Abstract
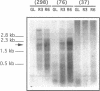
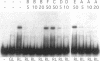
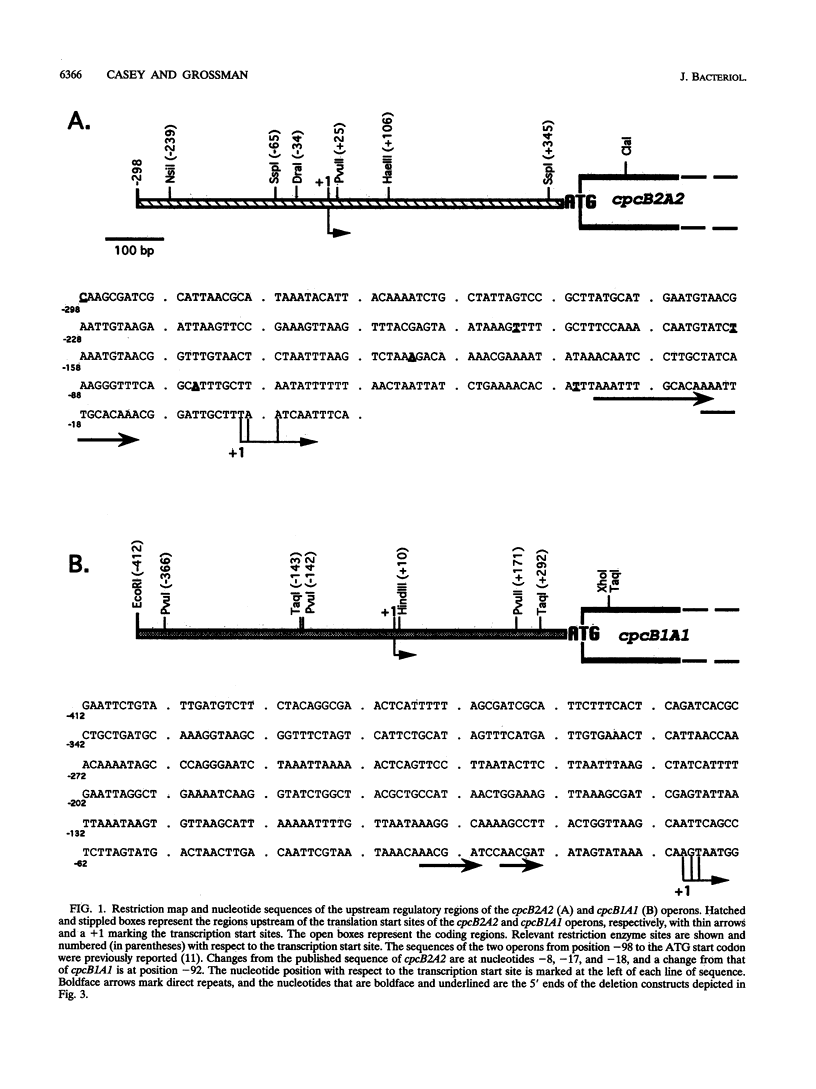
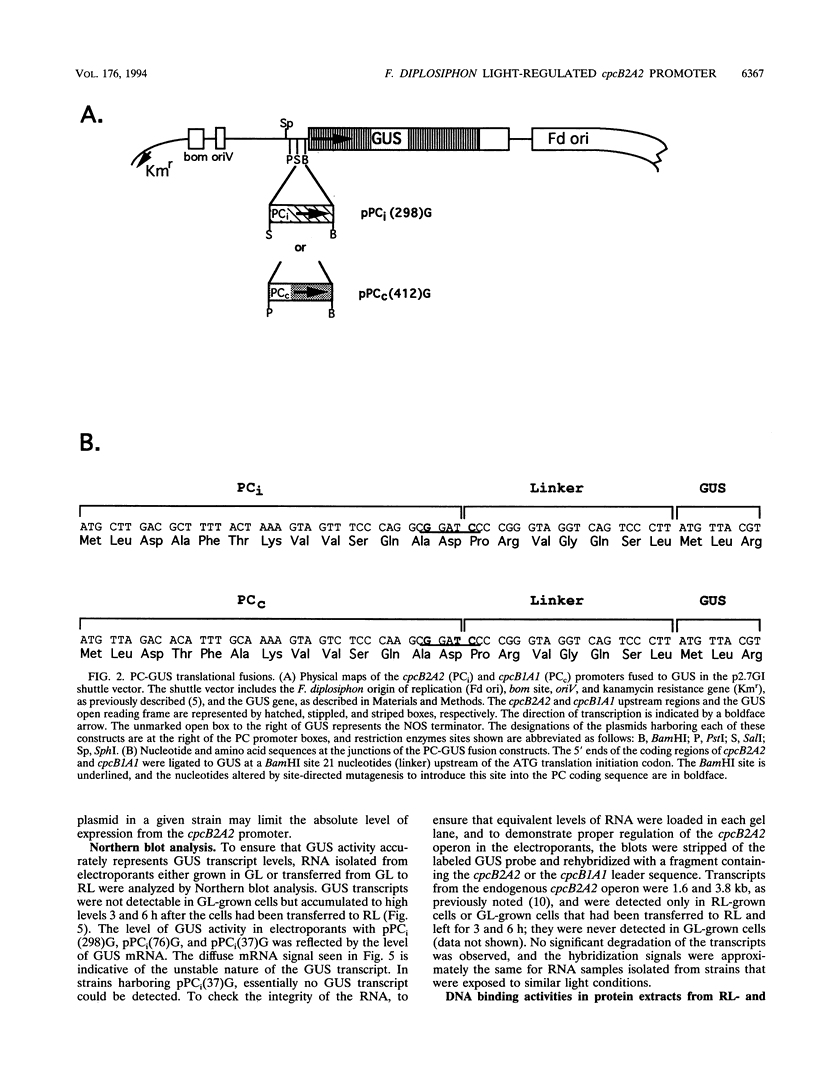
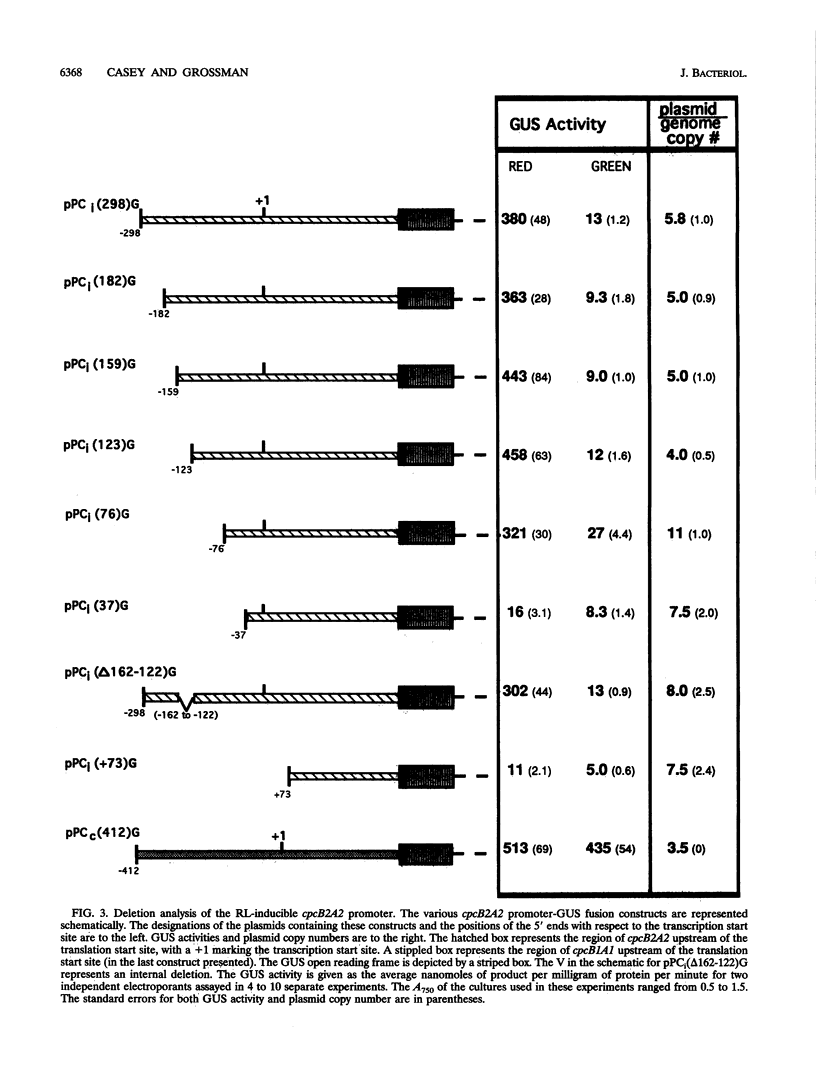
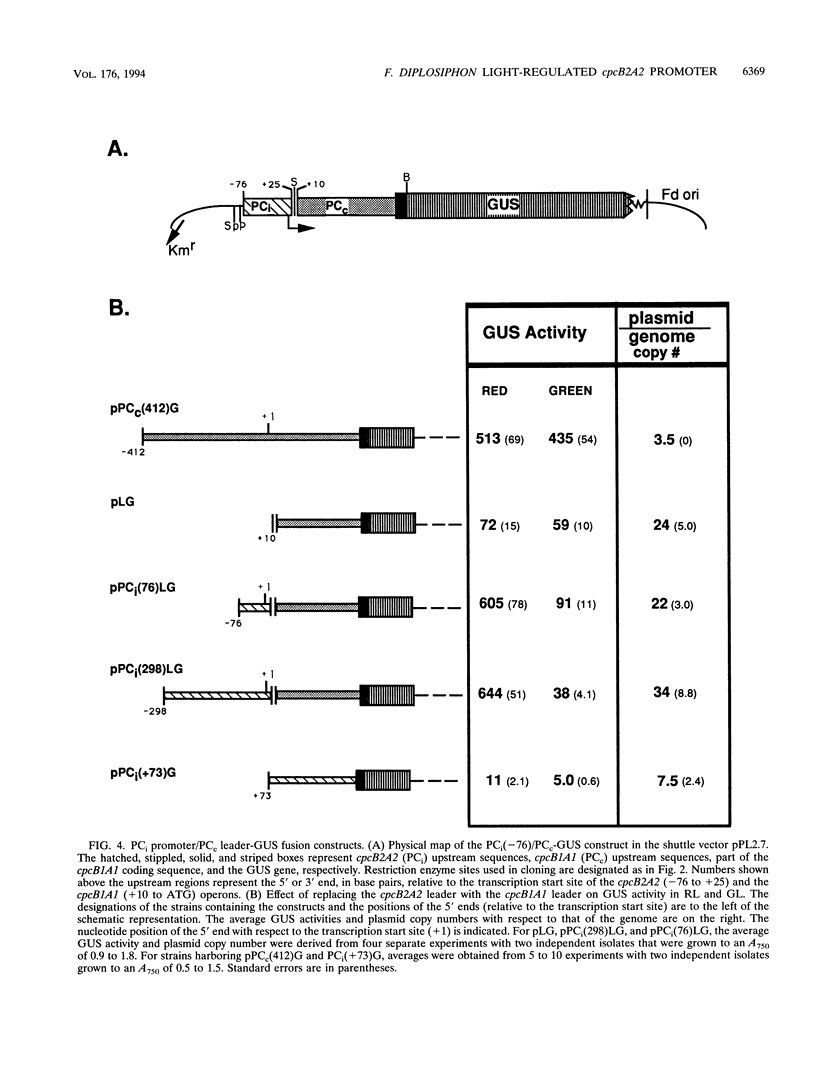
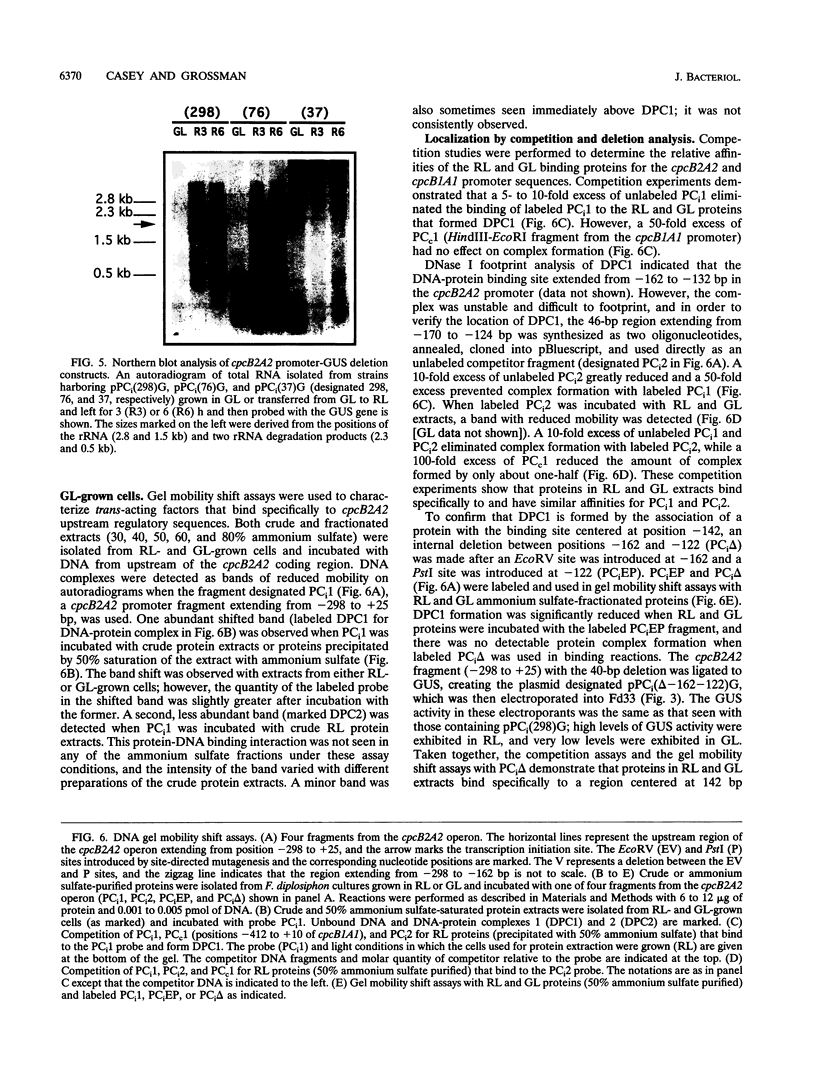
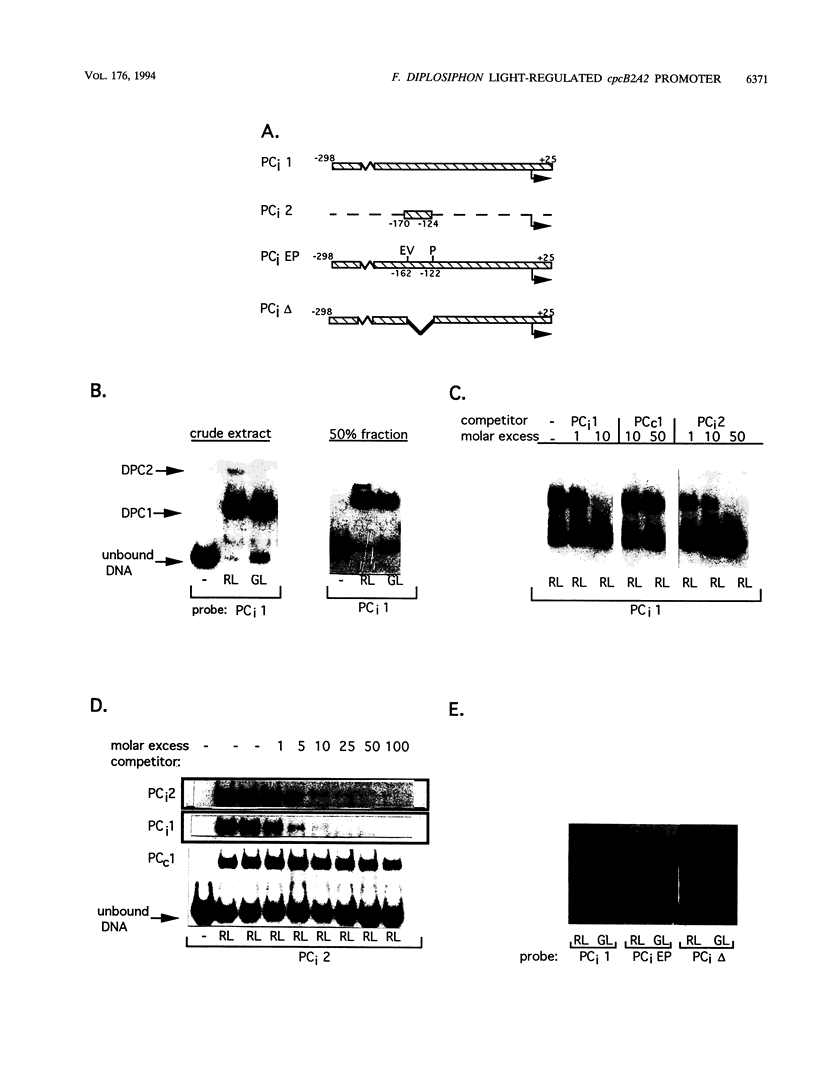
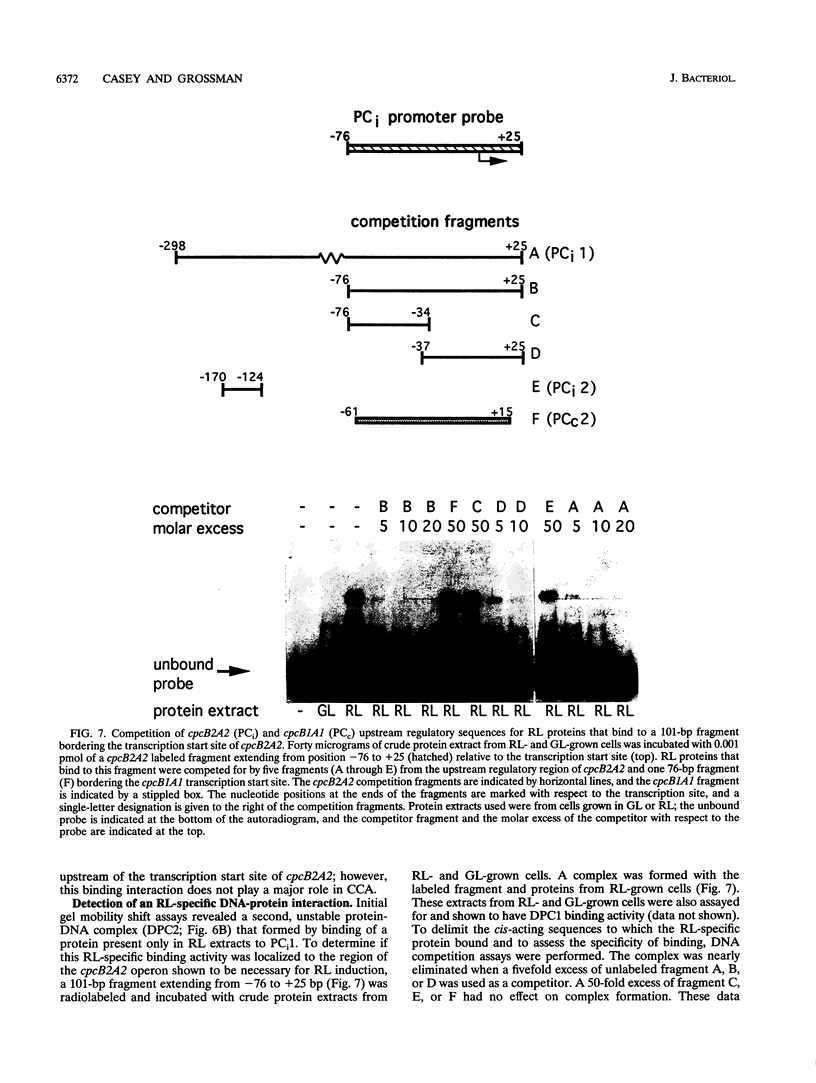
When exposed to different spectral qualities of light, many cyanobacteria dramatically alter their phycobilisome rod composition in a process termed complementary chromatic adaptation. In the cyanobacterium Fremyella diplosiphon, this response is associated with differential expression of the cpcB2A2, cpeBA, and cpeCDE operons, which code for the phycobiliproteins phycocyanin and phycoerythrin and the phycoerythrin linker polypeptides, respectively. To define components of the signal transduction pathway involved in light-regulated expression of genes encoding phycobilisome polypeptides, we have used in vivo and in vitro techniques to identify cis-acting sequences and trans-acting factors necessary for the regulation of the red-light-inducible cpcB2A2 operon. Deletion of the cpcB2A2 upstream sequences to -76 bp with respect to the transcription start site had no effect on red-light induction of a cpcB2A2-beta-glucuronidase (GUS) chimeric gene, while deletion to -37 bp abolished GUS expression. Furthermore, a fragment of the cpcB2A2 gene from -76 to +25 bp linked to the untranslated leader of cpcB1A1 (a constitutively expressed operon encoding phycocyanin) is sufficient to drive high-level GUS expression in red light. Therefore, the sequence between positions -76 and -37 is necessary for the expression of cpcB2A2, and the region extending from -76 to +25 is sufficient for red-light induction of the operon. Attempts were made to correlate the in vivo data with protein binding in the region upstream of the transcription start site of cpcB2A2. Using in vitro analysis, we detected two protein-binding sites in the cpcB2A2 promoter which were localized to positions -162 to -122 and -37 to +25. Proteins from both red- and green-light-grown cells interacted with the former site, while only proteins present in extracts from red-light-grown cells interacted with the latter site. The data from both the in vivo and in vitro analyses suggest that while two regions upstream of the cpcB2A2 transcription initiation site specifically bind proteins, only the binding site bordering the transcription start site is important for complementary chromatic adaptation.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Chiang G. G., Schaefer M. R., Grossman A. R. Complementation of a red-light-indifferent cyanobacterial mutant. Proc Natl Acad Sci U S A. 1992 Oct 15;89(20):9415–9419. doi: 10.1073/pnas.89.20.9415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cobley J. G., Miranda R. D. Mutations affecting chromatic adaptation in the cyanobacterium Fremyella diplosiphon. J Bacteriol. 1983 Mar;153(3):1486–1492. doi: 10.1128/jb.153.3.1486-1492.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collado-Vides J., Magasanik B., Gralla J. D. Control site location and transcriptional regulation in Escherichia coli. Microbiol Rev. 1991 Sep;55(3):371–394. doi: 10.1128/mr.55.3.371-394.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collier J. L., Grossman A. R. Chlorosis induced by nutrient deprivation in Synechococcus sp. strain PCC 7942: not all bleaching is the same. J Bacteriol. 1992 Jul;174(14):4718–4726. doi: 10.1128/jb.174.14.4718-4726.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conley P. B., Lemaux P. G., Grossman A. R. Cyanobacterial light-harvesting complex subunits encoded in two red light-induced transcripts. Science. 1985 Nov 1;230(4725):550–553. doi: 10.1126/science.3931221. [DOI] [PubMed] [Google Scholar]
- Conley P. B., Lemaux P. G., Grossman A. Molecular characterization and evolution of sequences encoding light-harvesting components in the chromatically adapting cyanobacterium Fremyella diplosiphon. J Mol Biol. 1988 Feb 5;199(3):447–465. doi: 10.1016/0022-2836(88)90617-1. [DOI] [PubMed] [Google Scholar]
- Conley P. B., Lemaux P. G., Lomax T. L., Grossman A. R. Genes encoding major light-harvesting polypeptides are clustered on the genome of the cyanobacterium Fremyella diplosiphon. Proc Natl Acad Sci U S A. 1986 Jun;83(11):3924–3928. doi: 10.1073/pnas.83.11.3924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Federspiel N. A., Grossman A. R. Characterization of the light-regulated operon encoding the phycoerythrin-associated linker proteins from the cyanobacterium Fremyella diplosiphon. J Bacteriol. 1990 Jul;172(7):4072–4081. doi: 10.1128/jb.172.7.4072-4081.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Federspiel N. A., Scott L. Characterization of a light-regulated gene encoding a new phycoerythrin-associated linker protein from the cyanobacterium Fremyella diplosiphon. J Bacteriol. 1992 Sep;174(18):5994–5998. doi: 10.1128/jb.174.18.5994-5998.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feinberg A. P., Vogelstein B. A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal Biochem. 1983 Jul 1;132(1):6–13. doi: 10.1016/0003-2697(83)90418-9. [DOI] [PubMed] [Google Scholar]
- Glazer A. N., Lundell D. J., Yamanaka G., Williams R. C. The structure of a "simple" phycobilisome. Ann Microbiol (Paris) 1983 Jul-Aug;134B(1):159–180. doi: 10.1016/s0769-2609(83)80103-3. [DOI] [PubMed] [Google Scholar]
- Henikoff S. Unidirectional digestion with exonuclease III in DNA sequence analysis. Methods Enzymol. 1987;155:156–165. doi: 10.1016/0076-6879(87)55014-5. [DOI] [PubMed] [Google Scholar]
- Ho Y. S., Rosenberg M. Characterization of a third, cII-dependent, coordinately activated promoter on phage lambda involved in lysogenic development. J Biol Chem. 1985 Sep 25;260(21):11838–11844. [PubMed] [Google Scholar]
- Ho Y. S., Wulff D. L., Rosenberg M. Bacteriophage lambda protein cII binds promoters on the opposite face of the DNA helix from RNA polymerase. Nature. 1983 Aug 25;304(5928):703–708. doi: 10.1038/304703a0. [DOI] [PubMed] [Google Scholar]
- Lomax T. L., Conley P. B., Schilling J., Grossman A. R. Isolation and characterization of light-regulated phycobilisome linker polypeptide genes and their transcription as a polycistronic mRNA. J Bacteriol. 1987 Jun;169(6):2675–2684. doi: 10.1128/jb.169.6.2675-2684.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazel D., Guglielmi G., Houmard J., Sidler W., Bryant D. A., Tandeau de Marsac N. Green light induces transcription of the phycoerythrin operon in the cyanobacterium Calothrix 7601. Nucleic Acids Res. 1986 Nov 11;14(21):8279–8290. doi: 10.1093/nar/14.21.8279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazel D., Marlière P. Adaptive eradication of methionine and cysteine from cyanobacterial light-harvesting proteins. Nature. 1989 Sep 21;341(6239):245–248. doi: 10.1038/341245a0. [DOI] [PubMed] [Google Scholar]
- Miller V. L., Taylor R. K., Mekalanos J. J. Cholera toxin transcriptional activator toxR is a transmembrane DNA binding protein. Cell. 1987 Jan 30;48(2):271–279. doi: 10.1016/0092-8674(87)90430-2. [DOI] [PubMed] [Google Scholar]
- O'Halloran T. V., Frantz B., Shin M. K., Ralston D. M., Wright J. G. The MerR heavy metal receptor mediates positive activation in a topologically novel transcription complex. Cell. 1989 Jan 13;56(1):119–129. doi: 10.1016/0092-8674(89)90990-2. [DOI] [PubMed] [Google Scholar]
- Oelmüller R., Conley P. B., Federspiel N., Briggs W. R., Grossman A. R. Changes in Accumulation and Synthesis of Transcripts Encoding Phycobilisome Components during Acclimation of Fremyella diplosiphon to Different Light Qualities. Plant Physiol. 1988 Dec;88(4):1077–1083. doi: 10.1104/pp.88.4.1077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rampersaud A., Norioka S., Inouye M. Characterization of OmpR binding sequences in the upstream region of the ompF promoter essential for transcriptional activation. J Biol Chem. 1989 Nov 5;264(31):18693–18700. [PubMed] [Google Scholar]
- Reed K. C., Mann D. A. Rapid transfer of DNA from agarose gels to nylon membranes. Nucleic Acids Res. 1985 Oct 25;13(20):7207–7221. doi: 10.1093/nar/13.20.7207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ronson C. W., Nixon B. T., Ausubel F. M. Conserved domains in bacterial regulatory proteins that respond to environmental stimuli. Cell. 1987 Jun 5;49(5):579–581. doi: 10.1016/0092-8674(87)90530-7. [DOI] [PubMed] [Google Scholar]
- Schmidt-Goff C. M., Federspiel N. A. In vivo and in vitro footprinting of a light-regulated promoter in the cyanobacterium Fremyella diplosiphon. J Bacteriol. 1993 Mar;175(6):1806–1813. doi: 10.1128/jb.175.6.1806-1813.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sobczyk A., Schyns G., Tandeau de Marsac N., Houmard J. Transduction of the light signal during complementary chromatic adaptation in the cyanobacterium Calothrix sp. PCC 7601: DNA-binding proteins and modulation by phosphorylation. EMBO J. 1993 Mar;12(3):997–1004. doi: 10.1002/j.1460-2075.1993.tb05740.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stock J. B., Ninfa A. J., Stock A. M. Protein phosphorylation and regulation of adaptive responses in bacteria. Microbiol Rev. 1989 Dec;53(4):450–490. doi: 10.1128/mr.53.4.450-490.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wanner B. L. Gene regulation by phosphate in enteric bacteria. J Cell Biochem. 1993 Jan;51(1):47–54. doi: 10.1002/jcb.240510110. [DOI] [PubMed] [Google Scholar]