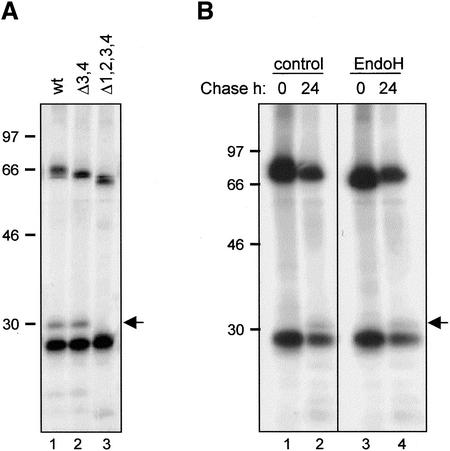
Figure 5.
Vacuolar delivery of IgA/G is mediated by the Golgi complex. (A) SR1 protoplasts were transfected with plasmids encoding κ chain and γ/α heavy chain (wt), γ/α lacking the two C-terminal glycosylation sites (Δ3,4) or γ/α lacking all four glycosylation sites (Δ1,2,3,4), respectively. Cells were pulse-labeled for 16 h, homogenized, and immunoprecipitated with anti-IgG antiserum. Proteins were visualized by SDS-PAGE and fluorography. (B) Cells were transfected with plasmids encoding κ and γ/α chains and treated as in A. Immunoprecipitates were subjected to treatment with endoglycosidase H (endo H) or buffer (control) before SDS-PAGE analysis. The arrow indicates the position of the 30-kDa vacuolar fragmentation product. Numbers at left indicate molecular mass markers in kilodaltons.