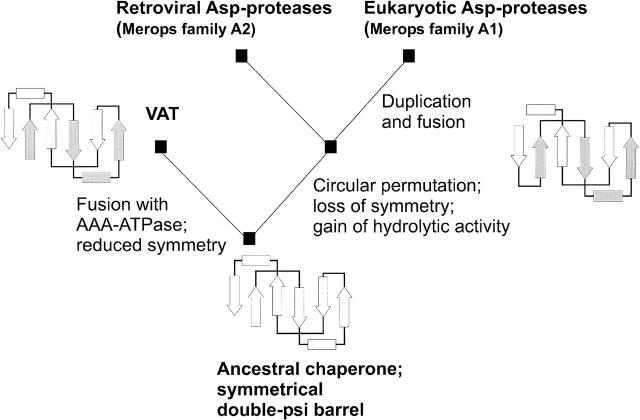
Figure 7.
Scenario of Cdc48 N-terminal domain-like double-ψ barrel and aspartic protease evolution. The scenario originates with an ancestral chaperone showing a symmetrical double-ψ barrel fold. The left branch leads to AAA-ATPases by fusion with AAA-domains, e.g., in VAT. The right branch leads to circular permutation, loss of internal symmetry, and gain of hydrolytic activity. Further branching leads to the retroviral proteases like HIV-1 protease, and via gene duplication and fusion, to the eukaryotic aspartic proteases like pepsin.