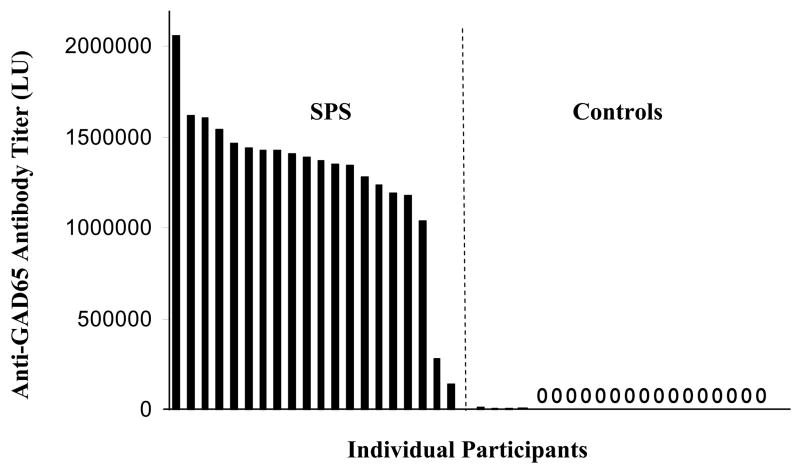
Fig. 2.
Detection of patient antibodies to GAD65. Forty blinded sera samples from controls and SPS patients were utilized to evaluate anti-GAD65 antibody titers. Sera were analyzed using LIPS in two independent assays, the values averaged, and samples rank ordered from high to low titers corresponding to left to right, respectively. By analyzing the anti-GAD65 antibody titers of the 40 coded sera, the samples were statistically segregated (P-value of 3.3 × E−16) into two groups: 20 sera with a high anti-GAD65 antibody titer (left panel) and 20 sera with a low anti-GAD65 antibody titer (right panel). Sixteen of the samples showed values of zero (0). For SPS disease status, samples on the left panel with high anti-GAD65 antibody titer were predicted as positive. Following unblinding, the exact disease status of the 40 sera samples was revealed, showing that the LIPS data predicted SPS status from controls with 100% sensitivity and 100% specificity.