Abstract
Synopsis
In insomnia, which is a very common sleep disorder, objective sleep measures, EEG activity, physiologic findings, HPA axis activity and inflammation markers suggest that it is not a state of sleep loss, but a disorder of hyperarousal present both during the night and the daytime. Several psychological and physiological factors contribute to the onset and perpetuation of insomnia, such as anxious-ruminative personality traits, stressful events, age-related sleep homeostasis weakening mechanisms, menopause and biologic – genetic diathesis of CNS hyperarousal. The therapeutic approach in insomnia should be multidimensional reducing the overall emotional and physiologic hyperarousal and its underlying factors present throughout the 24-h sleep/wake period.
Insomnia: Prevalence and Definitions
Insomnia is considered to be the most common sleep disorder. Its prevalence varies considerably based on the definition used. While one-fourth to one-third of the general population reports a complaint of difficulty falling and / or staying asleep [1-9], about 10% present chronic complaints and seek medical help for insomnia. [10, 11] However, insomnia has always been and still is an under-recognized and therefore under-treated problem, since about 60% of the people suffering from insomnia never talk to their physicians about their sleeping difficulties. [12-13] The inadequate identification and treatment of insomnia has significant medical and public health implications. Chronic insomnia results to impaired occupational performance and affects quality of life. [14-15]
Insomnia, according to the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision (DSM-IV TR), is defined as the complaint of difficulty initiating or maintaining sleep, early awakening, and interrupted or non-restorative sleep. Additionally, nighttime symptoms are accompanied by clinically significant impairment in daytime function, for which no identifiable cause i.e. other sleep disorder, psychiatric disorder, medical condition, is attributed. [16] The significance of insomnia is determined by its severity, frequency, duration and the daytime function impairment.[17-18] A presentation of insomnia for three or more times weekly is evaluated as clinically significant. Furthermore, the duration of a month or less is called transient insomnia and is frequently triggered by external stressors. Transient insomnia would usually be self-resolved after the person is adjusted to the stressful event or the events are resolved. Symptoms that last between one and six months are classified as sub-acute insomnia, whereas if they persist for more than six months, then they are classified as chronic insomnia. [16] In regard to impairment of daytime functioning there is no objective way to measure it and only subjective reports can indicate its significance.
Several factors, such as gender, age, socioeconomic status have been associated with the prevalence of insomnia. Insomnia occurs more frequently in women, both in terms of symptoms reported and of daytime consequences.[19] Menopause has been proposed as an explanation for this difference among mid-aged men and women but other underlying chronic conditions, e.g. increased prevalence of depression in women, seem to be also significant. Aging is another significant factor associated with increased prevalence of insomnia, present in up to 50% of people over 65 years old. In terms of socioeconomic status, insomnia is more frequent among individuals who are separated, divorced or widowed, especially in women, or have a lower educational / economical status or are unemployed. [19]
Etiologic factors and pathophysiologic models
1. The role of emotional stress and psychiatric disorders
The emotional factors involved in the development and persistence of insomnia have been assessed in numerous studies investigating stressful life events, personality patterns, and psychiatric diagnoses.
Stressful life events are closely associated with the onset of chronic insomnia and are mediated by certain predisposing personality factors. Insomniacs compared to controls, tend to be more discontent, both as children and as adults, have less satisfying interpersonal relations, and relatively poor self concepts, leading to inadequate coping mechanisms for dealing with stress.[20]
Minnesota Multiphasic Personality Inventory (MMPI) personality profiles in insomnia demonstrate a high specificity and consistency among patients. Patients with chronic insomnia exhibit high levels of psychopathology expressed by high scores in the depression (D), psychasthenia (Pt) and hysteria (Hy) scales of the MMPI. Therefore, the specific personality traits for insomniacs are depressed mood, rumination, chronic anxiety, inhibition of emotions, and inability to express anger.[21]
Insomnia is known to be more frequent in people with underlying psychiatric disorders. Insomnia due to a mental disorder, is the most common diagnosis among those who seek medical evaluation / treatment in sleep disorders' centers. [22] Among mental disorders associated with insomnia, the most common ones are mood disorders, such as dysthymic disorder, major depression disorder, bipolar disorder, cyclothymic disorder, as well as most anxiety disorders, and substance abuse disorders. [14,19, 22-23] In this case, when the patient's history is taken the course of the insomnia symptoms correlates to the course of the underlying mental disorder. Insomnia may be the first manifestation of the underlying mental disorder, and patients frequently tend to attribute their mental problems to poor sleep. [24] Therefore, in some cases it may be challenging for the physician to determine if insomnia is a manifestation of some mood or anxiety disorder, or if it is a cause for depressive and anxiety symptoms.
2. The role of primary sleep disorders
In the past, there have been reports claiming that other primary sleep disorders, such as sleep apnea and nocturnal myoclonus, are common causes of insomnia. However, large, well-controlled studies have shown that neither sleep apnea, nor nocturnal myoclonus, are more prevalent among insomniacs. [25-27] As a result, primary sleep disorders do not appear to be causative factors for insomnia.
3. The role of medical disorders and substance use
Medical conditions associated with chronic pain (e.g. arthritis, back pain), cardiovascular disease (heart failure), respiratory diseases (COPD, asthma), gastrointestinal disorders (duodenal ulcer), chronic renal insufficiency, endocrine conditions (thyroid dysfunction), and neurologic conditions (Parkinson's disease, headaches) are known to be associated with chronic insomnia. [20]
Also, smoking, alcohol, use of drugs, such as b-blockers, some SSRIs, some neuroleptics, amphetamines, and drug withdrawal (benzodiazepine, non-diazepine hypnotics) can cause insomnia. [19, 20]
4. The “Internalization Hypothesis” Model
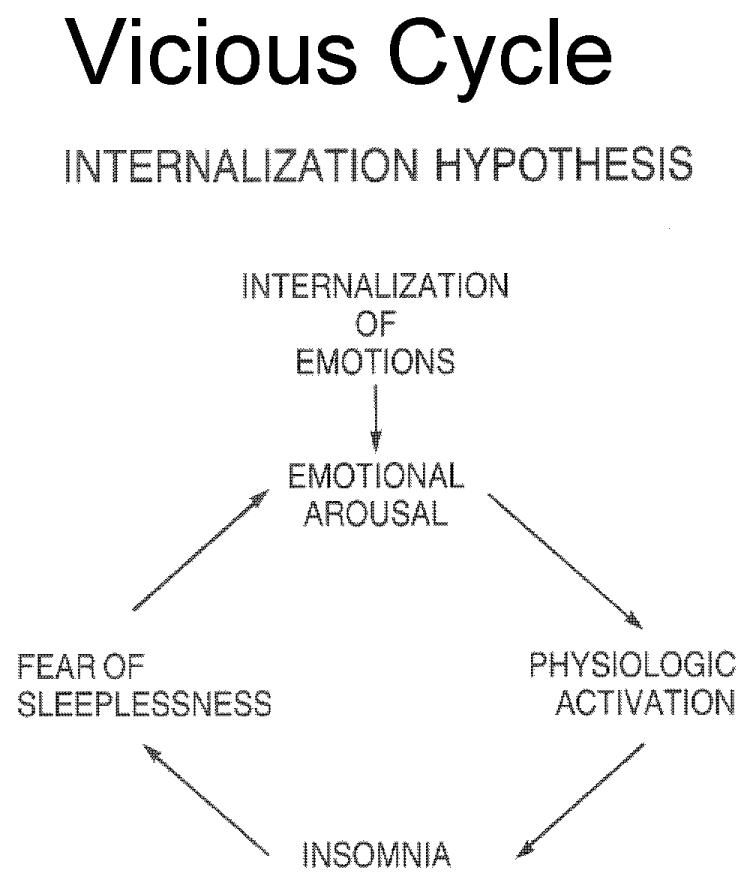
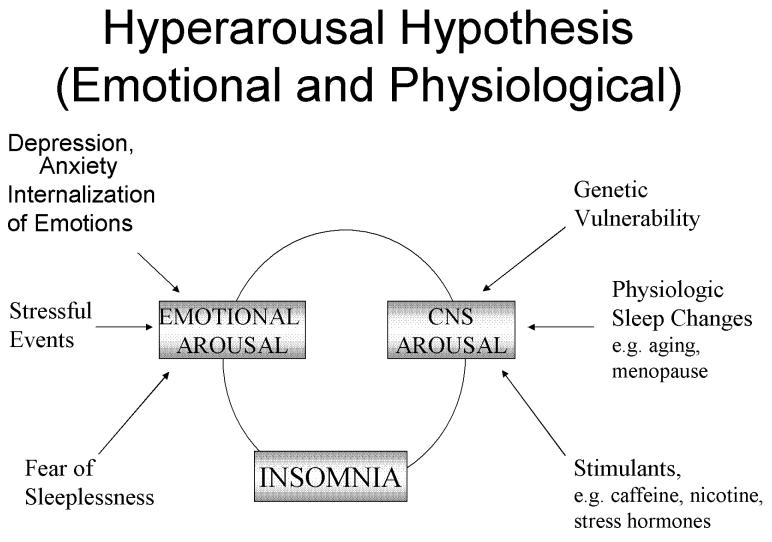
Based on clinical and psychometric studies that showed that insomnia is associated with inhibited and repressive personality traits[28], in early 1980's Kales proposed that patients with chronic insomnia are in a state of constant emotional arousal resulting to a physiological activation [20], such as increased heart rate, peripheral vasoconstriction, elevated rectal temperature and increased body movements before sleep initiation. [29-32] They generally tend to handle stress and conflicts through internalization of emotions, which leads to emotional arousal. At bedtime, they are characteristically tense, anxious, ruminative about issues associated with health, work, personal affairs, death etc. Since the emotional arousal causes physiological arousal, they face difficulties in sleep initiation or returning to sleep after awakenings during the night. As a result, they develop a ‘“fear of sleeplessness”, which intensifies their emotional arousal perpetuating their insomnia. In this model stressful life events are important in the initiation of the vicious cycle of emotional and physiologic arousal[figure 1].[20, 29] A similar model of pathogenesis of insomnia, known as the “3-P” model, was proposed by Spielmann [33-34], suggesting three distinct elements for the onset and course of insomnia: predisposing characteristics (i.e. acquired or inherited characteristics that render individuals more susceptible to develop a particular type of insomnia), precipitating events , such as health problems, family/ marital difficulties, work/ school stress and perpetuating attitudes and practices (i.e. behavioral practices that maintain sleep difficulty, mental worrying about sleeplessness or misconception about sleep). According to this model, insomnia may become independent of its origin over time.
Figure 1.
Internalization Hypothesis
5. The “Hyperarousal” Model
In the past, specialists of Sleep Medicine driven to a large extent by the chief complaints of their insomniacs, have focused their efforts to improve the quality and quantity of their sleep. Insomniacs, report significantly shorter sleep duration compared to normal sleepers. This subjective report has been repeatedly confirmed objectively by polysomnographic studies in this population. [35-37] Therefore, pharmacologic and /or psychotherapeutic treatments aimed to extend the sleep duration. This approach led to the perception, that insomnia is a disorder of sleep loss.[35]
In this section we present data from different domains showing that insomnia is a condition of hyperarousal rather than sleep loss.
A. Daytime Sleepiness and Fatigue
The terms of “sleepiness” and “fatigue” have been used interchangeably both by patients and physicians. However, based on clinical experience and findings from experimental studies, we have proposed a separate definition for these two terms.[38] Sleepiness is a subjective feeling of physical and mental tiredness associated with increased sleep propensity. Fatigue, on the other hand, is also a subjective feeling of physical and/or mental tiredness; however it is not associated with increased sleep propensity.
Daytime sleepiness measured in a objective way by the Multiple Sleep Latency Test (MSLT) is well known to be increased in individuals after sleep deprivation/ sleep loss, compared to non-deprived individuals.[39-40] However, insomniacs, in spite their subjective complaints of daytime fatigue, as well as their significantly less nocturnal sleep, do not show increased sleepiness as compared to normal sleepers. On the contrary, they are significantly more alert than normal sleepers, as shown by their longer sleep latencies compared to controls in the MSLT, suggesting that insomnia is a condition of 24h hyperarousal rather than a nighttime disorder. [36-37,41]
Insomniacs' frequent complaints of daytime fatigue have been verified in studies that fatigue was measured by scales, such as the Fatigue Severity Scale (FSS) and the Fatigue Scale of the Profile of Mood Sates (POMS). All studies showed that fatigue scores in insomniacs were elevated compared to normal sleepers. [42]
B. Physiologic changes
It has been shown that insomniacs compared to normal sleepers have significantly increased rectal temperature, heart rate, vasoconstriction, and increased skeletal muscles movements before and during sleep. [30,43,44]
On the contrary, in sleep deprivation, findings regarding physiological activation are inconsistent. While some studies report increased body temperature, others show minimal changes or even decrease of the temperature after sleep restriction.[45] Similarly, heart rate following sleep deprivation in some studies is reported as decreased, whereas in others it does not change. [46-47]
C. Quantitative EEG findings
Transition from wakefulness to sleep in normal sleepers is characterized by a decrease of high frequency and an increase of slow frequency activity in the EEG. [48] However, it has been shown that insomniacs seem to present different EEG patterns compared to normal sleepers, consistent with hyperarousal, both before sleep initiation and during sleep.
Specifically, during wakefulness at the sleep onset period, studies have shown that insomniacs have an elevated relative beta power, which is a high frequency EEG activity, while delta power – slow EEG activity- is decreased. [49-50] Additionally, in insomniacs compared to normal individuals, levels of alpha power are reduced during wakefulness, and remain rather constant at the sleep onset period. [49-50]
During NREM sleep, insomniacs present increased levels of beta and gamma power EEG activity – high frequencies-, not sharp decrease of alpha as usually seen in normal sleepers, and a decrease of theta and delta power. [49-52] During REM sleep, beta and alpha power are increased, while there is a deficit of theta and delta power. [52]
D. Neuroimaging
Functional neuroimaging studies have shown that transition from wakefulness to sleep, is associated with a decrease of brain activity in specific regions, such as the brain stem, the thalamus and the prefrontal cortex.[53] After sleep deprivation metabolic rates in the thalamus, frontal and parietal cortex are decreased and decreases seem to correlate with impaired cognition and greater fatigue. [54]
Fluorodeoxyglucose (F18) positron emission tomography (PET) was used to assess regional cerebral glucose metabolism in insomniacs vs. normal sleepers. Unlike after sleep deprivation, patients with insomnia showed elevation of the global cerebral metabolism during sleep compared to normal sleepers, consistent with a state of hyperarousal during their sleep. Furthermore, while shifting from wakefulness to sleep states, insomniacs had a smaller decline in areas that promote wakefulness compared to normal sleepers, whereas during wakefulness metabolic rate in the prefrontal, and not frontal as in sleep deprivation, regions is reduced. [55] As a result, it was suggested that elevated brain metabolism can explain subjectively reported disturbed sleep in insomniacs. Failure of the arousal system to decrease its activity, while shifting from wakefulness to sleep states, may explain the difficulty in sleep initiation documented in insomnia. Finally, decreased activity of prefrontal brain regions during wakefulness could be associated with daytime fatigue observed in insomniacs. [55]
E. Neurocognitive function
Insomniacs also complain of cognitive deficits, such as concentration, alertness and memory. Several studies focused on cognitive functions in insomniacs comparing them to normal sleepers, have failed to propose a definable and consistent pattern of dysfunction in the first group, both across studies and functions. While there is a few data pointing to the direction of a possible reduced attention span and vigilance performance, measures of alertness, focused, divided and sustained attention did not show consistent differences among the two groups. Furthermore, studies on verbal functions, concept formation, reasoning, and executive functions have inconsistent results and need to be further explored. [56]
Overall, most comparisons do not support significant daytime deficits in cognition and psychomotor performance in patients with insomnia. In the contrary, several studies in normal sleepers after sleep deprivation, consistently show that psychomotor performance is decreased and daytime napping may have a beneficial effect on it. [39,40, 57]
F. Hypothalamic –Pituitary –Adrenal (HPA) Axis
Stress has been associated with the activation of the Hypoathalamic- pituitary- adrenal (HPA) axis, while CRH and cortisol, products of the hypothalamus and the adrenals, respectively, are known to cause arousal and sleeplessness to humans and animals. On the other hand, sleep and particularly deep sleep, has an inhibitory effect on the stress system, including its two major components: the HPA axis and the sympathetic system.
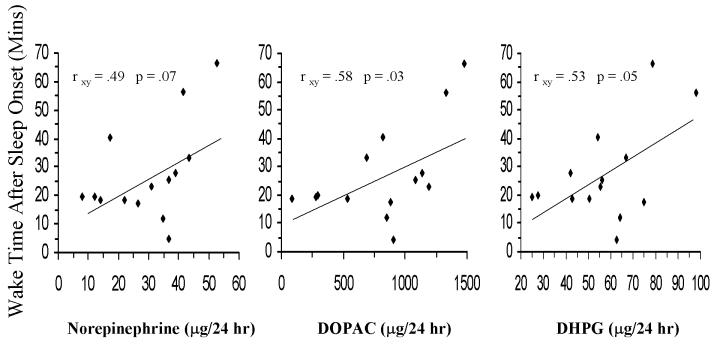
In the past, few studies have assessed cortisol levels in insomniacs and their results were inconsistent. The majority of these studies reported no difference between “poor” sleepers and normal individuals in the levels of 24-h cortisol and 17-hydorxysteroid excretion.[58] A more recent preliminary study investigated possible association between chronic insomnia and the activity of the stress system by measuring urinary free cortisol (UFC), catecholamines and Growth Hormone (GH) and found that 24-h UFC levels in insomniacs were positively correlated with the Total Wake Time (TWT) of the patients. Furthermore, catecholamine metabolites, DHPG and DOPAC were positively correlated with the percent of stage 1 (%ST1) of sleep and the Wake Time After Sleep Onset (WTASO), while norepinephrine tended to be significantly positively associated with %ST1 and WTASO and negatively associated with Slow Wave Sleep (SWS).[figure 2] GH was detectable in a small percent of the participants, suggesting that GH axis may be suppressed in chronic insomniacs. [59] These findings were further supported by a controlled study, which found that circulating levels of norepinephrine during the nocturnal period were elevated in patients with primary insomnia compared to patients with major depression and normal controls.[60]
Figure 2.
Correlation between wake time after sleep onset with 24-hr urinary NE, DHPG, and DOPAC
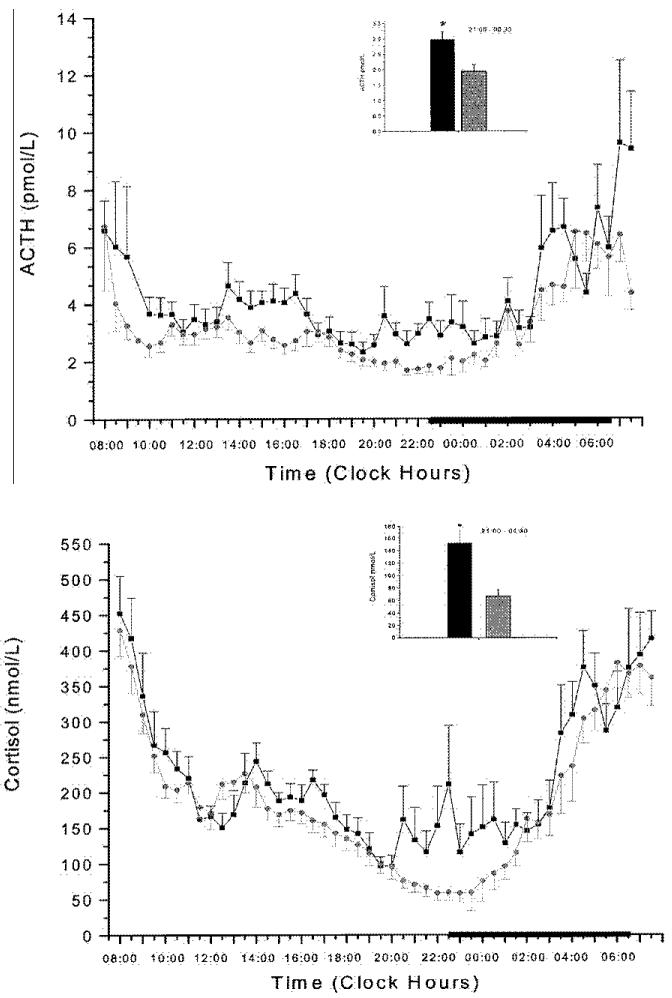
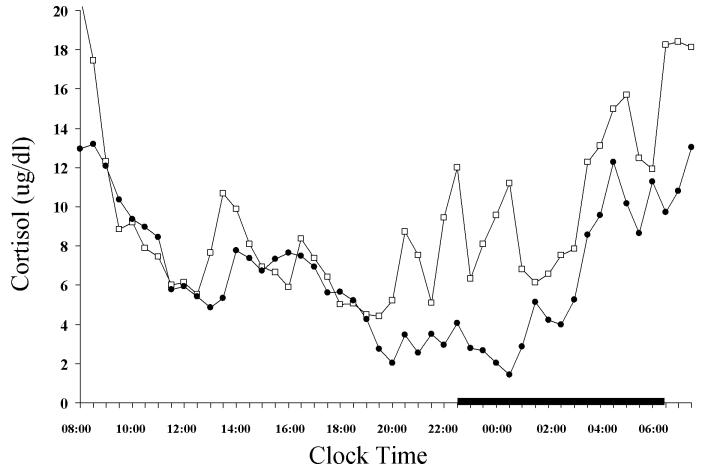
In a following controlled study 24-h serial ACTH and cortisol levels were significantly higher in insomniacs compared to normal sleepers.[31][figure 3] Within the 24-h period the greatest elevations were observed in the evening and during the first half of the night. Furthermore, within the group of insomniacs, the sub-group with high degree of objective sleep disturbance [percent of total sleep time (%TST) <70] had higher amount of cortisol compared to the subgroup with low degree of sleep disturbance.[figure 4] Finally, pulsatile analysis revealed a significantly higher number of ACTH and cortisol pulses in insomniacs compared to normal sleepers, while cosinor analysis indicated a significant circadian rhythm without differences in the temporal pattern of ACTH and cortisol secretion between the two groups. Conclusively, this study suggests that insomnia is associated with an overall 24h increase of the ACTH and cortisol secretion, which however retains a normal circadian pattern. Therefore, insomnia seems to be a disorder of 24-h hyperarousal rather than sleep loss. This approach is further supported by studies investigating the effects of sleep loss on cortisol secretion. Most of the studies have found no change [61-63], or decrease in the secretion of cortisol [39,64] after sleep deprivation, whereas in these studies that reported significant increase of cortisol in the evening following the night of sleep loss[65-66], sleep deprivation was associated with rather stressful experimental conditions, i.e. lying in bed in a dimly lit room and receiving calories through an iv catheter.
Figure 3.
24-hr plasma ACTH (top) and cortisol (bottom) concentrations in insomniacs (■) and controls (○). The thick black line on the abscissa indicates the sleep recording period. Error bar indicates SE. *P<0.01
Figure 4.
24-hr circadian secretory pattern of cortisol in insomniacs with low TST (□) and high TST(●).The thick black line on the abscissa indicates the sleep recording period. Error bar indicates SE. *P<0.05
Finally, a study that investigated the effects of the tricyclic antidepressant doxepin on nocturnal sleep and plasma cortisol levels in patients with primary insomnia showed that doxepin improved sleep and reduced mean cortisol levels, suggesting that beneficial effects of the medication are at least partially mediated by the normalization of the HPA axis. [67]
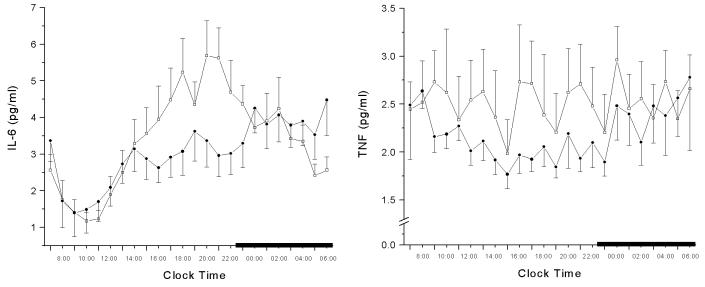
G. Proinflammatory cytokines in insomnia
Proinflammatory cytokines, Interleukin-6 (IL-6) and Tumor Necrosis Factor a (TNFa) are fatigue-inducing cytokines negatively influenced by the quantity and quality of sleep. In a study where levels of IL-6 and TNFa were compared between insomniacs and normal sleepers, it was found that, although mean 24-h cytokine's secretions were not different between the two groups, there was a significant increase of IL-6 from mid-afternoon to evening. Furthermore, cosinor analysis of IL-6 levels,showed a significant shift of the peak secretion from nighttime to evening in the insomnia group. [38] [figure 5] Moreover, the characteristic circadian TNFa secretion with a peak close to the sleep offset, that was found in the normal sleepers, was not present in the insomniacs. However, insomniacs' TNFa secretion was characterized by a regular 4 h rhythm, pattern not present in the normal sleepers group. The hypersecretion and / or circadian alteration of the cytokines secretion associated with a HPA axis activation may explain the fatigue and poor sleep associated with insomnia. In contrast, hypersecretion and / or circadian alteration of the cytokines, that is not associated with HPA axis activation, i.e. sleep deprivation in normal sleepers, may explain their true sleepiness and poor sleep. [36,68]
Figure 5.
24-hr circadian secretory pattern of IL-6 (left) and TNFa (right) in insomniacs (□) and controls (●). The thick black line on the abscissa indicates the sleep recording period. Error bar indicates SE. *P<0.05
In conclusion, data from studies on objective sleep, EEG activity, physiologic findings, neuroimaging, neuro-cognitive function and HPA axis and inflammation markers suggest that insomnia is not a state of sleep loss, but a disorder of hyperarousal present both during the night and the daytime.
H. Physiologic changes leading to hyperarousal and insomnia: aging and menopause
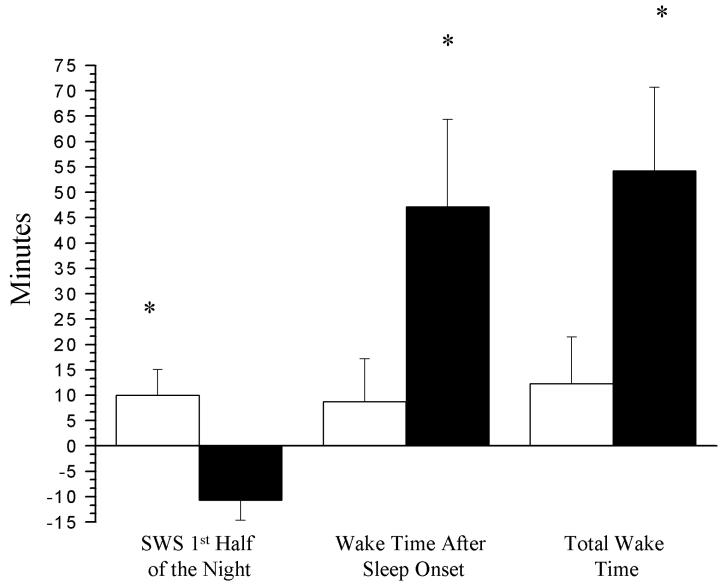
The emphasis on the role of psychologic characteristics, such as certain personality traits and inadequate coping mechanisms, as vulnerability factors increasing the susceptibility for insomnia, might have led to a relative neglect of physiologic changes that may underlie the pathogenesis of insomnia. Recently, studies have suggested that physiologic factors, such as aging, can also increase the vulnerability to insomnia. One study that examined the effects of exogenous administration of CRH- a hormone with an arousing/waking effect- on middle-aged v.s. young men, found that wakefulness increased significantly only in the middle-aged group.[69] This increase was more pronounced during the first half of the night. Moreover, CRH administration caused a significant reduction of SWS only in the middle-aged group.[figure 6] These findings suggest that middle-aged men compared with the young, are more vulnerable to the arousing effects of CRH. Therefore, the increased prevalence of insomnia in middle age may be the result of deteriorating sleep mechanisms associated with increased sensitivity to arousal-producing stress hormones rather than to increased life stresses during this period.
Figure 6.
Comparisons of changes in wakefulness and SWS from baseline (nights 2 and 3) to the night of CRH administration (night 4) between young (□) and middle-aged (■) healthy men after adjusting for baseline value. Values represent the least square mean ± SE. *, P<0.05
Disturbed sleep also is a common complaint of women entering the menopause. A recent study investigating the objective sleep patterns associated with menopause and hormonal replacement therapy (HRT) in a large population sample, found that while sleep latency (SL) among premenopausal women and postmenopausal women on HRT did not differ, it was significantly increased among postmenopausal women without HRT. [70] Furthermore, postmenopausal women without HRT were less likely to have slow wave sleep (SWS) compared to those with HRT. Conclusively, hormonal changes during menopause appear to correlate with increased SL, and decreased SWS. These objective findings support the increased frequency of complaints of poor sleep reported by postmenopausal women, as well as the symptomatic relief reported by those on HRT. [71]
I. Physiologic differences between insomnia with depression vs. depression alone
Insomnia is very frequently associated with depression. However, there is evidence that a primary complaint of insomnia associated with depression is different from depression without insomnia in terms of its pathophysiology. In chronic insomnia with depression, the primary polysomnographic variables affected are sleep efficiency measures [72], whereas in depression both sleep efficiency and REM sleep variables change. [73] Furthermore, in chronic insomnia cortisol secretion is related to indices of sleep disturbance, such as Total Wake Time (TWT) and percent of stage 1 sleep[31,59], whereas in depression hypercortisolism is related to REM sleep variable. [74] The degree of psychological distress in insomnia correlates with the objective sleep disturbance[20], while in depression without insomnia there is a dissociation between depth of depression and sleep abnormalities.[73] Finally, while sleep deprivation does not have a beneficial effect on mood in insomniacs with depression, it is mood elevating in depression without insomnia.[20] In conclusion, based on these preliminary observations, insomnia with depression and depression alone appear to be two distinct entities. Further studies are needed to understand the complex association between insomnia and depression.
All the above studies combined suggest that sleep loss is not the cause but only one of the manifestations of insomnia. It appears that (1) central nervous system hyperarousal, either as pre-existing, and/or induced by psychiatric pathology and deteriorated by stressful events, (2) specific personality traits and inadequate coping mechanisms, and (3) aging- and menopause-related physiological decline of sleep mechanisms, are at the core of this common sleep disorder.[figure 7]
Figure 7.
Heuristic Model of Psychologic and Physiologic Factors associated with Insomnia
6. Clinical Implications: Evaluation and Treatment
I. Evaluation
Chronic insomnia is a difficult disorder to treat and the importance of a multidimensional evaluation that includes thorough sleep history, medical history, physical examination and drugs / substances use, as well as psychiatric assessment has been stressed for many years, although frequently overlooked in daily practice. [20]
The data reviewed in the “etiologic factors and pathophysiologic models” section of this chapter, call for an emphasis in elements of the evaluation that should be investigated in the diagnostic work-up of an insomniac patient. It was shown that middle age and menopause are associated with sleep changes that make the subject more vulnerable to the effects of physical or emotional stress. For example, the use of beverages with stimulant substances, i.e. caffeine, will be associated with more sleep disturbances in a middle- aged or old individual compared to a young individual. Also, a post-menopausal woman would be expected to be more vulnerable in terms of sleep disturbance to life-related stressors, i.e. family difficulties, professional pressures etc. Moreover, eliciting information about the use or not of HRT in menopausal women, may explain differences in terms of sleep complaints in post-menopausal women. These physiological differences may also explain, why depression, a disorder with increased CRH and cortisol production, in middle age is associated with insomnia, whereas in the young is associated with hypersomnia. [75-76]
A frequent complaint of insomniacs is that they are tired during the day and they cannot meet the daily demands. The complaint of daytime fatigue should be carefully differentiated from the complaint of EDS. An easy way to differentiate these two symptoms is by asking the patient how likely it is for him/ her to fall asleep during the day, if given the opportunity. While for insomniacs, who frequently complain of fatigue, it is rather unlikely to nap, patients with disorders of excessive daytime sleepiness, such as narcolepsy or sleep apnea, are very likely to fall asleep during the day, especially if circumstances allow.
Psychologic factors are known to play a major role in the development of insomnia. Although insomnia is frequently associated with psychiatric disorders, it should be emphasized that in many cases psychopathology is subtle. The evaluation of subtle psychopathology is important in those that they are genetically or physiologically vulnerable to sleep disturbances, such as patients with a family history of insomnia and depression, middle-aged or older individuals, and peri- and post-menopausal women. Therefore, even though criteria for a DSM-IV diagnosis may not be fulfilled, traits of depression, anxiety, rumination, obsessive-compulsiveness or hypochondriasis may be present. For example, it is important to ask these patients, if they worry, feel dissatisfied or disappointed, have low self esteem, are angry or irritable.
The association of insomnia particularly its most severe form, with HPA axis activation, raises the possibility that insomnia is associated with medical morbidity not previously recognized. For example, insomniacs may be at higher risk for hypertension, visceral obesity and metabolic syndrome and osteoporosis. Thus, an evaluation for these disorders, in addition to the evaluation of psychosomatic type of illnesses previously emphasized, should be incorporated into the history.
It has been previously suggested that sleep laboratory testing is not necessary in the evaluation and diagnosis of most insomniacs and that sleep laboratory measurements are of limited value in distinguishing insomniacs from normal sleepers.[72] Furthermore, the findings of one of our studies suggest that although polysomongraphic measurements may provide a reliable index of the biological significance and severity of chronic insomnia, they may not be necessary in making the diagnosis in individual patients. Indeed, the use of the sleep laboratory to predict severity of chronic insomnia is costly and impractical, given that other simpler methods, such as actigraphy, may provide information that is just as useful to the practicing physician.[31] Larger studies are needed to establish the clinical utility, i.e. severity of chronic insomnia, of objective measures of sleep time.
Since insomnia is associated with high cortisol levels, particularly in the evening, it is possible that evening measures of cortisol may be a useful marker of the biological severity of insomnia. Such a marker may affect our treatment strategies in managing an insomnia patient. However, further studies are needed to establish cortisol or other hormones, as biological markers of this chronic condition.
II. Treatment
Most sleep specialists advocate a multidimensional approach, including education about the multi-factorial etiology of the disorder, with emphasis on the biologic-genetic predisposition, psychological-stress issues, and weakening of the sleep mechanisms associated with aging and menopause.[20,24] Patients should be supported, but unrealistic expectations of “perfect” sleep should be avoided. General measures for improving sleep hygiene, such as minimal use of caffeine, cigarettes, other stimulants and alcohol, maintaining a regular sleep schedule going to bed only when sleepy, regular exercise and avoiding daytime naps, should be explained to the patients. Since insomnia is a disorder of 24-h hyperarousal and not simple sleep loss, the therapeutic goal should be not just to improve the quality and quantity of nighttime sleep, but to decrease the 24-h emotional and physiologic hyperarousal.
In counseling the insomniac, it is helpful to explain how anxiety participates in the vicious cycle that exacerbates and maintains the condition. Patients can be taught to reduce stress and anxiety by managing emotions more effectively through stress management techniques. Psychotherapeutic and behavioral modalities should target the emotional hyperarousal that insomniacs present, not just before sleep, but throughout the 24h sleep/wake period. Behavioral-cognitive techniques have become increasingly popular and are reported to be effective, particularly in elderly insomniacs. In some cases though, insight-oriented psychotherapy may be appropriate.[24]
Since insomnia is associated with HPA axis activation, the use of medications that down regulate the HPA axis, such as antidepressants, may be a promising tool among the pharmacologic approach. Furthermore, antidepressants in contrast to the classical benzodiazepine receptor agonists, are not associated with tolerance- withdrawal phenomena and are safer in insomnia, which is often a chronic, non-remitting disorder and therefore long-term use of medication is needed. The sedative qualities of the old tricyclics, as well as of the newer antidepressants, such as trazodone, mirtazapine, administered in relatively low doses at bedtime, appear to improve nighttime sleep by reducing the physiological and mental hyperarousal of the insomniacs. However, a disadvantage of sedative antidepressants, particularly the tricyclics, is their strong anticholinergic effects, that include dry mouth, blurred vision, orthostatic hypotension, constipation and cognitive impairment, particularly in the elderly.[35]
Based on the thesis that insomniacs are hyperalert/ fatigued during the day, pharmacological strategies that decrease the central nervous system hyperarousal during the day may be useful. For example, low doses of a benzodiazepine during the day may improve the sleep in chronic insomnia.
The finding that pro-inflammatory cytokines' IL-6 and TNFa daytime secretion is elevated in insomniacs, considering their role in subjective complaints of fatigue and poor performance, may lead to novel approaches to treat insomnia. There are unpublished reports that insomniacs use anti-inflammatory medications to fight fatigue and even improve their sleep. These reports are more frequent among elderly insomniacs. Particularly, in the elderly insomniacs, even if they do not have an active inflammatory disease, strategies to reduce low-grade inflammation by administration of sex steroids, decreasing fat through diet and exercise, and controlling adequately chronic pain and inflammation with non-steroid anti-inflammatory agents, may improve sleep, daytime alertness, and performance, which in tern, may decrease the risk of common diseases of old age, such as metabolic and cardiovascular problems, cognitive disorders, and osteoporosis.[35]
Summary
Insomnia is considered to be the most common sleep disorder. Several different approaches suggest that insomnia is a state of 24-h hyperarousal rather than sleep loss : insomniacs compared to normal sleepers, are not sleepier during the day, have increased level of high frequency EEG rhythm during sleep, have elevated brain metabolism during sleep and decreased during wakefulness in functional neuroimaging studies, have similar cognitive function and performance, and also have a 24-h activation of the HPA axis combined with daytime cytokine hypersecretion and /or circadian alteration of cytokines' secretion as well as, increased sympathetic activity during the night. Although insomnia is frequently associated with depression, the pathophysiology of the two disorders is different. Several psychological and physiological factors contribute to the onset and perpetuation of insomnia, such as anxious-ruminative personality traits, stressful events, age-related sleep homeostasis weakening mechanisms, and biologic – genetic diathesis of CNS hyperarousal. Evaluation of insomnia is multidimensional and factors such as aging, menopause, differentiation between fatigue and sleepiness, subtle psychopathology and medical comorbidity potentially associated with HPA axis activation should be considered. The therapeutic approach in insomnia should also be multidimensional and should aim to reduce the overall emotional and physiologic hyperarousal and its underlying factors present throughout the 24-h sleep/wake period.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCE
- 1.US Department of HEW . Selected symptoms of psychological distress. US Department of HEW; Rockville(MD): 1970. [Google Scholar]
- 2.Karacan I, Thornby JI, Anch M, Holtzer CE, Warheit GJ, Schwab JJ, et al. Prevalence of sleep disturbances in a primarily urban Florida county. Arch Gen Psychiatry. 1976;10:239–44. doi: 10.1016/0037-7856(76)90006-8. [DOI] [PubMed] [Google Scholar]
- 3.Bixler EO, Kales A, Soldatos CR, Kales JD, Healey S. Prevalence of sleep disorders in the Los Angeles metropolitan area. Am J Psychiatry. 136:1257–62. doi: 10.1176/ajp.136.10.1257. 10979. [DOI] [PubMed] [Google Scholar]
- 4.Mellinger GD, Balter MB, Uhlenhuth EH. Insomnia and it's treatment. Prevalence and correlates. Arch Gen Psychiatry. 1985;42:225–32. doi: 10.1001/archpsyc.1985.01790260019002. [DOI] [PubMed] [Google Scholar]
- 5.Klink M, Quan SF. Prevalence of reported sleep disturbances in a general adult population and their relationship to obstructive airways diseases. Chest. 1987;91:540–6. doi: 10.1378/chest.91.4.540. [DOI] [PubMed] [Google Scholar]
- 6.Klink M, Quan SF, Kaltenborn WT, Lebowits MD. Risk factors associated with complaints of insomnia in a general adult population. Arch Intern Med. 1992;152:1634–7. [PubMed] [Google Scholar]
- 7.Dodge R, Cline MG, Quan SF. The natural history of insomnia and its relationship to respiratory symptoms. Arch Intern Med. 1995;155:1797–800. [PubMed] [Google Scholar]
- 8.Foley DJ, Monjan AA, Brown SL, Simonsick EM, Wallace RB, Blazer DG. Sleep complaints among elderly persons: an epidemiologic study of three communities. Sleep. 1995;18(6):425–32. doi: 10.1093/sleep/18.6.425. [DOI] [PubMed] [Google Scholar]
- 9.US Department of Health and Human Services (DHHS) NHANES III laboratory data file. Centers for Disease Control and Prevention; Hyattsville(MD): 1996. National Center for Health Statistics. Third National Health and Nutrition Examination Survey, 1988-1994. [Google Scholar]
- 10.Ancoli-Israel S, Roth T. Characteristics of insomnia in the United States: results of the 1991 National Sleep Foundation Survey Sleep. 1999;22(Suppl 2):S347–53. [PubMed] [Google Scholar]
- 11.Ford DE, Kamerow DB. Epidemiologic study of sleep disturbances and psychiatric disorders. JAMA. 1989;262:1479–84. doi: 10.1001/jama.262.11.1479. [DOI] [PubMed] [Google Scholar]
- 12.Cortoos A, Verstraeten E, Cluydts R. Neurophysiological aspects of primary insomnia: implications for its treatment. Sleep Medicine Reviews. 2006;10:255–266. doi: 10.1016/j.smrv.2006.01.002. [DOI] [PubMed] [Google Scholar]
- 13.Ohayon MM. Prevalence of DSM-IV diagnostic criteria of insomnia: distinguishing insomnia related to mental disorders from sleep disorders. J Psychiatr Res. 1997;31:333–46. doi: 10.1016/s0022-3956(97)00002-2. [DOI] [PubMed] [Google Scholar]
- 14.Simon GE, Von Korff M. Prevalence, burden, and treatment of insomnia in primary care. Am J Psychiatry. 1997;154:1417–23. doi: 10.1176/ajp.154.10.1417. [DOI] [PubMed] [Google Scholar]
- 15.Chevalier H, Los F, Boichut D, Bianchi M, Nutt DJ, Hajak G, et al. Evaluation of severe insomnia in the general population : results of a Europeanmultinational survey. J Psychopharmacol. 1999;13:S21–S24. doi: 10.1177/026988119901304S04. [DOI] [PubMed] [Google Scholar]
- 16.American Psychiatric Association . Diagnostic and statistical manual of mental disorders, text revision. 4th ed American Psychiatric Association; Washington, DC: 2000. [Google Scholar]
- 17.Morin CM. Insomnia: psychological assessment and management. The Guilford Press; New York: 1993. [Google Scholar]
- 18.Roth T, Roehrs T. Insomnia: epidemiology, characteristics, and consequences. Clin Cornerstone. 2003;5:5–15. doi: 10.1016/s1098-3597(03)90031-7. [DOI] [PubMed] [Google Scholar]
- 19.Ohayon MM. Epidemiology of insomnia : what we know and what we still need to learn. Sleep Med Rev. 2002;6:97–111. doi: 10.1053/smrv.2002.0186. [DOI] [PubMed] [Google Scholar]
- 20.Kales A, Kales J. Evaluation and treatment of insomnia. Oxford University Press; New York: 1984. [Google Scholar]
- 21.Kales A, Caldwell AB, Soldatos CR, Bixler EO, Kales JD. Biopsychobehavioral correlates of insomnia II. Pattern specificity and consistency with the Minnesota Multiphasic Personality Invetory. Psychosomatic Med. 1983;45(4):341–5. doi: 10.1097/00006842-198308000-00008. [DOI] [PubMed] [Google Scholar]
- 22.Buysse DJ, Reynolds CF, 3rd, Kupfer DJ, Thorpy MJ, Bixler EO, Manfredi R, et al. Clinical diagnoses in 216 insomnia patients using the international classification of sleep disorders (ICSD), DSM-IV and ICD-10 categories: a report form the APA/NIMH DSM-IV trial. Sleep. 1994;17:630–637. doi: 10.1093/sleep/17.7.630. [DOI] [PubMed] [Google Scholar]
- 23.Breslau N, Roth T, Rosenthal L, Andreski P. Sleep disturbance and psychiatric disorders: a longitudinal epidemiological study of young adults. Biol Psychiatry. 1996;39:411–8. doi: 10.1016/0006-3223(95)00188-3. [DOI] [PubMed] [Google Scholar]
- 24.Summers MO, Crisostomo MI, Stepanski EJ. Recent developments in the classification, evaluation, and treatment of insomnia. Chest. 2006;130:276–86. doi: 10.1378/chest.130.1.276. [DOI] [PubMed] [Google Scholar]
- 25.Kales A, Bixler EO, Soldatos CR, Vela-Bueno A, Caldwell AB, Cardieux RJ. Biopsychobehavioral correlates of insomnia, part1: role of sleep apnea and nocturnal myoclonus. Psychosomatics. 1982;23(6):589–600. doi: 10.1016/S0033-3182(82)73359-6. [DOI] [PubMed] [Google Scholar]
- 26.Vgontzas AN, Kales A, Bixler EO, Manfredi RL, Vela-Bueno A. Usefulleness of polysomnographic studies in the differential diagnosis of insomnia. Int J Neurosci. 1995;82:47–60. doi: 10.3109/00207459508994289. [DOI] [PubMed] [Google Scholar]
- 27.Buysse DJ, Reynolds CF, III, Hauri PJ, Roth T, Stepanski EJ, Thorpy MJ, et al. Diagnostic concordance for insomnia patients among sleep specialists using proposed DSM-IV, proposed ICD-10, and ICSD diagnostic systems: report from the APA/NIMH DSM-IV field trial. In: Widiger TA, et al., editors. DSM-IV Sourcebook. Vol. 4. American Psychiatric Association; Washington, DC: pp. 869–889. [Google Scholar]
- 28.Kales A, Cadwell AB, Preston TA, Healey S, Kales JD. Personality patterns in insomnia. Theoretical implications. Arch Gen Psychiatry. 1976;33:1128–1134. doi: 10.1001/archpsyc.1976.01770090118013. [DOI] [PubMed] [Google Scholar]
- 29.Kales A, Soldatos K, Kales JD. Sleep disorders: insomnia, sleepwalking, night terrors, nightmares and enuresis. Ann Inter Med. 1987;106:582–92. doi: 10.7326/0003-4819-106-4-582. [DOI] [PubMed] [Google Scholar]
- 30.Bonnet MH, Arand DL. Insomnia, metabolic rate and sleep restoration. J Intern Med. 2003;254:23–31. doi: 10.1046/j.1365-2796.2003.01176.x. [DOI] [PubMed] [Google Scholar]
- 31.Vgontzas AN, Bixler EO, Lin HM, Prolo P, Mastorakos G, Vela-Bueno A, et al. Chronic insomnia is associated with nyctohemeral activation of the hypothalamic- pituitary – adrenal axis: clinical implications. J Clin Endocrinol Metab. 2001;86:3787–94. doi: 10.1210/jcem.86.8.7778. [DOI] [PubMed] [Google Scholar]
- 32.Rodenbeck A, Huether G, Ruether E, Hajak G. Interactions between evening and nocturnal cortisol secretion and sleep parameters in patients with severe chronic primary insomnia. Neurosci Lett. 2002;324:163–459. doi: 10.1016/s0304-3940(02)00192-1. [DOI] [PubMed] [Google Scholar]
- 33.Spielman AJ. Assessment of insomnia. Clin Psych Review. 1986;6:11–26. [Google Scholar]
- 34.Spielman AJ, Glovinsky PB. The varied nature of insomnia. In: Hauri PJ, editor. Case studies in insomnia. Plenum Press; New York: 1991. pp. 1–15. [Google Scholar]
- 35.Vgontzas AN. Understanding Insomnia in the Primary Care Setting: a new model. Insomnia Series. 2004;9(2):1–7. [Google Scholar]
- 36.Stepanski E, Zorick F, Roehrs T, Young D, Roth T. Daytime alertness in patients with chronic insomnia compared to asymptomatic controls subjects. Sleep. 1988;11(1):54–60. doi: 10.1093/sleep/11.1.54. [DOI] [PubMed] [Google Scholar]
- 37.Edinger JD, Glenn DM, Bastian LA, Marsh GR, Daile D, Hope V, et al. Sleep in laboratory and sleep at home II: comparisons of middle-aged insomnia sufferers and normal sleepers. Sleep. 2001;24(7):761–70. doi: 10.1093/sleep/24.7.761. [DOI] [PubMed] [Google Scholar]
- 38.Vgontzas AN, Zoumakis E, Papanicolaou DA, Bixler EO, Prolo P, Lin HM, et al. Chronic insomnia is associated with a shift of interleukin-6 and tumor necrosis factor secretion from nighttime to daytime. Metabolism. 2002;51(7):887–92. doi: 10.1053/meta.2002.33357. [DOI] [PubMed] [Google Scholar]
- 39.Vgontzas AN, Zoumakis E, Bixler EO, Lin HM, Follet H, Kales A, et al. Adverse effects of modest sleep restriction on sleepiness, performance, and inflammatory cytokines. JCME. 2004;89:2119–2126. doi: 10.1210/jc.2003-031562. [DOI] [PubMed] [Google Scholar]
- 40.Vgontzas AN, Pejovic S, Zoumakis E, Lin HM, Bixler EO, Basta M, et al. Daytime napping after a night on sleep loss decreases sleepiness, improves performance, and causes beneficial changes in cortisol and interleukin-6 secretion. Am J Physiol Endocrin Metab. 2007;292:253–261. doi: 10.1152/ajpendo.00651.2005. [DOI] [PubMed] [Google Scholar]
- 41.Bonnet MH, Arand DL. 24-hour metabolic rate in insomniacs and matched normal sleepers. Sleep. 1995;18(7):581–88. doi: 10.1093/sleep/18.7.581. [DOI] [PubMed] [Google Scholar]
- 42.Riedel BW, Lichstein KL. Insomnia and daytime functioning. Sleep Medicine Reviews. 2000;4(3):277–98. doi: 10.1053/smrv.1999.0074. [DOI] [PubMed] [Google Scholar]
- 43.Monroe LJ. Psychological and physiological differences between good and poor sleepers. J Abnorm Psychol. 1967;72:255–64. doi: 10.1037/h0024563. [DOI] [PubMed] [Google Scholar]
- 44.Freedman RR, Sattler HL. Physiological and psychological factors in sleep-onset insomnia. J Abnorm Psychol. 1982;91:380–9. doi: 10.1037//0021-843x.91.5.380. [DOI] [PubMed] [Google Scholar]
- 45.Landis CA, Savage MV, Lentz MJ, Brengelmann GL. Sleep deprivation alters body temperature dynamics to mild cooling and heating not sweating threshold in women. Sleep. 1998;21(1):101–8. doi: 10.1093/sleep/21.1.101. [DOI] [PubMed] [Google Scholar]
- 46.Muenter NK, Watenpaugh DE, Wasmund WL, Wasmund SL, Maxwell SA, Smith ML. Effect of sleep restriction on orthostatic cardiovascular control in humans. J Appl Physiol. 2000;88(3):966–72. doi: 10.1152/jappl.2000.88.3.966. [DOI] [PubMed] [Google Scholar]
- 47.Kato M, Phillips BG, Sigurdsson G, Narkiewicz K, Pesek CA, Somers VK. Effects of sleep deprivation on neural circulatory control. Hypertension. 2000;35:1173–5. doi: 10.1161/01.hyp.35.5.1173. [DOI] [PubMed] [Google Scholar]
- 48.De Gennaro L, Ferrara M, Bertini M. The boundary between wkefulness and sleep : quantitative electroencephalographic changes during the sleep onset period. Neuroscience. 2001;107:1–11. doi: 10.1016/s0306-4522(01)00309-8. [DOI] [PubMed] [Google Scholar]
- 49.Lamarche CH, Ogilvie RD. Electrophysiological changes during sleep onset of psychophysiological insomniacs, psychiatric insomniacs, and normal sleepers. Sleep. 1997;20:724–33. [PubMed] [Google Scholar]
- 50.Staner L, Cornette F, Maurice D, Viardot G, Le Bon O, Haba J, et al. Sleep microstructure around sleep onset differentiates major depressive insomnia from primary insomnia. J Sleep Res. 2003;12:319–30. doi: 10.1046/j.0962-1105.2003.00370.x. [DOI] [PubMed] [Google Scholar]
- 51.Perlis ML, Smith MT, Andrews PJ, Orff H, Giles DE. Beta / gamma EEG activity in patients with primary and secondary insomnia and good sleeper controls. Sleep. 2001;24:110–7. doi: 10.1093/sleep/24.1.110. [DOI] [PubMed] [Google Scholar]
- 52.Merica H, Blois R, Gaillard JM. Spectral characteristics of sleep EEG in chronic insomnia. Eur J Neurosci. 1998;10:1826–34. doi: 10.1046/j.1460-9568.1998.00189.x. [DOI] [PubMed] [Google Scholar]
- 53.Nofzinger EA, Buysse DJ, Miewald JM, Meltzer CC, Price JC, Sebrat RC, et al. Human regional cerebral glucose metabolism during non rapid eye movement sleep in relation to waking. Brain. 2002;125(part 5):1105–1115. doi: 10.1093/brain/awf103. [DOI] [PubMed] [Google Scholar]
- 54.Thomas M, Sing H, Belenky G, Holcomb H, Mayberg H, Dannals R, et al. Neural basis of alertness and cognitive performance impairments during sleepiness, I: effects of 24h of sleep deprivation on waking human regional brain activity. J Sleep Res. 2000;9:335–52. doi: 10.1046/j.1365-2869.2000.00225.x. [DOI] [PubMed] [Google Scholar]
- 55.Nofzinger EA, Buysse DJ, Germain A, Price JC, Miewald JM, Kupfer DJ. Functional neuroimaging evidence for hyperarousal in insomnia. Am J Psychiatry. 2004;161:2126–29. doi: 10.1176/appi.ajp.161.11.2126. [DOI] [PubMed] [Google Scholar]
- 56.Fulda S, Schulz H. Cognitive dysfunction in sleep disorders. Sleep Medicine. 2001;5(6):423–445. doi: 10.1053/smrv.2001.0157. [DOI] [PubMed] [Google Scholar]
- 57.Van Dongen HP, Maislin G, Mullington JM, Dinges DF. The cumulative cost of additional wakefulness: dose-response effects on neurobehavioral functions and sleep physiology from chronic sleep restriction and total sleep deprivation. Sleep. 2003;2:117–26. doi: 10.1093/sleep/26.2.117. [DOI] [PubMed] [Google Scholar]
- 58.Adams K, Tomeny M, Oswald I. Physiological and psychological differences between good and poor sleepers. J Psychiatr Res. 1986;20:301–16. doi: 10.1016/0022-3956(86)90033-6. [DOI] [PubMed] [Google Scholar]
- 59.Vgontzas AN, Tsigos C, Bixler EO, Stratakis CA, Zachman K, Kales A, et al. Chronic insomnia and activity of the stress system: a preliminary study. J Psychosom Res. 1998;45(1):21–31. doi: 10.1016/s0022-3999(97)00302-4. [DOI] [PubMed] [Google Scholar]
- 60.Irwin M, Clark C, Kennedy B, Gillin C, Ziegler M. Nocturnal catecholamines and immune function in insomniacs, depressed patients, and controls subjects. Brain Behavior Immunity. 2003;17:365–72. doi: 10.1016/s0889-1591(03)00031-x. [DOI] [PubMed] [Google Scholar]
- 61.Moldofsky H, Lue FA, Davidson JR, Gorczynski R. Effects of sleep deprivation on human immune functions. FASEB J. 1989;3:1927–77. doi: 10.1096/fasebj.3.8.2785942. [DOI] [PubMed] [Google Scholar]
- 62.Scheen AJ, Byrne MM, Plat L, Leproult R, Van Cauter E. Relationships between sleep quality and glucose regulation in normal humans. Am J Physiol. 1997;271:E261–E270. doi: 10.1152/ajpendo.1996.271.2.E261. [DOI] [PubMed] [Google Scholar]
- 63.Brun J, Chambe G, Khalfallah Y, Girard P, Boissy I, Bastuji H, et al. Effect of modafinil on plasma melatonin, cortisol and growth hormone rhythms, rectal temperature and performance in healthy subjects during 36h sleep deprivation. J Sleep Res. 1998;7:105–114. doi: 10.1046/j.1365-2869.1998.00100.x. [DOI] [PubMed] [Google Scholar]
- 64.Vgontzas AN, Mastorakos G, Bixler EO, Kales A, Gold PW, Chrousos GP. Sleep deprivation effects on the activity of hypothalamic –pituitary- adrenal and growth axes: potential clinical implications. Clin Endocrinol (Oxf) 1999;51:205–15. doi: 10.1046/j.1365-2265.1999.00763.x. [DOI] [PubMed] [Google Scholar]
- 65.Leproult R, Copinschi G, Buxton O, Van Cauter E. Sleep loss results in an elevation of cortisol levels the next evening. Sleep. 1997;20:865–70. [PubMed] [Google Scholar]
- 66.Spiegel K, Lerpoult R, Van Cauter E. Impact of sleep debt on metabolic and endocrine function. Lancet. 1999;354:1435–1439. doi: 10.1016/S0140-6736(99)01376-8. [DOI] [PubMed] [Google Scholar]
- 67.Rodenberg A, Cohrs S, Jordan W, Huether G, Ruether E, Hajak G. The sleep-improving effects of doxepin are paralleled by a normalized plasma cortisol secretion in primary insomnia. Psychopharmacology. 2003;170:423–28. doi: 10.1007/s00213-003-1565-0. [DOI] [PubMed] [Google Scholar]
- 68.Vgontzas AN, Papanicolaou DA, Bixler EO, Lotsikas A, Zachman K, Kales A, et al. Circadian interleukin-6 secretion and quantity and depth of sleep. J Clin Endocrinol Metab. 1999;84:2603–7. doi: 10.1210/jcem.84.8.5894. [DOI] [PubMed] [Google Scholar]
- 69.Vgontzas AN, Bixler EO, Wittman AM, Zachman K, Lin HM, Vela-Bueno A, et al. Middle-aged men show higher sensitivity of sleep to the arousing effects of corticotrophin-releasing hormone that young men: clinical implications. J Clin Endocrinol Metab. 2001;86:1489–95. doi: 10.1210/jcem.86.4.7370. [DOI] [PubMed] [Google Scholar]
- 70.Bixler EO, Vgontzas AN, Lin HM, Vela-Bueno A. Menopause and Hormone Replacement therapy: effects in objective sleep patterns; 21st annual meeting of the APSS 2007; Minneapolis. [Google Scholar]
- 71.Lugaresi E, Zucconi M, Bixler EO. Epidemiology of sleep disorders. Psychiatric Ann. 1987;17:446–453. [Google Scholar]
- 72.Vgontzas AN, Bixler EO, Kales A, Manfredi RL, Tyson K. Validity and clinical utility of sleep laboratory criteria for insomnia. Inter J Neuroscience. 1994;77:11–21. doi: 10.3109/00207459408986015. [DOI] [PubMed] [Google Scholar]
- 73.Thase ME. Treatment and issues related to sleep and depression. J Clin Psychiatry. 2006;61(suppl 11):46–50. [PubMed] [Google Scholar]
- 74.Chrousos GP, Gold PW. The concepts of stress and stress system disorders. Overview of physical and behavioral homeostasis. JAMA. 1992;267:1244–52. [PubMed] [Google Scholar]
- 75.Gold PW, Goodwin FK, Chrousos GP. Clinical and biochemical manifestations of depression: relationship to the neurobiology of stress (part 1) N Eng J Med. 1988;319:348–53. doi: 10.1056/NEJM198808113190606. [DOI] [PubMed] [Google Scholar]
- 76.Gold PW, Goodwin FK, Chrousos GP. Clinical and biochemical manifestations of depression: relationship to the neurobiology of stress (part 2) N Eng J Med. 1988;413:20. doi: 10.1056/NEJM198808183190706. [DOI] [PubMed] [Google Scholar]