Figure 4.
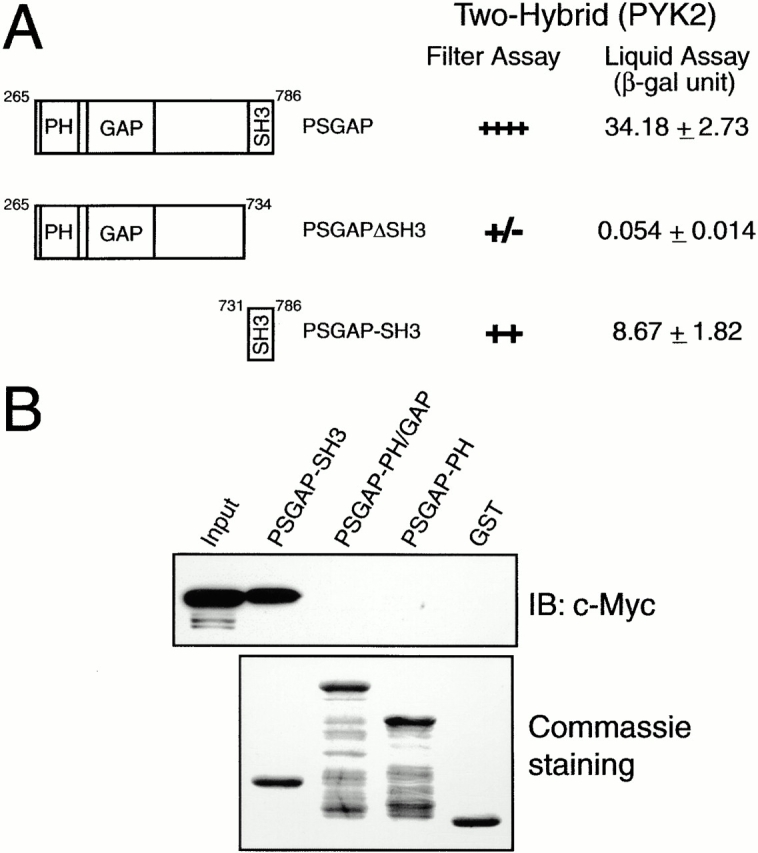
SH3 domain of PSGAP is essential for the interaction with PYK2. (A) Mapping of the binding region in PSGAP by yeast two-hybrid assays. Yeast cells were cotransformed with a vector encoding the Gal4 binding domain fused to different PSGAP constructs and Gal4AD fused to PYK2. Transformed yeast cells were seeded in Leu−, Trp−, and His− plates and assayed for β-Gal activity. Transformed yeast cells were also grown in Leu− and Trp− medium for liquid β-Gal assays. (B) Analysis of PSGAP binding to PYK2 by in vitro GST pull-down assays. HEK293 cells were transfected with Myc-tagged PYK2. Lysates of transfected cells were incubated with various GST-PSGAP fusion proteins (SH3, amino acids 731–786; PH-GAP, amino acids 265–590; and PH, amino acids 265–454) immobilized on beads. Lysate input and bound proteins were resolved on SDS-PAGE and subjected to immunoblotting with an anti–Myc antibody (top). An equal amount of GST fusion proteins was used as revealed by Coomassie staining (bottom).
