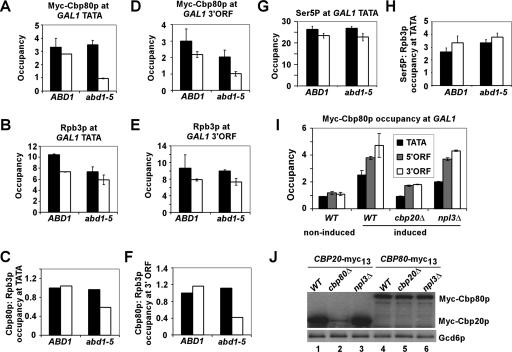
FIG. 3.
Optimal Cbp80p recruitment to GAL1 requires the cap guanine 7-methyltransferase Abd1p. (A to H) Isogenic CBP80-myc strains CMY015 (ABD1) and CMY016 (abd1-5) were cultured under the same conditions described for Fig. 2 for galactose induction and heat treatment, and ChIP analysis was conducted as described for Fig. 1 using antibodies to Myc (to detect Myc-Cbp80p) (A and D) or Rpb3p (B and E). Occupancies of Myc-Cbp80p were normalized to those for Rpb3p for the GAL1 TATA (C) and GAL1 3′ ORF (F). ChIP analysis of the same samples was conducted using H14 antibodies specific for the Ser5-phosphorylated CTD (Ser5P) (G). Occupancies of Ser5P were normalized to those for Rpb3p for the GAL1 TATA in panel B (H). Optimal Cbp80p recruitment to GAL1 requires Cbp20p but not Npl3p. (I) Isogenic CBP80-myc strains CMY006 (WT), CMY011 (cbp20Δ), and CMY012 (npl3Δ) were cultured under the conditions described for Fig. 2 for galactose induction except that cells were grown at 30°C, and ChIP analysis was conducted as described for Fig. 1 with the use of antibodies to Myc (to detect Myc-Cbp80p). The average results obtained from two independent cultures and two PCR amplifications for each culture were plotted in the histograms with standard errors shown as error bars. (J) Western blot analysis of Myc-Cbp20p and Myc-Cbp80p expression. Whole-cell extracts of isogenic CBP20-myc strains CMY005 (WT), CMY009 (cbp80Δ), and CMY010 (npl3Δ) and CBP80-myc strains CMY006 (WT), CMY011 (cbp20Δ), and CMY012 (npl3Δ) were subjected to Western blot analysis using antibodies to Myc (to detect Myc-Cbp20p or Myc-Cbp80p) or Gcd6p (loading control).