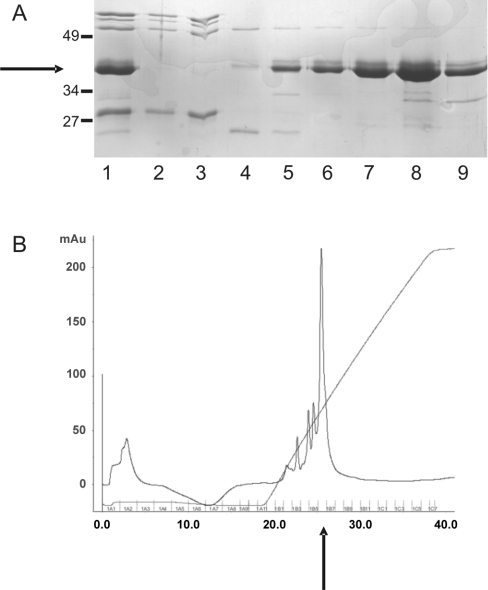
Figure 1. Purification of His–centerin.
Bacterial lysate containing recombinant His–centerin was chromatographed on a nickel-HisTrap column and the pooled eluate (A; lane 1) was then applied to a mono-S cation-exchange column. (A) The Figure shows flow through (lanes 2 and 3) and fractions eluted with a 0.125–1 M NaCl gradient (lanes 4–9) (Coomassie-stained SDS/PAGE gel). Recombinant His–centerin is arrowed on left. Molecular mass standards are shown to the left of the Figure. Sizes are in kDa. (B) The Figure shows a protein elution profile [A280 (absorbance)] from mono-S cation-exchange chromatography step. The major protein peak (arrowed) corresponds to fraction 8 (A).