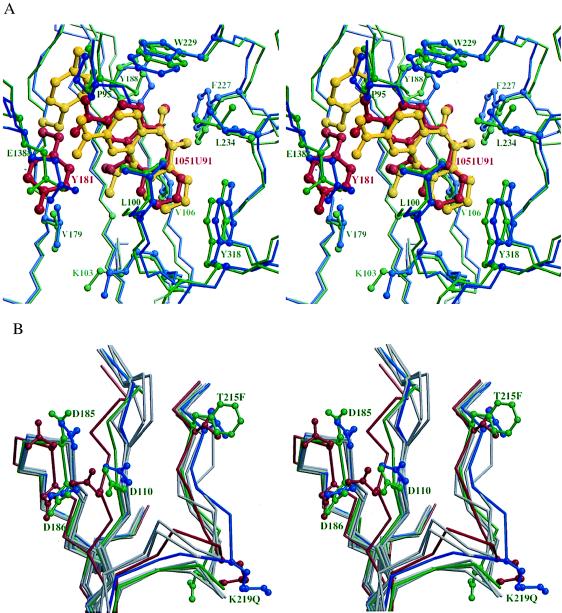
Figure 3.
The NNI binding site and polymerase active site. (a) A stereodiagram showing the superposition of the NNI binding site in RTMC and wild-type RT. The protein backbone is shown by thin sticks. The NNIs (thick bonds) and side chains that have contacts with the NNIs are shown as ball-and-stick representations. The RTMC is colored in green with residue 181 and the bound 1051U91 highlighted in red. The wild-type RT is colored in blue with residue 181 and bound 1051U91 highlighted in yellow. (b) A stereodiagram of the superposition of the active sites in RTMC (green), the wild type unliganded (red), and six NNI-bound RT structures (blue for RT-1051U91, gray for others) showing the structural changes at the active site in RTMC caused by 215 and 219 mutations. The Cα trace and side chains for residues 110, 185, 186, 215, and 219 are shown for RTMC, wild-type unliganded RT, and RT-1051U91; the Cα traces only are shown for RT-Cl-TIBO, RT-BHAP, RT-nevirapine, RT-MKC-442, and RT-α-APA. In the p51 subunit, residues 215 and 219 are disordered whereas residues 67 and 70 do not show significant rearrangement from the wild-type p51.