psychiatric factors affecting the medical condition |
| 3 | anxiety disorder |
| 3 | emotional and behavioral problems during chronic ventilation |
| 2 | adjustment disorders with depressed mood |
| 1 | mood disorder |
| 1 | adjustment disorder with anxiety |
| 1 | sleeping problem |
| 1 | feeding problem |
Table 3 summarizes the population characteristics of the sample diagnosed with delirium, while Table 4 lists the underlying somatic disease or causative pharmacological treatment.
Table 3.
Population characteristics of the 40 PICU cases with delirium, 2002–2005
| Characteristics | Frequency (total n = 40) |
|---|---|
| Age (mean ± SD) | 7.6 ± 5.9 |
| Male | |
| n | 25 |
| age (mean ± SD) | 7.6 ± 6.1 |
| Female | |
| n | 15 |
| age (mean ± SD) | 7.6 ± 5.8 |
| Ethnicity | |
| White | 36 (90%) |
| African | 3 (7.5%) |
| Asian | 1 (2.5%) |
| Mechanical ventilation | 34 (85%) |
| PIM score (mean ± SD) | 9.96 ± 16.20 |
| PRISM score (mean ± SD) | 23.54 ± 24.94 |
| Major somatic pharmacological features | |
| Recent increase or decrease of analgosedative medication | 22 (55%) |
| Neurological disorders | 21 (52%) |
| Infectious disorders | 20 (50%) |
| Respiratory disorders | 12 (30%) |
PIM, Pediatric Index of Mortality; PRISM, Pediatric Risk of Mortality
Table 4.
Patient characteristics of the 40 PICU cases with delirium 2002–2005
| No. | Sex | Age | Primary diagnosis on admission PICU | Mechanical ventilation | Delirium type | Treatment |
|---|---|---|---|---|---|---|
| 1 | M | 3 months | Multiple congenital malformations | + | Emerging | Haloperidol |
| 2 | F | 4 months | Meningococcal septic shock | + | Emerging | Risperidone |
| 3 | M | 4.5 months | Severe CLD | + | Emerging | Risperidone |
| 4 | F | 10 months | Near drowning | + | Emerging | Haloperidol |
| 5 | F | 1 year | Pneumonia | + | Emerging | Haloperidol |
| 6 | M | 1 year | Sepsis due to perforated appendicitis | + | Emerging | Haloperidol |
| 7 | M | 1 year | Subarachnoid bleeding | + | Emerging | Haloperidol |
| 8 | M | 1 year | Pericardial effusion with pretamponade | + | Hyperactive | Haloperidol |
| 9 | M | 1 year | Multiple dysmorphia, upper airway obstruction | – | Emerging | Haloperidol |
| 10 | F | 2 years | Meningococcal septic shock | + | Hyperactive | Haloperidol |
| 11 | M | 2 years | ADEM | + | Hyperactive | Risperidone |
| 12 | M | 2 years | Cervical mass, upper airway obstruction | + | Emerging | Haloperidol |
| 13 | M | 2 years | Aspiration and pneumothorax | + | Hypoactive | – |
| 14 | F | 2 years | Meningococcal meningitis with sepsis and DIC | + | Hyperactive | Haloperidol |
| 15 | F | 3 years | Cystic fibrosis and pneumonia | + | Hypoactive | Haloperidol |
| 16 | M | 4 years | Intracerebral hemorrhage, Marfan syndrome | + | Hypoactive | Haloperidol |
| 17 | M | 5 years | Medulloblastoma post surgery | + | Emerging | Risperidone |
| 18 | M | 5 years | Upper respiratory tract infection | + | Hyperactive | Haloperidol |
| 19 | M | 6 years | Multiple trauma | + | Emerging | Risperidone |
| 20 | F | 8 years | Meningo-encephalitis | + | Hyperactive | Risperidone |
| 21 | F | 8 years | Viral encephalitis | + | Hyperactive | – |
| 22 | F | 9 years | Status asthmaticus | + | Hyperactive | Risperidone |
| 23 | F | 9 years | TBI, gunshot wound | + | Hypoactive | Risperidone |
| 24 | M | 9 years | Status asthmaticus | + | Hyperactive | Haloperidol |
| 25 | M | 9 years | Neural tube defect and drain dysfunction | – | Emerging | first Haloperidol, then Risperidone |
| 26 | M | 11 years | Hypovolemic shock, typhus abdominalis | – | Hyperactive | Haloperidol |
| 27 | F | 12 years | TBI | + | Hypoactive | Haloperidol |
| 28 | M | 12 years | TBI and fracture of lower leg | + | Emerging | Haloperidol |
| 29 | M | 13 years | Sepsis, paronychia | – | Hyperactive | Haloperidol |
| 30 | M | 13 years | Status epilepticus | + | Emerging | Haloperidol |
| 31 | F | 14 years | TBI | + | Hyperactive | Haloperidol |
| 32 | F | 15 years | Post TBI | + | Hypoactive | Haloperidol |
| 33 | M | 15 years | Postoperative state | + | Hypoactive | Risperidone |
| 34 | M | 15 years | Acute lymphoblastic leukemia | – | Hypoactive | Haloperidol |
| 35 | M | 15 years | TBI | + | Emerging | Haloperidol |
| 36 | F | 15 years | Status asthmaticus | + | Hyperactive | Haloperidol |
| 37 | M | 16 years | Multiple trauma | – | Hypoactive | Haloperidol |
| 38 | F | 16 years | Bacterial meningitis | + | Hyperactive | Risperidone |
| 39 | M | 16 years | Respiratory failure, Duchenne disease | + | Emerging | Haloperidol |
| 40 | M | 17 years | Septic shock | + | Emerging | Haloperidol |
CLD, chronic lung disease; ADEM, acute disseminated encephalomyelitis; DIC, diffuse intravascular coagulation; TBI, traumatic brain injury
The underlying features were: recent increase or decrease in analgosedative medication (n = 22), neurological disorders (n = 21), infections (n = 20) and respiratory disorders (n = 12). Usually, a combination of these existed.
All but two patients were treated with an antipsychotic drug. Twenty-seven children were given haloperidol, 10 risperidone, and 1 child received both drugs in succession. In most cases, the beneficial results were observed rapidly, especially in the hyperactive forms, sometimes even after a single dose [11]. Sometimes it took a few hours or days. Two patients experienced acute dystonia as a likely side effect of the haloperidol, responding well to biperidene. Two children received no medication: one because of lack of consensus in our expert team, and one because of an endotracheal intubation at the time that medication was indicated. In most cases, the medication was stopped or tapered off successfully during hospitalization or afterwards in an outpatient setting. Five children (12.5%) with delirium died of their underlying disease; the mean PIM was 10% and the mean PRISM 24% (Table 3).
Discussion
This is the first systematic multidisciplinary study of the phenomenology and treatment of delirium in 40 critically ill children in a PICU context. The low cumulative incidence of 5% is mainly the result of the low incidence in the younger age groups (< 9 years old), this segment constituting the majority of the total sample (80%). A clearly higher incidence is seen in the older age groups. However, in critically ill adult patients substantially higher incidences have been reported, ranging from 10–30% in general hospital settings to 50% in postoperative patients and up to 80% in the terminally ill [2].
There are several possible explanations for this difference. First, the incidence of delirium in young critically ill children may be truly low: differences in age-related resilience and underlying conditions may contribute to true differences in the incidence between the very young and the very old. However this explanation seems unlikely, given the tendency in the very young to develop delirium under even much less severe circumstances [1, 11, 24]. A second explanation may relate to the fact that extensive psychosocial interventions are provided as a routine in Dutch PICU settings, with a possibly preventive effect on delirium in much the same way as observed in geriatric patients [25]. A third factor may be an anti-delirium effect of the routinely used analgosedative medication, although especially benzodiazepines may have excitatory side effects in children. A fourth factor may relate to reluctance on the part of the intensivists and/or child neurologists to request psychiatric evaluation in the case of behavioral changes for fear of adding stigmatization to an already burdened system. Perhaps the fifth, most likely and important explanation is that parents, nurses, pediatric intensivists and child neurologists do not easily recognize delirium, because the medical condition of these critically ill PICU children is so complex and changeable. It is possible that a psychiatric consultation was readily requested only in cases of anxiety and/or hyperactive delirium, not in the hypoactive and or veiled ones.
The differential diagnosis of pediatric delirium consists of acute stress reactions, acute anxiety states, adjustment disorders with mixed emotions, dissociative and/or regressive states and childhood-onset psychosis (see also Table 2). However, differentiating delirium from extreme stress and agitation due to acute and life-threatening conditions is not only impossible, it is also unwanted, because it is usually irrelevant at that point of time in the process of medical care. Causative treatment, if possible, is always the first step to be taken. Furthermore, the diagnosis of delirium in children is complicated by the fact that the criteria for adult delirium are not always easily applicable to children because of important differences in age and developmental levels. The DSM-IV criteria for delirium are not always useful in the case of pediatric delirium, especially in a PICU context. This is a source of concern. Moreover, delirium is not mentioned in the DSM-IV section on child psychiatry. DSM-IV describes as an essential feature of delirium the “disturbance of consciousness” leading to “impairment of the ability to focus, shift and sustain attention”. This, however, is of little relevance in the critically ill in a PICU context, where disturbance of attention is routinely present due to the disorder(s) itself. In fact, attention is often the first “to go” [17, 26]. In addition, patients almost always require treatment with opioids and benzodiazepines, which also have a strong impact on attention. Furthermore, it has been hypothesized that a disturbance of consciousness is not a discriminating characteristic of delirium in an ICU setting [27].
Our case series suggests that in addition to the hyper- and hypoactive forms of delirium, a third form may be characterized by anxiety, moaning, and/or restlessness. This was referred to as an “emerging” or veiled delirium, as described previously [17]. In the PICU population this state did not develop into a frank hyper- or hypoactive form, but appeared to exist in its own right, accompanied by disturbances of consciousness and cognition.
Although atypical presentations of disorders are often not captured in classification systems, the high prevalence of “emerging” delirium in our sample (17/40) stresses the importance of further phenomenological study. Adhering too strongly to DSM-IV criteria for adult delirium, for clinical use in a PICU context, may result in persistent under-diagnosis. On the other hand, the incidence of delirium may be overestimated by using the CAM-ICU, so most important is the issue of what constitutes delirium in critical illness [28].
There are accumulating indications that pediatric delirium can be subtle and accompanied or even dominated by other neuropsychiatric signs such as: reduction of awareness of the caregiver and/or the surrounding environment, purposeless actions, restlessness, inconsolability, signs of autonomic dysregulation and other subtle higher cortical dysfunctions [29–32]. Parents and nurses have a way of discriminating patterns in their children that may be diagnostically important. Thus, parents sometimes state: “This is not my child anymore.” Pediatric delirium therefore may have various subtle presentations and can be considered a spectrum disorder, which makes it sometimes difficult to diagnose [33]. Neither haloperidol nor risperidone is registered for the treatment of childhood delirium, although both are mentioned as the treatment of choice for adults [1]. Moreover, haloperidol is not registered for i.v. administration, even though it is used in this way in many places. We prefer risperidone in non-acute situations because of the theoretically lower risk of extrapyramidal side effects. Haloperidol and risperidone have been used for other indications in young children as well, such as childhood psychosis [34] or aggression in autism [35]. There are two limitations regarding our observations of treatment response. First, no severity scale for pediatric delirium was used, because none exists for this PICU population. Second, because no studies on the natural course of childhood delirium exist that have established the rate of spontaneous remission, spontaneous remissions may have been misclassified as response to treatment. In our opinion, however, the time frame of response points towards a medication effect.
Most medications employed in pediatrics and child psychiatry are used off-label [36], which means that special attention should be paid to information and informed consent procedures. Given the relatively high incidence of extrapyramidal symptoms with haloperidol, the “off-label” use needs further study [37].
This study has several limitations. First, it was a study of routine clinical practice. Observations were based on referrals emerging from care as usual. Although the focus on delirium may have altered referral paths and rates, we did not actively advocate any change, nor did we screen all admissions for delirium. In the absence of a hard clinical indication, no routine blood level measurements were performed to rule out persistent high levels of sedatives as a possible explanation for any neuropsychiatric symptom. Next, the lack of DSM-IV criteria for pediatric delirium and the disputed relevance of its main criterion in a (P)ICU setting make a standardized diagnosis difficult. Finally, treatment was provided in an open setting and based on consensus, especially among child psychiatrist, pediatric intensivist and child neurologist.
In conclusion, critically ill children in a PICU can develop delirium, with a hyperactive, hypoactive or veiled presentation, despite adequate analgosedation and intensive psychosocial interventions. The approach using an algorithmic structuring and an intensifying of daily clinical care, including the use of multidisciplinary daily consensus meetings, appeared effective in assessing, diagnosing and treating childhood delirium at the PICU. The findings suggest that the incidence is much lower than in adults, but a likely explanation is that delirious states requiring child psychiatric referral are still frequently under-diagnosed. There is also still a great need for developing delirium criteria in critically ill patients, children and adults alike, in a (P)ICU setting. Treatment with haloperidol or risperidone was successful in all patients. Future research is necessary to identify the risk factors for pediatric delirium in a multivariate prospective approach, to develop “easy to use” bedside tools for non-psychiatric trained team members for the early detection of delirium in all pediatric PICU patients, and to study the effects of interventions in a double-blind and ideally placebo-controlled fashion.
Disclosure
Jim van Os is a speaker or member of the advisory board for Lilly, BMS, Janssen-Cilag and AstraZeneca. He received grant or research support from Lilly, GSK, BMS and AstraZeneca. Albert Leentjens participated in research for Boehringer Ingelheim and is on the advisory board of a study related to Parkinson's disease by the same company. The other authors have no financial relationships to disclose.
Acknowledgements
We gratefully acknowledge the cooperation with, and help from, our colleagues in the study group on pediatric delirium, especially Kirsten Venrooij and Richel Lousberg, M.Sc., Ph.D, and our colleagues in the PICU: the pediatric intensivists, child neurologists and nurses.
Footnotes
J. N. M. Schieveld and P. L. J. M. Leroy contributed equally to this paper.
References
- 1.American Psychiatric Association (2000) Diagnostic and statistical manual of mental disorders, 4th edition text, revision (DSM-IV-TR). American Psychiatric Association, Washington DC
- 2.American Psychiatric Association (1999) Practice guidelines for the treatment of patients with delirium. Am J Psychiatry 5:1–20 [PubMed]
- 3.Leentjens AFG, Van der Mast RC (2005) Delirium in the elderly: an update. Curr Opin Psychiatry 18:325–330 [DOI] [PubMed]
- 4.Trzepacz PT, Van der Mast RC (2002) Pathophysiology of delirium. In: Lindesay J, Rockwood K, MacDonald A (eds) Delirium in old age. Oxford University Press, Oxford, pp 51–90
- 5.Wise MG, Hilty DM, Cerda GM, Trzepacz PT (2002) Delirium (confusional states). In: Wise MG, Rundell JR (eds) Textbook of consultation–liaison psychiatry in the medically ill. American Psychiatric Publishing, Washington, DC, pp 257–272
- 6.Ely EW, Shintani A, Truman B, Speroff T, Gordon SM, Harrell Jr FE, Inouye SK, Bernard GR, Dittus RS (2004) Delirium as a predictor of mortality in mechanically ventilated patients in the intensive care unit. J Am Med Assoc 291:1753–1762 [DOI] [PubMed]
- 7.Ely E, Inouye SK, Bernard GR, Gordon S, Francis J, May L, Truman B, Speroff T, Gautam S, Margolin R, Hart RP, Dittus R (2001) Delirium in mechanically ventilated patients: validity and reliability of the confusion assessment method for the intensive care unit (CAM-ICU). J Am Med Assoc 286:2703–2710 [DOI] [PubMed]
- 8.Jacobi J, Fraser GL, Coursin DB, Riker RR, Fontaine D, Wittbrodt ET, Chalfin DB, Masica MF, Bjerke HS, Coplin WM, Crippen DW, Fuchs BD, Kelleher RM, Marik PE, Nasraway Jr SA, Murray MJ, Peruzzi WT, Lumb PD (2002) Clinical practise guidelines for the sustained use of sedatives and analgesics in the critically ill adult. Crit Care Med 30:119–141 [DOI] [PubMed]
- 9.Andrews P, Azoulay E, Antonelli M, Brochard L, Brun-Buisson C, Dobb G, Fagon J, Gerlach H, Groeneveld J, Mancebo J, Metnitz P, Nava S, Pugin J, Pinsky M, Radermacher P, Richard C, Tasker R, Vallet B (2005) Year in review in intensive care medicine, 2004. III. Outcome, ICU organisation, scoring, quality of life, ethics, psychological problems and communication in the ICU, immunity and hemodynamics during sepsis, pediatric and neonatal critical care, experimental studies. Intensive Care Med 31:356–372 [DOI] [PubMed]
- 10.Martini RC (2005) Commentary: The diagnosis of delirium in pediatric patients. J Am Acad Child Adolesc Psychiatry 44:395–398 [DOI] [PubMed]
- 11.Schieveld JNM, Leentjens AFG (2005) Delirium in severely ill young children in the pediatric intensive care unit. J Am Acad Child Adolesc Psychiatry 44:392–394 [DOI] [PubMed]
- 12.Turkel SB, Trzepacz PT, Tavare CJ (2006) Comparing symptoms of delirium in adults and children. Psychosomatics 47:320–324 [DOI] [PubMed]
- 13.Turkel SB, Tavare CJ (2003) Delirium in children and adolescents. J Neuropsychiatry Clin Neurosci 15:431–435 [DOI] [PubMed]
- 14.Tobias JD (1999) Sedation and analgesia in pediatric intensive care units: a guide to drug selection and use. Pediatr Drugs 1:109–126 [DOI] [PubMed]
- 15.Tobias JD (2000) Tolerance, withdrawal, and physical dependency after long-term sedation and analgesia of children in the pediatric intensive care unit. Crit Care Med 28:2122–2132 [DOI] [PubMed]
- 16.Anand KJ, Arnold JH (1996) Opioid tolerance and dependence in infants and children. Crit Care Med 22:334–342 [DOI] [PubMed]
- 17.Lishman WA (1998) Cardinal psychological features of cerebral disorder. In: Lishman WA (ed) Organic psychiatry: the psychological consequences of cerebral disorder, 3rd edn. Blackwell Science, Oxford, vol 8, pp 3–20
- 18.Cole M, McCusker J, Dendukuri N, Han L (2003) The prognostic significance of subsyndromal delirium in elderly medical inpatients. J Am Geriatr Soc 51:754–760 [DOI] [PubMed]
- 19.Lipowski ZV (1990) Delirium. Acute confusional state. Oxford University Press, New York
- 20.Gemke RJ, van Vught J (2002) Scoring systems in pediatric intensive care: PRISM III versus PIM. Intensive Care Med 28:204–207 [DOI] [PubMed]
- 21.Nederlandse Vereniging voor Psychiatrie (2004) Richtlijn Delirium. Uitgeverij Boom, Amsterdam
- 22.Brown RL, Henke A, Greenhalgh DG, Warden GD (1996) The use of haloperidol in the agitated, critically ill pediatric patient with burns. J Burn Care Rehabil 17:34–38 [DOI] [PubMed]
- 23.Harrison AM, Lugo RA, Lee WE, Appachi E, Bourdakos D, Davis SJ, McHugh MJ, Weise KL (2002) The use of haloperidol in agitated, critically ill children. Clin Pediatr 41:51–54 [DOI] [PubMed]
- 24.Williams DT (2002) Neuropsychiatric signs, symptoms, and syndromes. In: Lewis M (ed) Child and adolescent psychiatry, a comprehensive textbook. Lippincott Williams & Wilkins, Philadelphia, pp 399–404
- 25.Inouye SK, Bogardus ST, Williams CS, Leo-Summers L, Agostini JV (2003) The role of adherence on the effectiveness of non-pharmacologic interventions. Arch Intern Med 163:958–964 [DOI] [PubMed]
- 26.Weiss M, Weiss G (2002) Attention deficit hyperactivity disorder. In: Lewis M (ed) Child and adolescent psychiatry, a comprehensive textbook. Lippincott Williams & Wilkins, Philadelphia, pp 645–670
- 27.Bergeron N, Skrobik YK, Dubois MJ (2005) Is disturbance of consciousness an important feature of ICU delirium? Intensive Care Med 31:887 [DOI] [PubMed]
- 28.Quimet S, Kavanagh BP, Gottfried SB, Skrobik Y (2007) Incidence, risk factors and consequences of ICU delirium. Intensive Care Med 33:66–73 [DOI] [PubMed]
- 29.Stoddard FJ, Wilens TE (1995) Delirium. In: Jellinek MS, Herzog DB (eds) Psychiatric aspects of general hospital pediatrics. Yearbook, Chicago, pp 254–259
- 30.Sikich N, Lerman J (2004) Development and psychometric evaluation of the pediatric anesthesia emergence delirium scale. Anesthesiology 100:1138–1145 [DOI] [PubMed]
- 31.Wise MG (1987) Delirium. In: Hales RE, Yudofsky SC (eds) Textbook of neuropsychiatry. The American Psychiatric Press, Washington, DC, pp 89–105
- 32.Trzepacz PT, Meagher DJ (2005) Delirium. In: Levinson JL (ed) Textbook of psychosomatic medicine. American Psychiatric Publishing, Washington, DC, pp 92–130
- 33.Schieveld JNM (2005) Case reports with a child psychiatric exploration of catatonia, autism, and delirium. In: Dhossche DM, Wing L, Ohta M, Neumarker K (eds) Catatonia in autism spectrum disorders. International review of neurobiology, vol 72. Elsevier Academic Press, Amsterdam, pp 195–206 [DOI] [PubMed]
- 34.Carlson GA (2002) Clinical aspects of child and adolescent psychopharmacology. In: Kutcher S (ed) Practical child and adolescent psychopharmacology. Cambridge University Press, Cambridge, pp 70–90
- 35.Lynn D, King BH (2002) Aggressive behavior. In: Kutcher S (ed) Practical child and adolescent psychopharmacology. Cambridge University Press, Cambridge, pp 305–327
- 36.Hill P (2005) Off licence and off label prescribing in children: litigation fears for physicians. Arch Dis Child 90[Suppl 1]:i17–i18 [DOI] [PMC free article] [PubMed]
- 37.Ratcliff SL, Meyer WJ, Cuervo LJ, Villarreal C, Thomas CR, Herndon DN (2004) The use of Haloperidol and associated complications in the agitated, acutely ill pediatric burn patient. J Burn Care Rehabil 25:472–478 [DOI] [PubMed]


