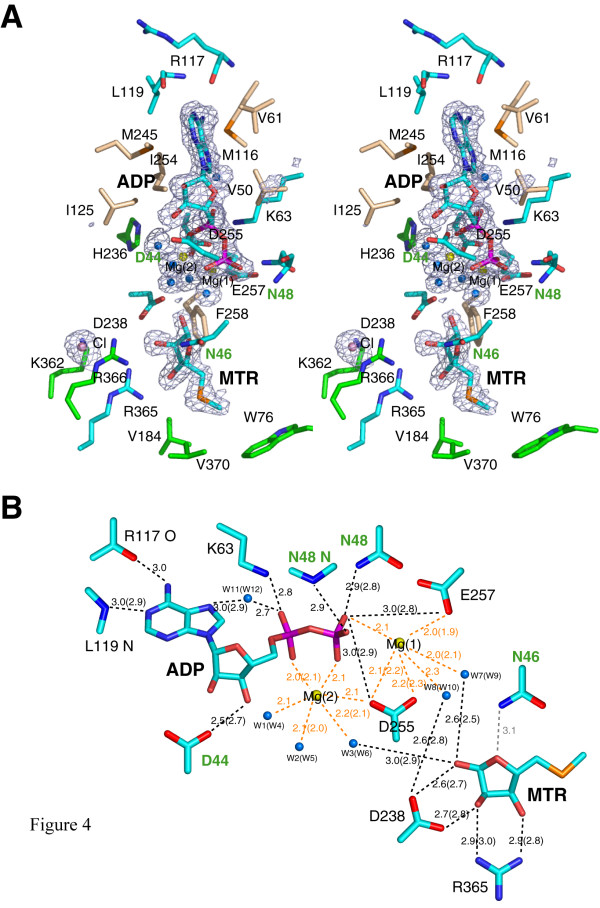
Figure 4.
The active site of A. thaliana MTR kinase. (a) Stereo representation of the active site. The product ADP and the substrate MTR and nearby interacting residues are drawn in stick representation with C, N, O, P and S atoms coloured in cyan, blue, red, magenta, and orange, respectively. Hydrophobic residues are shown with their C atoms coloured in wheat. Residues that do not directly interact with ADP or MTR but are discussed in the text are shown with their C atoms coloured in green. The magnesium ions, chloride ion and water molecules are shown as yellow, violet and blue spheres, respectively. The σA-weighted Fo-Fc ligand (and water) omit map in blue mesh was calculated using CCP4 [52] and is contoured at 3σ. (b) Schematic diagram showing the hydrophilic interactions less than 3 Å between the ligands and the protein. The colour schemes of the atoms are as described in Panel (a). The distances in Å between the ligands and the protein are for monomer A while those for monomer B are in parentheses. If the two distances are the same, only one value is shown. The distances around the two octahedrally coordinated Mg(II) ions are labelled in orange. Residues from the G-loop are labelled in green. The distance between the Nδ amide nitrogen of N46 in the G-loop and the O4 of MTR is greater than 3 Å and hence is shown in grey instead of black. Both panels were prepared using PyMOL [51].