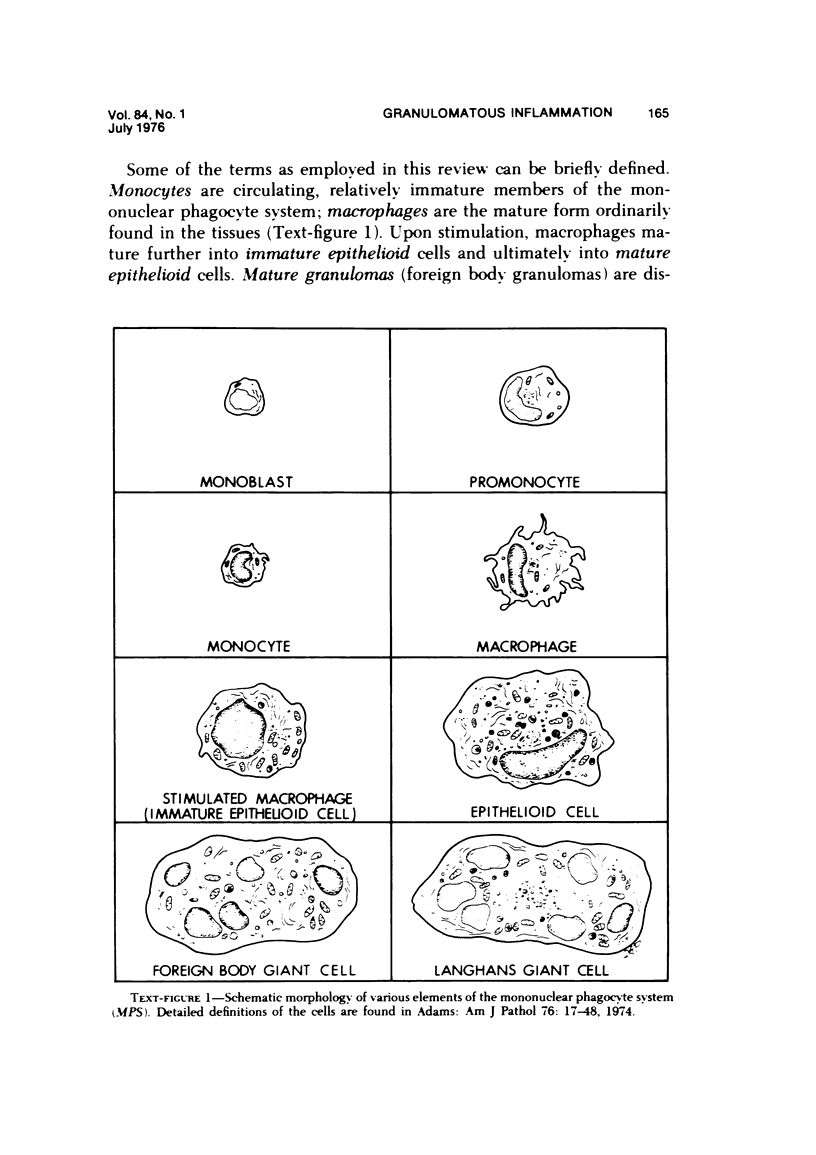
Full text
PDF




























Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adams D. O., Biesecker J. L., Koss L. G. The activation of mononuclear phagocytes in vitro: immunologically mediated enhancement. J Reticuloendothel Soc. 1973 Dec;14(6):550–570. [PubMed] [Google Scholar]
- Adams D. O. Experimental pine pollen granulomatous pneumonia in the rat. Am J Pathol. 1966 Jul;49(1):153–165. [PMC free article] [PubMed] [Google Scholar]
- Adams D. O. The structure of mononuclear phagocytes differentiating in vivo. I. Sequential fine and histologic studies of the effect of Bacillus Calmette-Guerin (BCG). Am J Pathol. 1974 Jul;76(1):17–48. [PMC free article] [PubMed] [Google Scholar]
- Ando M., Dannenberg A. M., Jr, Shima K. Macrophage accumulation, division, maturation and digestive and microbicidal capacities in tuberculous lesions. II. Rate at which mononuclear cells enter and divide in primary BCG lesions and those of reinfection. J Immunol. 1972 Jul;109(1):8–19. [PubMed] [Google Scholar]
- Axline S. G. Functional biochemistry of the macrophage. Semin Hematol. 1970 Apr;7(2):142–160. [PubMed] [Google Scholar]
- Bast R. C., Jr, Zbar B., Borsos T., Rapp H. J. BCG and cancer. N Engl J Med. 1974 Jun 27;290(26):1458–1469. doi: 10.1056/NEJM197406272902605. [DOI] [PubMed] [Google Scholar]
- Berry C. L. The development of the granuloma of histoplasmosis. J Pathol. 1969 Jan;97(1):1–10. doi: 10.1002/path.1710970102. [DOI] [PubMed] [Google Scholar]
- Black M. M., Epstein W. L. Formation of multinucleate giant cells in organized epitheloid cell granulomas. Am J Pathol. 1974 Feb;74(2):263–274. [PMC free article] [PubMed] [Google Scholar]
- Blanden R. V. Modification of macrophage function. J Reticuloendothel Soc. 1968 Jun;5(3):179–202. [PubMed] [Google Scholar]
- Bloom B. R. In vitro approaches to the mechanism of cell-mediated immune reactions. Adv Immunol. 1971;13:101–208. doi: 10.1016/s0065-2776(08)60184-4. [DOI] [PubMed] [Google Scholar]
- Boros D. L., Warren K. S., Pelley R. P. The secretion of migration inhibitory factor by intact schistosome egg granulomas maintained in vitro. Nature. 1973 Nov 23;246(5430):224–226. doi: 10.1038/246224a0. [DOI] [PubMed] [Google Scholar]
- Calderon J., Kiely J. M., Lefko J. L., Unanue E. R. The modulation of lymphocyte functions by molecules secreted by macrophages. I. Description and partial biochemical analysis. J Exp Med. 1975 Jul 1;142(1):151–164. doi: 10.1084/jem.142.1.151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carbone P. P., Zipkin I., Sokoloff L., Frazier P., Cook P., Mullins F. Fluoride effect on bone in plasma cell myeloma. Arch Intern Med. 1968 Feb;121(2):130–140. [PubMed] [Google Scholar]
- Carr I., Clarke J. A., Salsbury A. J. The surface structure of mouse peritoneal cells--a study with the scanning electron microscope. J Microsc. 1969;89(1):105–111. doi: 10.1111/j.1365-2818.1969.tb00654.x. [DOI] [PubMed] [Google Scholar]
- Cohn Z. A. The structure and function of monocytes and macrophages. Adv Immunol. 1968;9:163–214. doi: 10.1016/s0065-2776(08)60443-5. [DOI] [PubMed] [Google Scholar]
- Dannenberg A. M., Jr, Ando M., Shima K. Macrophage accumulation, division, maturation, and digestive and microbicidal capacities in tuberculous lesions. 3. The turnover of macrophages and its relation to their activation and antimicrobial immunity in primary BCG lesions and those of reinfection. J Immunol. 1972 Nov;109(5):1109–1121. [PubMed] [Google Scholar]
- Dannenberg A. M., Jr Cellular hypersensitivity and cellular immunity in the pathogensis of tuberculosis: specificity, systemic and local nature, and associated macrophage enzymes. Bacteriol Rev. 1968 Jun;32(2):85–102. doi: 10.1128/br.32.2.85-102.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dannenberg A. M., Jr, Meyer O. T., Esterly J. R., Kambara T. The local nature of immunity in tuberculosis, illustrated histochemically in dermal BCG lesions. J Immunol. 1968 May;100(5):931–941. [PubMed] [Google Scholar]
- Dumont A., Sheldon H. Changes in the fine structure of macrophages in experimentally produced tuberculous granulomas in hamsters. Lab Invest. 1965 Nov;14(11):2034–2055. [PubMed] [Google Scholar]
- Elias P. M., Epstein W. L. Ultrastructural observations on experimentally induced foreign-body and organized epithelioid-cell granulomas in man. Am J Pathol. 1968 Jun;52(6):1207–1223. [PMC free article] [PubMed] [Google Scholar]
- Epstein W. L. Granulomatous hypersensitivity. Prog Allergy. 1967;11:36–88. [PubMed] [Google Scholar]
- Epstein W. L., Krasnobrod H. The origin of epithelioid cells in experimental granulomas of man. Lab Invest. 1968 Feb;18(2):190–195. [PubMed] [Google Scholar]
- Epstein W. L. Methal-induced granulomatous hypersensitivity in man. Adv Biol Skin. 1971;11:313–335. [PubMed] [Google Scholar]
- Fedorko M. E., Hirsch J. G. Structure of monocytes and macrophages. Semin Hematol. 1970 Apr;7(2):109–124. [PubMed] [Google Scholar]
- Feldman J. D., Tubergen D. G., Pollock E. M., Unanue E. R. Distribution of a macrophage-specific antigen. Cell Immunol. 1972 Oct;5(2):325–337. doi: 10.1016/0008-8749(72)90058-5. [DOI] [PubMed] [Google Scholar]
- Galindo B., Imaeda T. Cellular response to Freund's adjuvant in the rabbit lung. An electron microscope study. Lab Invest. 1966 Nov;15(11):1659–1681. [PubMed] [Google Scholar]
- Golde D. W., Finley T. N., Cline M. J. Production of colony-stimulating factor by human macrophages. Lancet. 1972 Dec 30;2(7792):1397–1399. doi: 10.1016/s0140-6736(72)92966-2. [DOI] [PubMed] [Google Scholar]
- Gordon S., Cohn Z. A. The macrophage. Int Rev Cytol. 1973;36:171–214. doi: 10.1016/s0074-7696(08)60218-1. [DOI] [PubMed] [Google Scholar]
- Gusek W. Histologie und elektronenmikroskopische komparative Zytologie tuberkulöser und epitheloidzelliger Granulome. Bibl Tuberc. 1965;21:97–156. [PubMed] [Google Scholar]
- Hanifin J. M., Epstein W. L., Cline M. J. In vitro studies on granulomatous hypersensitivity to beryllium. J Invest Dermatol. 1970 Oct;55(4):284–288. doi: 10.1111/1523-1747.ep12260036. [DOI] [PubMed] [Google Scholar]
- Hanna M. G., Jr Immunologic aspects of BCG-mediated regression of established tumors and metastases in guinea pigs. Semin Oncol. 1974 Dec;1(4):319–335. [PubMed] [Google Scholar]
- James D. G., Neville E., Walker A. Immunology of sarcoidosis. Am J Med. 1975 Sep;59(3):388–394. doi: 10.1016/0002-9343(75)90397-6. [DOI] [PubMed] [Google Scholar]
- James E. M., Williams W. J. Fine structure and histochemistry of epithelioid cells in sarcoidosis. Thorax. 1974 Jan;29(1):115–120. doi: 10.1136/thx.29.1.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- James S. L., Colley D. G. Eosinophils and immune mechanisms: production of the lymphokine eosinophil stimulation promoter (ESP) in vitro by isolated intact granulomas. J Reticuloendothel Soc. 1975 Nov;18(5):283–293. [PubMed] [Google Scholar]
- Levy M. H., Wheelock E. F. The role of macrophages in defense against neoplastic disease. Adv Cancer Res. 1974;20:131–163. doi: 10.1016/s0065-230x(08)60110-4. [DOI] [PubMed] [Google Scholar]
- Lin H., Stewart C. C. Colony formation by mouse peritoneal exudate cells in vitro. Nat New Biol. 1973 Jun 6;243(127):176–177. doi: 10.1038/newbio243176a0. [DOI] [PubMed] [Google Scholar]
- Loewi G., Papamichail M. Studies of mononuclear cells from inflammatory joint effusions. Int Arch Allergy Appl Immunol. 1973;45(1):285–289. doi: 10.1159/000231043. [DOI] [PubMed] [Google Scholar]
- Mariano M., Spector W. G. The formation and properties of macrophage polykaryons (inflammatory giant cells). J Pathol. 1974 May;113(1):1–19. doi: 10.1002/path.1711130102. [DOI] [PubMed] [Google Scholar]
- Mauel J., Defendi V. Regulation of DNA synthesis in mouse macrophages. II. Studies on mechanisms of action of the macrophage growth factor. Exp Cell Res. 1971 Apr;65(2):377–385. doi: 10.1016/0014-4827(71)90016-4. [DOI] [PubMed] [Google Scholar]
- McDougal J. S., Azar H. A. Tritiated proline in macrophages. In vivo and in vitro uptake by foreign-body granulomas. Arch Pathol. 1972 Jan;93(1):13–17. [PubMed] [Google Scholar]
- McGregor D. D. Cytokinetics and fate of sensitized lymphocytes. J Reticuloendothel Soc. 1975 Feb;17(2):126–132. [PubMed] [Google Scholar]
- Myrvik Q. N. Adjuvants. Ann N Y Acad Sci. 1974;221:324–330. doi: 10.1111/j.1749-6632.1974.tb28233.x. [DOI] [PubMed] [Google Scholar]
- Page R. C., Davies P., Allison A. C. Participation of mononuclear phagocytes in chronic inflammatory diseases. J Reticuloendothel Soc. 1974 May;15(5):413–438. [PubMed] [Google Scholar]
- Papadimitriou J. M., Archer M. The morphology of murine foreign body multinucleate giant cells. J Ultrastruct Res. 1974 Dec;49(3):372–386. doi: 10.1016/s0022-5320(74)90051-3. [DOI] [PubMed] [Google Scholar]
- Papadimitriou J. M., Spector W. G. The origin, properties and fate of epithelioid cells. J Pathol. 1971 Nov;105(3):187–203. doi: 10.1002/path.1711050305. [DOI] [PubMed] [Google Scholar]
- Papadimitriou J. M., Wyche P. A. An examination of murine foreign body giant cells using cytochemical techniques and thin layer chromatography. J Pathol. 1974 Oct;114(2):75–83. doi: 10.1002/path.1711140204. [DOI] [PubMed] [Google Scholar]
- Parakkal P., Pinto J., Hanifin J. M. Surface morphology of human mononuclear phagocytes during maturation and phagocytosis. J Ultrastruct Res. 1974 Aug;48(2):216–226. doi: 10.1016/s0022-5320(74)80078-x. [DOI] [PubMed] [Google Scholar]
- Perper R. J., Sanda M., Stecher V. J., Oronsky A. L. Physiologic and pharmacologic alterations of rat leukocyte chemotaxis (Cx) in vivo. Ann N Y Acad Sci. 1975 Jun 13;256:190–209. doi: 10.1111/j.1749-6632.1975.tb36047.x. [DOI] [PubMed] [Google Scholar]
- Quie P. G. Pathology of bactericidal power of neutrophils. Semin Hematol. 1975 Apr;12(2):143–160. [PubMed] [Google Scholar]
- Reid J. D., Mackay J. B. The role of delayed hypersensitivity in granulomatous reactions to mycobacteria. 2. Reactions to intradermal injections of intact and disintegrated organisms. Tubercle. 1967 Jun;48(2):109–113. doi: 10.1016/s0041-3879(67)80005-9. [DOI] [PubMed] [Google Scholar]
- Rocklin R. E. Clinical applications of in vitro lymphocyte tests. Prog Clin Immunol. 1974;2:21–67. [PubMed] [Google Scholar]
- Roth S. A molecular model for cell interactions. Q Rev Biol. 1973 Dec;48(4):541–563. doi: 10.1086/407816. [DOI] [PubMed] [Google Scholar]
- Rothwell T. L., Spector W. G. The effect of neonatal and adult thymectomy on the inflammatory response. J Pathol. 1972 Sep;108(1):15–21. doi: 10.1002/path.1711080103. [DOI] [PubMed] [Google Scholar]
- Ryan G. B., Spector W. G. Natural selection of long-lived macrophages in experimental granulomata. J Pathol. 1969 Oct;99(2):139–151. doi: 10.1002/path.1710990208. [DOI] [PubMed] [Google Scholar]
- SHELLEY W. B., HURLEY H. J., Jr Experimental sarcoid reactions in human skin. Am Rev Respir Dis. 1961 Nov;84(5):45–48. doi: 10.1164/arrd.1961.84.5P2.45. [DOI] [PubMed] [Google Scholar]
- SUTER E. Interaction between phagocytes and pathogenic microorganisms. Bacteriol Rev. 1956 Jun;20(2):94–132. doi: 10.1128/br.20.2.94-132.1956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SUTER E., RAMSEIAR H. CELLULAR REACTIONS IN INFECTION. Adv Immunol. 1964;27:117–173. doi: 10.1016/s0065-2776(08)60707-5. [DOI] [PubMed] [Google Scholar]
- Shima K., Dannenberg A. M., Jr, Ando M., Chandrasekhar S., Seluzicki J. A., Fabrikant J. I. Macrophage accumulation, division, maturation, and digestive and microbicidal capacities in tuberculous lesions. I. Studies involving their incorporation of tritiated thymidine and their content of lysosomal enzymes and bacilli. Am J Pathol. 1972 Apr;67(1):159–180. [PMC free article] [PubMed] [Google Scholar]
- Simon H. B., Sheagren J. N. Cellular immunity in vitro. I. Immunologically mediated enhancement of macrophage bactericidal capacity. J Exp Med. 1971 Jun 1;133(6):1377–1389. doi: 10.1084/jem.133.6.1377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slauson D. O., Dahlstrom M. A. The pulmonary inflammatory response. Cellular events in experimental pulmonary arterial hypersensitivity disease. Am J Pathol. 1975 Apr;79(1):119–130. [PMC free article] [PubMed] [Google Scholar]
- Smith J. B., McIntosh G. H., Morris B. The migration of cells through chronically inflamed tissues. J Pathol. 1970 Jan;100(1):21–29. doi: 10.1002/path.1711000104. [DOI] [PubMed] [Google Scholar]
- Snyderman R., Shin H., Dannenberg A. M., Jr Macrophage proteinase and inflammation: the production of chemotactic activity from the fifth complement by macrophage proteinase. J Immunol. 1972 Oct;109(4):896–898. [PubMed] [Google Scholar]
- Spector W. G., Heesom N., Stevens J. E. Factors influencing chronicity in inflammation of rat skin. J Pathol Bacteriol. 1968 Jul;96(1):203–213. doi: 10.1002/path.1700960121. [DOI] [PubMed] [Google Scholar]
- Spector W. G., Heesom N. The production of granulomata by antigen-antibody complexes. J Pathol. 1969 May;98(1):31–39. doi: 10.1002/path.1710980105. [DOI] [PubMed] [Google Scholar]
- Spector W. G. The granulomatous inflammatory exudate. Int Rev Exp Pathol. 1969;8:1–55. [PubMed] [Google Scholar]
- Spector W. G., Walters M. N., Willoughby D. A. The origin of the mononuclear cells in inflammatory exudates induced by fibrinogen. J Pathol Bacteriol. 1965 Jul;90(1):181–192. doi: 10.1002/path.1700900119. [DOI] [PubMed] [Google Scholar]
- Stewart C. C., Lin H. S., Adles C. Proliferation and colony-forming ability of peritoneal exudate cells in liquid culture. J Exp Med. 1975 May 1;141(5):1114–1132. doi: 10.1084/jem.141.5.1114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sutton J. S., Weiss L. Transformation of monocytes in tissue culture into macrophages, epithelioid cells, and multinucleated giant cells. An electron microscope study. J Cell Biol. 1966 Feb;28(2):303–332. doi: 10.1083/jcb.28.2.303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsuda T., Dannenberg A. M., Jr, Ando M., Rojas-Espinosa O., Shima K. Enzymes in tuberculous lesions hydrolyzing protein, hyaluronic acid and chondroitin sulfate: a study of isolated macrophages and developing and healing rabbit BCG lesions with substrate film techniques; the shift of enzyme pH optima towards neutrality in "intact" cells and tissues. J Reticuloendothel Soc. 1974 Oct;16(4):220–231. [PubMed] [Google Scholar]
- Turk J. L., Bryceson A. D. Immunological phenomena in leprosy and related diseases. Adv Immunol. 1971;13:209–266. doi: 10.1016/s0065-2776(08)60185-6. [DOI] [PubMed] [Google Scholar]
- UNGAR J., MUGGLETON P. W. The production of experimental granulomatous lesions induced by injections of fatty acids and fractions of tubercle bacilli. Am Rev Respir Dis. 1961 Nov;84(5):76–80. doi: 10.1164/arrd.1961.84.5P2.76a. [DOI] [PubMed] [Google Scholar]
- Unanue E. R., Benacerraf B. Immunologic events in experimental hypersensitivity granulomas. Am J Pathol. 1973 Jun;71(3):349–364. [PMC free article] [PubMed] [Google Scholar]
- Unanue E. R. The regulatory role of macrophages in antigenic stimulation. Adv Immunol. 1972;15:95–165. doi: 10.1016/s0065-2776(08)60684-7. [DOI] [PubMed] [Google Scholar]
- Von Lichtenberg F., Smith T. M., Lucia H. L., Doughty B. L. New model for schistosome granuloma formation using a sluble egg antigen and bentonite particles. Nature. 1971 Jan 15;229(5281):199–200. doi: 10.1038/229199a0. [DOI] [PubMed] [Google Scholar]
- Wanstrup J., Christensen H. E. Sarcoidosis. 1. Ultrastructural investigations on epithelioid cell granulomas. Acta Pathol Microbiol Scand. 1966;66(2):169–185. doi: 10.1111/apm.1966.66.2.169. [DOI] [PubMed] [Google Scholar]
- Ward P. A. Complement-dependent phlogistic factors in rheumatoid synovial fluids. Ann N Y Acad Sci. 1975 Jun 13;256:169–176. doi: 10.1111/j.1749-6632.1975.tb36045.x. [DOI] [PubMed] [Google Scholar]
- Ward P. A. Leukotaxis and leukotactic disorders. A review. Am J Pathol. 1974 Dec;77(3):520–538. [PMC free article] [PubMed] [Google Scholar]
- Werb Z., Gordon S. Elastase secretion by stimulated macrophages. Characterization and regulation. J Exp Med. 1975 Aug 1;142(2):361–377. doi: 10.1084/jem.142.2.361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Werb Z., Gordon S. Secretion of a specific collagenase by stimulated macrophages. J Exp Med. 1975 Aug 1;142(2):346–360. doi: 10.1084/jem.142.2.346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- White R. G. Granuloma formation and its relation to chemotaxis. A discussion of the role of two groups of micro-organisms: the mycobacteria and the anaerobic coryneforms. Antibiot Chemother (1971) 1974;19:382–408. [PubMed] [Google Scholar]
- Wiener E. DNA-synthesis in peritoneal mononuclear leucocytes. Exp Cell Res. 1967 Feb;45(2):450–459. doi: 10.1016/0014-4827(67)90193-0. [DOI] [PubMed] [Google Scholar]
- Williams D., Williams W. J., Williams J. E. Enzyme histochemistry of epithelioid cells in sarcoidosis and sarcoid-like granulomas. J Pathol. 1969 Apr;97(4):705–709. doi: 10.1002/path.1710970416. [DOI] [PubMed] [Google Scholar]
- Williams W. J., Fry E., James E. M. The fine structure of beryllium granulomas. Acta Pathol Microbiol Scand Suppl. 1972;233:195–202. [PubMed] [Google Scholar]
- Williams W. J., James E. M., Erasmus D. A., Davies T. The fine structure of sarcoid and tuberculous granulomas. Postgrad Med J. 1970 Aug;46(538):496–500. doi: 10.1136/pgmj.46.538.496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Furth R., Cohn Z. A., Hirsch J. G., Humphrey J. H., Spector W. G., Langevoort H. L. The mononuclear phagocyte system: a new classification of macrophages, monocytes, and their precursor cells. Bull World Health Organ. 1972;46(6):845–852. [PMC free article] [PubMed] [Google Scholar]
- van Furth R. Origin and kinetics of monocytes and macrophages. Semin Hematol. 1970 Apr;7(2):125–141. [PubMed] [Google Scholar]
- von Heyden H. W., von Heyden D. Characteristics of macrophages in vitro derived from peripheral blood cells. Blut. 1974 Jul;29(1):37–42. doi: 10.1007/BF01631579. [DOI] [PubMed] [Google Scholar]


